Ask & Answer: India's Largest Education Community
All Questions
New Question
6 months agoContributor-Level 10
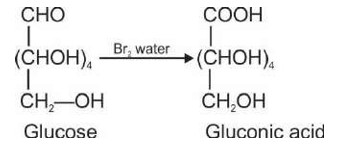
The carbonyl compounds which contain at least two a-hydrogen will give aldol condensation.
New Question
6 months agoContributor-Level 10
There exist different types of cancer, some of which have a remedy to be treated and some of which may never have in the near future. Technologies have advanced, the field of Science and biomedical sector have witnessed significant improvements in the previous decades. Many cancers which seemed impossible to be treated years ago have today a reliable cure. So it depends on which type of cancer and which stage of cancer are you specifically talking about.
New Question
6 months agoContributor-Level 7
KARTET syllabus is prescribed by the examination authority. KARTET Paper 1 syllabus comprises five sections-Child Development and Pedagogy, Language 1, Language 2, Mathematics, and Environmental Science. KARTET Paper 2 Syllabus comprises four sections, Child Development and Pedagogy, Language 1, Language 2, Mathematics, and Science or Social Science.
New Question
6 months agoContributor-Level 10
Yes. Indeed there are various scientists who are trained to prepare and handle these carcinogens safely in a laboratory. These carcinogens are used to prepare various useful substances such as drugs and medicines so their usage needs to be monitored regularly and handled carefully. The production must comply with legal rules and standards.
New Question
6 months agoContributor-Level 7
The process to download KARTET hall ticket is given below. The hard copy of the admit card is not sent to candidates by post.
Click the admit card download link
Enter the required credentials
Admit card appears on the screen
Download and save it
New Question
6 months agoContributor-Level 8
KARTET admit card 2025 is released a week before the exam. Candidates can download their admit cards online by logging with the required credentials.
New Question
6 months agoContributor-Level 10
Kindly go through the solution
B2 ->s1s2 s*1s2 s2s2 s*2s2 p2p1= p2p1
New Question
6 months agoContributor-Level 10
Ion | l°/S cm2 mol–1 |
OH– | 199.1 |
SO2- 4 | 160.0 |
Cl– | 76.3 |
CH3COO– | 40.9 |
Order of limiting molar conductivity
OH- > SO42- > Cl- > CH3COO-
New Question
6 months agoContributor-Level 10
Higher the acidic strength, lower will be the value of pKa.
Therefore, Correct order of pKa is
CH3COOH > C6H5COOH > HCOOH > O2NCH2COOH
New Question
6 months agoContributor-Level 10
The following are the real-world applications of electrochemistry - military applications such as thermal batteries, digital watches, hearing aids, digital cameras, electrical appliances such as cellphones, and torches.
New Question
6 months agoNew Question
6 months agoContributor-Level 10
It depends on students. Though it is not a tough chapter to study but for students who have misconceptions and those who struggle with visualization can find it challenging.
New Question
6 months agoContributor-Level 10
There are two types of electrochemical cells - Electrolytic and Galvanic or Voltaic cells. The electrolytic cells need an external source such as AC power source or DC battery and it involve non-spontaneous reactions. The galvanic cells gets its energy from redox reactions which is spontaneous.
New Question
6 months agoContributor-Level 10
Redox reactions is the basic principle of the electrochemistry. The redox reactions is the process where electrons are transferred between substances. In this process chemical energy gets converted into electrical energy and vice versa.
New Question
6 months agoContributor-Level 10
[Co (NH3)6]3+, NH3 becomes strong ligand due to +3 oxidation state of cobalt ion. Hence electronic configuration of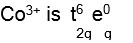
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts