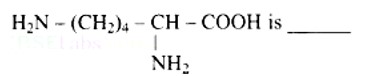Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Yes, Takshashila University offers an MSc programme for students to pursue. Offered at the PG level, this course is spread across various specialisations, including Computer Science, Chemistry, Physics, and more. The duration of all the MSc courses is two years. Moreover, the university admits students to these courses based on scores obtained in the qualifying examination.
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Correct options are a and c.
The cyclic structure of ribose and fructose is five-membered (furanose structures). Like compound furans, they have a five-membered ring.
New Question
10 months agoLysine,
(a) α - Amino acid
(b) Basic amino acid
(c) Amino acid synthesized in body
(d) β- Amino acid
Contributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Correct options are a, b and c.
Lysine is an amino acid with the structural formula of (a).
(b) Because the number of groups (two) is more than the number of -COOH groups (one), it is a basic amino acid (one).
Because it is created in human bodies, it is a non-essential amino acid.
New Question
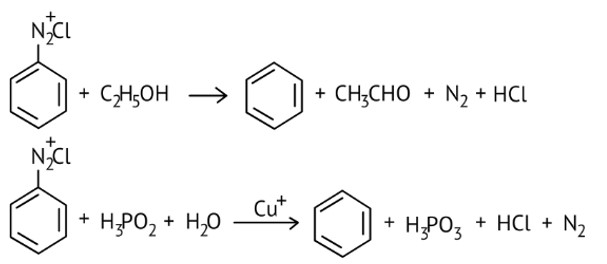
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (B and C)
Certain mild reducing agents like hypophosphorous acid (phosphinic acid) or ethanol reduce diazonium salts to arenes and themselves get oxidised to phosphorous acid and ethanol, respectively.
New Question
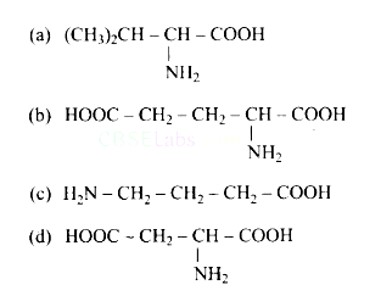
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Correct options are b and d.
Acidic amino acids are divided into two groups, one -COOH group against the other - NH2 group. As a result, the acidic amino acids (b) and (d) are the only ones among the supplied structures.
New Question
10 months agoContributor-Level 10
Group studies are helpful for the students to understand the difficult topics in easy way as the friends can help understand topics easily. The students can also play quiz or explain lessons to one another to check the preparation so far and where they need to focus.
New Question
10 months agoContributor-Level 10
Staying motivated can be hard during long study hours, but setting small goals can help. Reward yourself when you complete a chapter or achieve a target. Surround yourself with positive people who encourage you. Watching motivational videos and reading success stories of toppers can also inspire you. Remember, your efforts today will shape your future success.
New Question
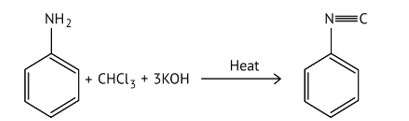
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (A and B)
When aliphatic and aromatic primary amines are heated with chloroform and ethanolic potassium hydroxide, they form isocyanides or carbylamines, which have a foul odor. This reaction does not occur in secondary or tertiary amines.
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Correct options are b and d.
Amylopectin and Glycogen are the correct choices since they are carbohydrate storage forms found in animal tissues, whereas amylose is made up of 80-85% starch. It's also a branched glucose polymer that branches via C1 - C6 glycosidic linkage.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (A, B & C)
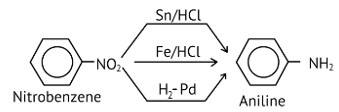
Nitro compounds are reduced to amines by passing hydrogen gas through an acidic medium containing finely divided nickel, palladium, or platinum, as well as by reduction with metals in an acidic medium.
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Correct options are a and c.
Globular protein is a protein structure that forms when a chain of polypeptides coils around to produce a spherical shape. Insulin and albumin are two examples of water-soluble globular proteins. As a result, (a) and (c) are the correct answers.
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Correct options are b and d.
Sucrose is a common disaccharide that breaks down into an equimolar mixture of D- (+)- glucose and D- (-)- fructose when hydrolyzed. These two monosaccharides are held together by a glycosidic bond between C1 of α - glucose and C2 of β fructose. Because the reducing groups of glucose and fructose are involved in the formation of glycosidic linkages, sucrose is a non-reducing sugar.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (C and D)
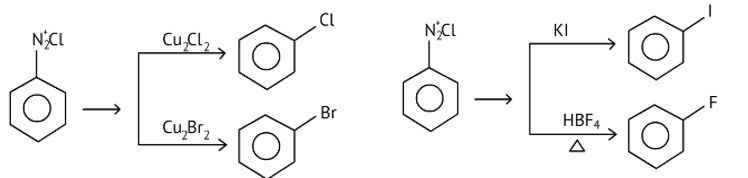
Chlorobenzene and bromobenzene are prepared by the sandmeyer's reaction.
While fluorobenzene and iodobenzene are prepared by simple heating of diazonium salt with aqueous KI Solution.
New Question
10 months agoContributor-Level 10
MBA at the School of Business Management (NIU) covers a wide range of core and advanced subjects, including Principles of Management, Financial Accounting, HRM, Managerial Economics, Business Analytics, Data Science, Corporate Governance, and more. Depending on the specialisation, students also explore industry-focused topics such as Pharmaceutical Marketing, Agri Supply Chain, or International Trade to gain deeper domain-specific knowledge.
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Correct option is C.
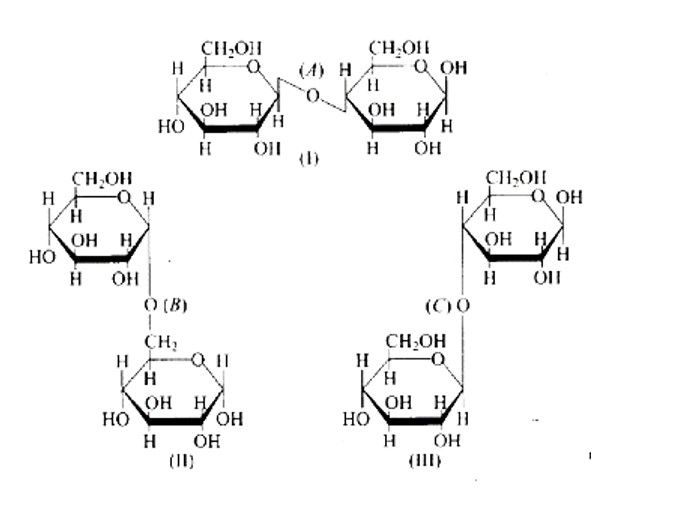
The links between C1 and C4 of glucose are shown as 'A' and ' C, ' respectively, whilst the linkage between C1 and C6 of the glucose units is shown as ' B. ' Furthermore, option (c) is clearly correct based on the structures.
Because ' B ' is the bond or link between the glucose units of C1 and C6 and, option (b) is wrong.
Option (a) is wrong because the relationship between C1 and C4 and is represented by 'C.'
Because 'A' is the hyperlink between C1 and C4 the glucose units of and, option (d) is wrong.
New Question
10 months agoContributor-Level 7
Last year the OBC cutoff for B.Sc courses was around 14,579. So for OBC girls, a rank within the top 15,000 in CUET is usually required for admission in B.Sc Mathematics at GGU
New Question
10 months agoNew Question
10 months agoBeginner-Level 5
The Atal Bihari Vajpayee Medical University (ABVMU), Lucknow, releases the result of UP CPET 2025 in online mode only. Students will be able to check their UP CPET results by visiting the official website only; there is no other alternative source for the result.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
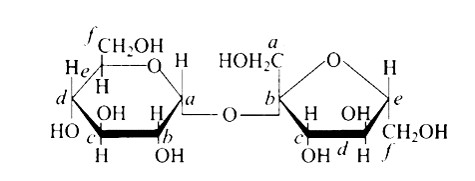
The Correct option is c.
In the cyclic structure of glucose or fructose, anomeric carbon is carbon that is close to an oxygen atom. As shown in the structure above, atoms 'a' and 'b' are close to the oxygen atom, and the hydroxyl groups of both carbon atoms have different orientations.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts