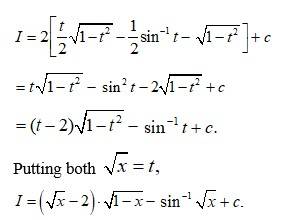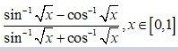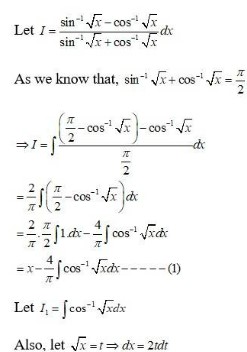Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 7
Yes, students can use old GRE books for their preparation. Concepts of math and verbal section concepts, and question types, haven't changed much in the year 2025. However, be aware that the GRE duration is now 1 hour and 58 minutes. Old books won't reflect the new, shorter test format or the removal of one essay in Analytical Writing Assesement section.
New Question
10 months agoContributor-Level 10
The application fee for the BSc prorgamme at Whistling Woods International (WWI) is INR 3,500. Additionally, the tuition fee for the whole course duration is INR 8.5 lakh. However, the mentioned fee is taken from the official website/ sanctioning body. It is still subject to change and, hence, is indicative.
New Question
10 months agoContributor-Level 10
The question paper of IILET have sections such as English Language and GK & Current Affairs. The questions from Legal and Logical Reasoning and Quantitative Techniques will be asked. On the basis scores, the selected candidates will have to appear for the personal interview rounds.
New Question
10 months agoContributor-Level 10
Length of scale = 20 cm = 20 x 107 nm = 2 x 108 nm
Diameter of carbon atom = 0.15 nm
∴ Number of carbon atoms which can be placed side by side in a straight line across a length of a scale of length 20 cm = (2 x 108 nm) / (0.15 nm) = 1.33 x 109
New Question
10 months agoContributor-Level 10
The expression for the ionization energy atom,
En = (2.18 * 10-18 x Z2) / n2 J atom-1
For H atom, (Z = 1). So,
En =2.18 * 10-18 * (l)2 J atom-1 (given)
For He+ ion (Z = 2). So,
En =2.18 * 10-18 * (2)2 = 8.72 * 10-18 J atom-1 (one electron species)
New Question
10 months agoContributor-Level 9
Dronacharya Group of Institutions is a private college and was established in 2006. The college is approved by the All-India Council for Technical Education (AICTE) and is aimed at imparting high-quality education in the field of Engineering and Management. Further, the college has been recognised in the band 'Excellent' by ARIIA, MHRD, Govt of India.
New Question
10 months agoContributor-Level 10
For an atom? = 1/ λ = RH Z2 [ (1/n12) – (1/n22)]
For He+ spectrum: Z = 4, n2 = 4, n1 = 2.
∴? = 1/ λ = RH Z2 [ (1/22) – (1/42)]
= RH 22 [ (1/22) – (1/42)]
= 3RH /4
For hydrogen spectrum:
∴? = 1/ λ = RH 12 [ (1/n12) – (1/n22)] = 3RH /4
=> (1/n12) – (1/n22) = 3/4
This corresponds to n1 = 1 and n2 =2 and means that the transition has taken place in the Lyman series from n = 2 to n =1.
New Question
10 months agoContributor-Level 10
IILET exam is conducted by the Indore Institute of Law (IIL). This exam is conducted for admission to five-year integrated BBA LLB (Hons.) with Global & Transnational Studies, BA LLB and BBA LLB courses. The IILET 2026 application could be filled on the official website, indoreinstituteoflaw.org.
New Question
10 months agoContributor-Level 10
The overall vibe of LSR is quite good. Being a part of the Delhi University and located in the North Campus, makes the college a preferred one. LSR College is spread across 15 acres of land and has a cafeteria, auditorium, girls hostel, gym, hospital / medical facilities, shuttle service, sports complex, hostel, labs, library, and wi-fi campus. The Lady Shri Ram College also provides various other facilities to postgraduate students. The college arranges various fests and seminars for students.
New Question
10 months agoContributor-Level 10
According to Bohr’s theory,
mvr = nh / 2π
=> 2πr = nh/mv
=> mv = nh / 2πr - (i)
According to de Broglie equation,
h / λ - (ii)
Comparing equations (i) and (ii)
nh / 2πr = h / λ
=> 2πr = n λ
Thus, the circumference (2πr) of the Bohr orbit for hydrogen atom is an integral multiple of the de Broglie wavelength.
New Question
10 months agoContributor-Level 6
There is no requirement for a passport copy when registering for SAT exam. Applicants only need to fill in their details - academic, coursework and personal. This is followed by optional photo uploading (can be uploaded later on also), and paying for the exam. The passport will be required at the test centre for identification.
New Question
10 months agoContributor-Level 10
Lady Shri Ram College for Women (LSR) offers placement assistance to students. Lady Shri Ram College for Women invites top recruiters to take part in the placement drive conducted by LSR DU. The below-mentioned are the Lady Shri Ram College for Women (LSR) placement statistics:
| Particulars | Statistics 2025 |
|---|---|
| the highest package | INR 45 LPA |
| Average package | INR 12.18 LPA |
| Median package | INR 10.2 LPA |
New Question
10 months agoContributor-Level 9
The University of Edinburgh is far better than the University of Dundee based on the global, country rankings. The University of Edinburgh ranked #38 among the best global universities, where the University of Dundee ranked #362. Students can check the comparison between the University of Edinburgh and the University of Dundee below:
Particulars (US news 2025) | University of Dundee | University of Edinburgh |
|---|---|---|
Best Global Universities | #362 | #38 |
Best Universities in Europe | #140 | #8 |
Best Global Universities in the UK | #31 | #6 |
New Question
10 months agoContributor-Level 10
(a) For n = 4
Total number of electrons = 2n2 = 2 × 16 = 32
Half out of these will have ms = -1/2
∴ Total electrons with ms (-1/2) = 16
(b) For n = 3
l= 0; ml = 0, ms +1/2, -1/2 (two e–)
New Question
10 months agoContributor-Level 9
UDundee offers various popular programs especially in fields like Science Medicine Arts and Design. As per unofficial source university ranked within top 10 in UK for subjects like Anatomy and Physiology Dentistry, Social Work etc. The university also provides strong business and economics programs along with CS, Nursing, and Architecture.
New Question
10 months agoContributor-Level 9
Dronacharya Group of Institutions application process is available in online mode only. Applicants need to visit the official website of DGI, Greater Noida to register themselves. Once the application form is filled and submitted, an application fee needs to be paid via online mode only. Mentioned below is a step-by-step process of filling out the application form:
Step 1: Visit the official website of DGI, Greater Noida (gnindia.dronacharya.info) and click on 'Apply Now' (if applying via desktop/laptop). Students applying via mobile phones need to click on the menu tab followed by clicking on 'Admission' and then on 'Apply Online'
New Question
10 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts