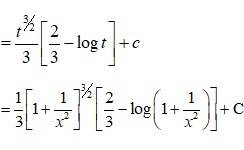Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
(a) Frequency of emission, ν = c/λ = (3.0 x 108 ms-1) / (616 x 10-9 m) = 4.87 x 1014 s-1
(b) Speed of radiation, c = 3 x 108 ms-1
Distance travelled by this radiation in 30s = 3 x 108 ms-1 x 30 s = 9.0 x 109 m
(c) Energy of quantum, E = hν =hc/λ = [ (6.626 x 10-34Js) x (3 x 108 ms-1)] / (616 x 10-9 m) = 32.27 x 10-20 J
(d) Number of quanta present if it produces 2 J of energy
&nbs
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
If arenes would undergo electrophilic addition reaction then they would lose their aromaticity which would lead to comparatively less stability. However, alkenes undergo electrophilic addition reaction via carbocation intermediate.
New Question
10 months agoContributor-Level 10
Power of the laser E = Nhv = Nh c/λ, where N is the number of photos emitted
= [ (5.6 x 1024) x (6.626 x 10-34Js) x (3 x 108 ms-1)] / (337.1 x 10-9 m)
= 3.3 x 106 J
New Question
10 months agoContributor-Level 10
Let I =
=
=
=
=
Putting tan2x = t =>2 tan x?sec2xdx = dt
When x = 0, t = tan2x = tan2 0 = 0
x = t = tan2 = 12 = 1.
? I =
=
=
New Question
10 months agoContributor-Level 10
The Sasurie College of Engineering is an institution known for its BE/BTech programme and its placements. Check out the table below to know about the BE/BTech placement information recorded at Sasurie College of Engineering placements during 2023-2025:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
the highest Package | INR 4.7 LPA (BE CSE) INR 4.1 LPA (BE ECE) INR 4.7 LPA (BE EE) INR 3.7 LPA (BE ME) INR 4.6 LPA (BE CE) | INR 4.3 LPA (BE CSE) INR 4.4 LPA (BE ECE) INR 4.3 LPA (BE EE) INR 4.3 LPA (BE ME) INR 4.5 LPA (BE CE) INR 4.6 LPA (BTech AIDS) | INR 4.7 LPA (BE CSE) INR 4.5 LPA (BE ECE) INR 4.5 LPA (BE EE) INR 4.5 LPA (BE ME) INR 4.5 LPA (BE CE) INR 4.7 LPA (BTech AIDS) |
the lowest Package | INR 2.7 LPA (BE CSE) INR 2.5 LPA (BE ECE) INR 2.7 LPA (BE EE) INR 2.4 LPA (BE ME) INR 2 LPA (BE CE) | INR 2.3 LPA (BE CSE) INR 2.2 LPA (BE ECE) INR 2.3 LPA (BE EE) INR 2.6 LPA (BE ME) INR 2.4 LPA (BE CE) INR 2.5 LPA (BTech AIDS) | INR 2.7 LPA (BE CSE) INR 2.3 LPA (BE ECE) INR 2.4 LPA (BE EE) INR 2.7 LPA (BE ME) INR 2.7 LPA (BE CE) INR 2.6 LPA (BTech AIDS) |
Placement Rate | 85% | 90% | 90% |
New Question
10 months agoNew Question
10 months agoContributor-Level 10
The given radiations in increasing order of wavelength are:
Cosmic rays < X-rays < radiation from microwave oven < amber light from traffic signal < radiation from FM radio
New Question
10 months agoNew Question
10 months agoContributor-Level 10
IILET 2026 application forms window is closed. Candidates will have to submit an online application form along with the required documents including qualifying exam details, category, residential address, contact details, etc.
New Question
10 months agoContributor-Level 10
Let the no. of electrons in the ion= x
∴ the no. of the protons= x + 3 (as the ion has three units positive charge)
and the no. of neutrons= x + (x×30.4 / 100) = x+ 0.304 x
Now, mass no. of ion = No. of protons + No. of neutrons = (x + 3) + (x + 0.304x)
∴ 56 = (x + 3) + (x + 0.304x)
=>2.304x = 56 – 3 = 53
=>x = 53 / 2.304 = 23
Atomic no. of the ion (or element) = 23 + 3 = 26
The element with atomic number 26 is iron (Fe) and the corresponding ion is Fe3+.
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
The bond energy of HCl is higher than that of HBr thus it is not cleaved by free radical mechanism to exhibit peroxide effect. However in case of HI the bond energy is so low that the iodine radical forms readily and after formation it combines to form an iodine molecule.
New Question
10 months agoContributor-Level 10
Sasurie College of Engineering placement report for BE/BTech is out on the official website. Check out the table below to know the key highlights of the Sasurie College of Engineering placements for BE/BTech between 2023 to 2025:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
the highest Package | INR 4.7 LPA (BE CSE) INR 4.1 LPA (BE ECE) INR 4.7 LPA (BE EE) INR 3.7 LPA (BE ME) INR 4.6 LPA (BE CE) | INR 4.3 LPA (BE CSE) INR 4.4 LPA (BE ECE) INR 4.3 LPA (BE EE) INR 4.3 LPA (BE ME) INR 4.5 LPA (BE CE) INR 4.6 LPA (BTech AIDS) | INR 4.7 LPA (BE CSE) INR 4.5 LPA (BE ECE) INR 4.5 LPA (BE EE) INR 4.5 LPA (BE ME) INR 4.5 LPA (BE CE) INR 4.7 LPA (BTech AIDS) |
the lowest Package | INR 2.7 LPA (BE CSE) INR 2.5 LPA (BE ECE) INR 2.7 LPA (BE EE) INR 2.4 LPA (BE ME) INR 2 LPA (BE CE) | INR 2.3 LPA (BE CSE) INR 2.2 LPA (BE ECE) INR 2.3 LPA (BE EE) INR 2.6 LPA (BE ME) INR 2.4 LPA (BE CE) INR 2.5 LPA (BTech AIDS) | INR 2.7 LPA (BE CSE) INR 2.3 LPA (BE ECE) INR 2.4 LPA (BE EE) INR 2.7 LPA (BE ME) INR 2.7 LPA (BE CE) INR 2.6 LPA (BTech AIDS) |
Placement Rate | 85% | 90% | 90% |
New Question
10 months agoContributor-Level 10
Let the no. of electron in the ion = x
∴ The no. of protons = x – 1 (as the ion has one unit negative charge)
and the no. of neutrons = x + (x×11.1 / 100) = 1.111 x
Mass of the ion = No. of protons + No. of neutrons
(x – 1) + (1.111 x)
Given mass of the ion = 37
∴ (x – 1) + (1.111 x) = 37
=> 2.111 x = 37 + 1 = 38
x = 38 / 2.111 = 18
No. of electrons = 18; No. of protons = 18 – 1 = 17
Atomic no. of the ion = 17; atom corresponding to ion = Cl
Symbol of the ion = 3717Cl–
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
The structure of A is
The reaction involved are as follows:
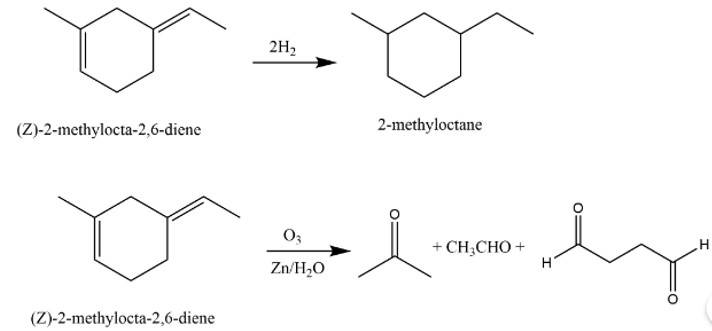
New Question
10 months agoContributor-Level 10
An element can be identified by its atomic number only. Let us find the atomic number.
Let the number of protons = x
Number of neutrons = x + (x×31.7/100) = 1.371x
Now, Mass no. of element = no. of protons + no. neutrons
⇒ x + 1.317x = 81
⇒ x = 34.958
x ? 35
∴ No. of protons = 35, No. of neutrons = 81 – 35 =46
Atomic number of element (Z) = No. of protons = 35
The element with atomic number (Z) 35 is bromine (3579Br)
New Question
10 months agoContributor-Level 10
Some the top recruiters for Sasurie College of Engineering placement are listed below:
Sasurie College of Engineering Top Recruiters | ||
|---|---|---|
Cognizant | HP | Hexaware |
HCL | L & T | Tech Mahindra |
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts