Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
As we know displacement y=sinwt-coswt
=
=
=
To comparing with standard equation
Y= asin (wt+ )
So T=2 /w
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: The octahedral and tetrahedral splitting up of d orbitals is different.
Δt= ( )Δ0
Δt < Δ0
Δt= CFSE in tetrahedral field
Δ0= CFSE in octahedral field
Comparing CFSE with energy=
New Question
10 months agoContributor-Level 10
The total tuition fee for Ranchi University BA courses ranges from INR 966 to INR 5.2 lakh. This fee is given in a range, as the fee varies from institute to institute.
Note: This fee has been sourced from official sources. However, it is subject to change. Hence, it is indicative.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: When light wavelengths from a specific part of the spectrum are absorbed by a substance, the result is a complimentary colour. When a complex absorbs a wavelength of light, it reflects a complementary colour. If a violent colour is absorbed, for example, yellow is conveyed. The CFSE value, often known as the colour definer for any complex, comes next. To determine the wavelength value in order to determine which colour absorbs the most energy.
Δe=
As λ has shorter wavelength.
Low spin complexes absorb shorter wavelengths, while high spin complexes
New Question
10 months agoContributor-Level 10
The Symbiosis Institute of Computer Studies and Research has been ranked by recognised ranking bodies for the MBA category. The Business Today have ranked the SICSR Pune based on various parameters. In 2024, the institute claim 13 ranks to #103 in the 'MBA' category by the Business Today. Check out the following table for more information:
| Publisher | 2022 | 2023 | 2024 |
|---|---|---|---|
| Business Today | 121 | 116 | 103 |
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
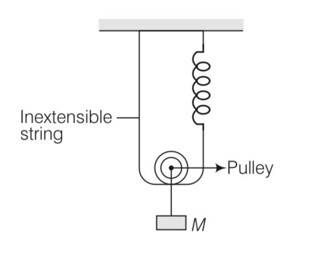
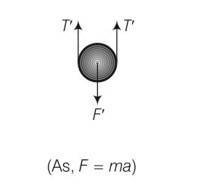
For calculation purpose, in this situation we will neglect gravity because it is constant throughout will not affect the net restoring force.
Let in the equilibrium position, the spring has extended by an amount xo
Let displacement by spring is and string be x .
But string is extensible so only spring will contribute in extension x+x=2x
So net extension is 2x+xo
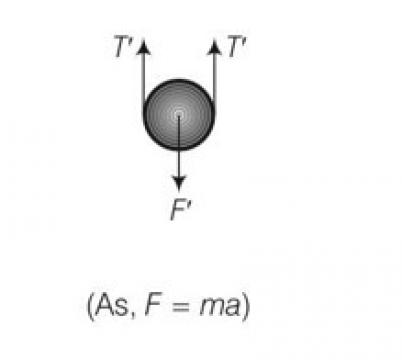
So force is F= 2T
T=kxo
F=2kxo
But when mass is lowered down further by x
F’=2T’ but spring length is 2x+xo
F’=2k (2x+xo)
Restoring force on the system
Frestoring=- (F’-F)
So using above equati
New Question
10 months agoContributor-Level 7
The PTET counselling starts after the results and merit list are declared. The PTET counselling is conducted across multiple rounds to accommodate candidates with different ranks for the BEd course from participating colleges in Rajasthan. Candidates should regularly check the official website for updates regarding the exact schedule and important dates to avoid missing any deadlines. We will also be sharing the direct link for candidates to view the counselling schedule.
New Question
10 months agoContributor-Level 10
The IIITDM Jabalpur BDes cutoff for 2025, in Round 3, has been released. The closing rank for the general AI category is 202. Moreover, after comparing the round 3 closing ranks of the same course, they were 170 and 165 in 2024 and 2023, respectively. Therefore, in the year 2025, the rank increases, which reflects that the competition slightly decreases.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: (i) 'A' is [Co (NH3)5SO4]Cl
'B' is [Co (NH3)5Cl]SO4
(ii) Type of isomerism is ionisation isomerism
(iii) IUPAC name of isomer 'A' is pentaaminesulphatocobalt (III)chloride
IUPAC name of isomer 'B' is pentaaminechlorocobalt (III)sulphate
New Question
10 months agoContributor-Level 10
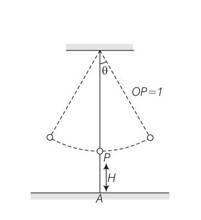
This is a long answer type question as classified in NCERT Exemplar
Let us assume t=0 when o then = 0coswt
Given a seconds pendulum w=2
=
At time t, let
Cos2 , t1=1/6
=-( )sin2
t=t1=1/6
d =- sin
the linear velocity is u=-
the vertical component is Uy=
the horizontal component Ux=-
at the time it snaps the vertical height is
H’=H+l(1-cos )
Let the time require for fall be t , then
H’= H+l (1 )
Let the time required for fall be at t then
H’=uyt+1/2 gt2
1/2gt2+
t=
given that is small , hence neglecting terms of order and higher
t=
H’
t=
the distance travelled in the x direction is
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans:
[Mn (CN)6]3−
Electronic configuration is Mn3+= [Ar]3 d4 hence box electronic structure
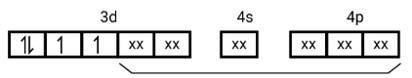
(i) Type of hybridisation d2sp3
(ii) Inner orbital complex
(iii) paramagnetic, due to presence of three unpaired electrons.
(iv) Spin only magnetic moment is calculated using the formula : n=2 in this case, we get spin only magnetic moment in BMas = = = 2.87BM
[Co (NH3)6]3+
Electronic configuration of Co3+= [Ar]3 d6
(i) Hyb As shown in the above box electronic structure the type of hybridisation is . d2sp3
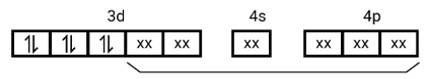
(ii) Inner orbital complex
(iii) Diama
New Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
The gravitational force on the particle at a distance r from the centre of the earth arises entirely from that portion of matter of the earth in shells internal to the positiin of the particles . the external shells exert no force on the particle.
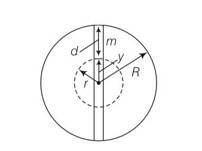
Let g’ be the acceleration at P
So g’ =g (1-d/R)=g (R-d/R)
R-d=y
g’=gy/R’
F=-mg’= -mgy/R
F
Ma=-Mgy/R, a = -gy/R
Comparing a=-w2y
w2=g/R
T=2
New Question
10 months agoContributor-Level 10
Indian Institute of Management Jammu offers undergraduate, postgradute, and Diploma courses. The institute accepts admission based on merit and entrance exams. IIM Jammu accepts national entrance exams such as JEE Main, CAT, MAT, GATE, and others. Aspiring candidates must qualify the eligibility criteria set by the institute for the desired course.
New Question
10 months agoContributor-Level 10
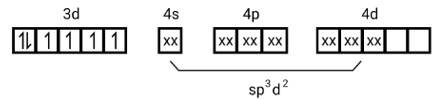
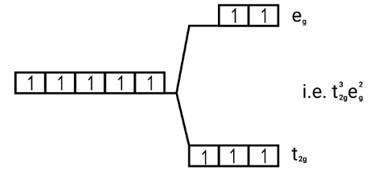
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: (i) Electronic cnfiguration: Co3+ =[Ar]3d6
Energy level diagram:
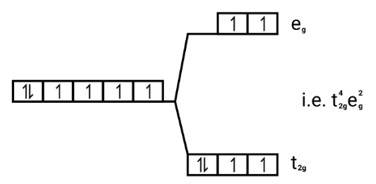
Magnetic moment:
Number of unpaired electrons (n)=4
Magnetic moment = μ= =
= = 4.9 BM
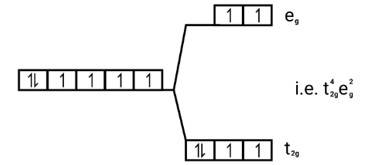
[Co(H2O)6]2+
Electronic cnfiguration: Co2+=[Ar]3 d7
Energy level diagram:
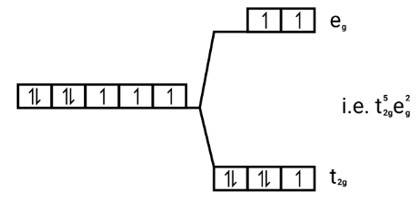
Magnetic moment: Since ,number of unpaired electrons (n)=3, therefore magnetic moment = = = 3.87BM
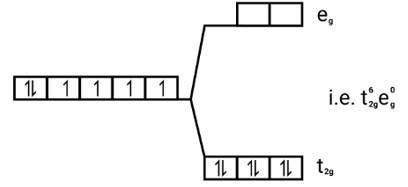
[Co(CN)6]3−
Electronic configuration: [Ar]Co3+=3 d6
Energy level diagram:
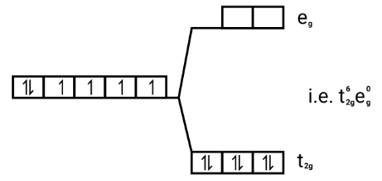
Ans: [FeF6]3−
Electronic configuration: Fe3+=[Ar]3 d5
Ener
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
Let us consider an infinitesimal liquid column of length dx at a height x from horizontal line.
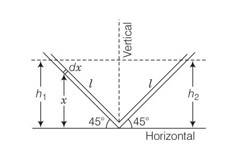
If density of the liquid
PE= dmgx=A
So total PE of the column
= =
But h1=lsin45
PE=A gl2sin245/2
Similarly PE of right column = A gl2sin245/2
Total PE = A gl2sin245/2+ A gl2sin245/2
= A gl2/2
If due to pressure difference is created y element of left side moves on the right side then liquid present in the left arm =l-y
But liquid present in the right arm =l+y
Total PE = PEfinal-PEinitial
Change in PE = ]
= =A
Change in KE = ½ mv2
m=A
change in KE= 1/2A =A
s
New Question
10 months agoContributor-Level 7
To register for the counselling process candidates will have to follow the steps mentioned below:
Visit the official website of IGNOU and check the dates.
Visit the regional centre in offline mode.
Pay the counselling registration fee.
Fill your preferences for the centre.
Submit and lock your choices before the deadline.
Ensure that all details are accurate and the choices are finalized before submission.
New Question
10 months agoContributor-Level 6
It is not confirmed that you will get admission into the MA Clinical Psychology programme at Gujarat University's Navrangpura campus with a 63% score. The eligibility criteria for most postgraduate courses, including psychology, usually require a minimum of 55% to 60%, and some programs, like Amity's, specify 65%. Since the third round is happening, it's possible that the seats are limited and the competition is high.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Sol:
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts