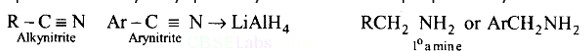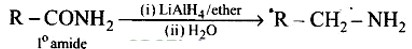Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (D)
Aliphatic and arylalkyl primary amines can be prepared by the reduction of the corresponding nitriles with LiAlH4
Heating alkyl halide with primary, secondary and tertiary amine can be prepared by reduction of LiAlH4 followed by treatment with water.
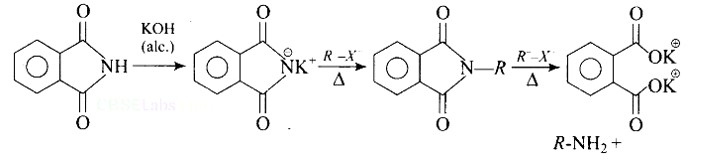
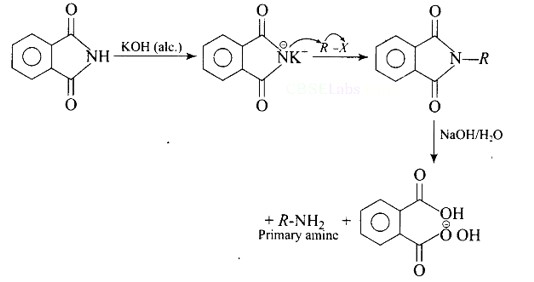
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis produces primary amine. This process is known as Gabriel phthalimide reaction. The number of carbon atoms in the chain of amines of product is same as reactant.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (B)
Among primary, secondary, and tertiary amines, tertiary amines are the most volatile compounds as they don't have any strong intermolecular H-bonding between N-H like the primary and secondary amines have.
So due to weak dipole-dipole interactions, (B) will be the most volatile.
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is a.
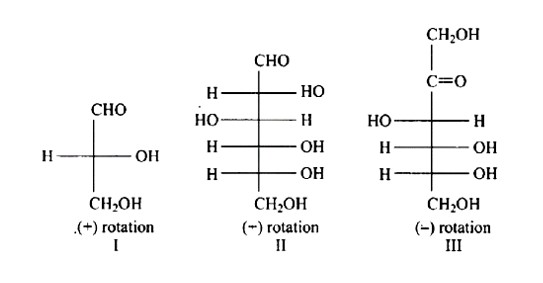
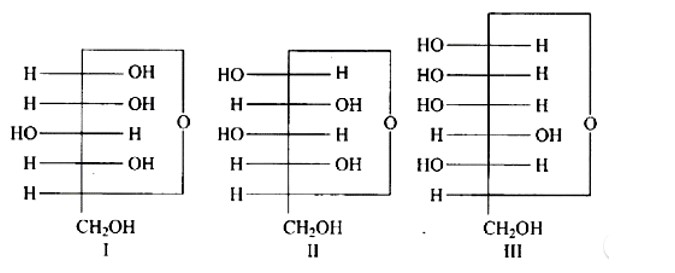
In the same way as (+) glyceraldehyde has a group on the lowest asymmetric carbon on the right side, the I, II, and III structures have a (-OH) group on the lowest asymmetric carbon on the right side giving them a D-configuration.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (A)
The basic strength of an atom is determined by its electron-donating capacity; in this case, the amide is the most basic due to the presence of a negative charge and two lone pairs of electrons on the nitrogen atom.
New Question
10 months agoContributor-Level 10
NEU Boston is considered among the best universities in the US for higher studies. Several prominent university ranking bodies have ranked it on a national and global level. Given below is how the university ranks in 2026.
Ranking Authorities | Rankings |
|---|---|
Shiksha Popularity Rankings | #295 in the world |
QS World University Rankings 2026 | #384 |
US News & World Report | #46 in the US |
THE World University Rankings 2026 | #201 |
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is c.
The absence of a free (-CHO) aldehyde group is indicated by the fact that glucose pentaacetate does not react with hydroxylamine. The open structure of glucose cannot account for this, although the open chain structure of glucose can account for all other features.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (D)
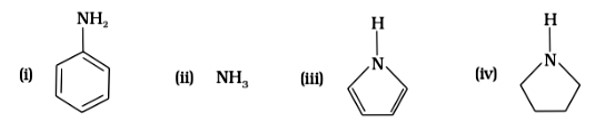
Pyrrolidine is the strongest of two bases because the lone pair of nitrogen does not involve sin resonance, and the presence of two alkyl basic compounds increases the basic strength among the given four compounds.
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Ans: The Correct option is a.
Anomers are cyclic monosaccharide structures that differ structurally at carbon-1. In this scenario, I and II are anomers since they differ only at carbon -1.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (D)
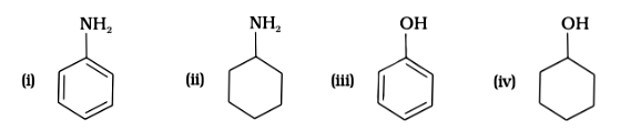
Since phenol is the strongest acid among the four options listed above, it is the weakest Brönsted base. The stronger the acid, the weaker the conjugate base.
Amines have a strong tendency to accept electrons thus they are a strong bronsted base while phenol is the strongest acid among all, therefore as per the relation of conjugative strong acids and weak bases, phenol is the weakest base.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is d.
Adenine, guanine, thymine, and cytosine are the four bases found in DNA. As a result, uracil is not found in DNA as we cannot identify it among the other nucleotides that are present in the DNA. Uracil is, however, seen in RNA, another nucleic acid found in most of the organisms.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (D)
Weak electrophile such as Diazonium cation readily reacts with electron-rich compounds which are having electron-rich compounds such as hydroxyl group, amino group. They don't react with electron-withdrawing groups like nitro groups. Therefore, nitrobenzene will not undergo an azo coupling reaction with benzene diazonium chloride.
New Question
10 months agoContributor-Level 10
The institution has not released the course-wise placement packages as of yet. Meanwhile the NIRF report has been released. Check out the table below to know more:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) | Placement Statistics (2024) |
|---|---|---|---|
Total Students | 5,983 (UG 3-year) 166 (PG 1-year) 1,731 (PG 2-year) | 6,000 (UG 3-year) 39 (PG 1-year) 1850 (PG 2-year) | 6,156 (UG 3-year) 364 (PG 1-year) 2,124 (PG 2-year) |
No. of Students Placed | 114 (UG 3-year) 3 (PG 1-year) 72 (PG 2-year) | 120 (UG 3-year) 5 (PG 1-year) 94 (PG 2-year) | 59 (UG 3-year) 23 (PG 1-year) 112 (PG 2-year) |
Median Salary | INR 3.50 LPA (UG 3-year) INR 3.60 LPA (PG 1-year) INR 4 LPA (PG 2-year) | INR 3.50 LPA (UG 3-year) INR 3 LPA (PG 1-year) INR 3.6 LPA (PG 2-year) | INR 3.70 LPA (UG 3-year) INR 3.20 LPA (PG 1-year) INR 4 LPA (PG 2-year) |
NOTE: The above data is obtained from the NIRF report 2025
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is d.
Water-soluble vitamins are those in the B group. B group vitamins cannot be stored in our systems since they are quickly excreted; but, because they are B12 insoluble in water, our bodies may store the vitamin. Because our systems are unable to store these vitamins, options (a), (b), and (c) are excluded.
New Question
10 months agoContributor-Level 10
For MBA admission at the School of Business Management (NIU) students must upload and later submit documents such as a recent passport-size photograph, graduation marksheet, signature, valid ID proof, and academic transcripts (Class 10 and 12 mark sheets). These documents are essential for both the online application and final admission formalities, like counselling and verification.
New Question
10 months agoContributor-Level 10
The table below contains the data from the NIRF report on Patna University:
Particulars | 2022 | 2023 | 2024 |
|---|---|---|---|
Total Students | 5,983 (UG 3-year) 166 (PG 1-year) 1,731 (PG 2-year) | 6,000 (UG 3-year) 39 (PG 1-year) 1850 (PG 2-year) | 6,156 (UG 3-year) 364 (PG 1-year) 2,124 (PG 2-year) |
No. of Students Placed | 114 (UG 3-year) 3 (PG 1-year) 72 (PG 2-year) | 120 (UG 3-year) 5 (PG 1-year) 94 (PG 2-year) | 59 (UG 3-year) 23 (PG 1-year) 112 (PG 2-year) |
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is c.
The four bases contained in DNA are adenine, guanine, thymine, and cytosine. Adenine, uracil, guanine, and cytosine are the four bases of RNA. As a result, RNA does not include thymine as in place of thymine, it has uracil which is a lesser stable nucleotide base and is somewhat responsible for RNA's instability but also lends it some flexibility to change.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (B)
The reaction of transformation of primary alkyl halides to primary amines using potassium phthalimide is the Gabriel phthalimide reaction.
New Question
10 months agoContributor-Level 10
Patna University provides good placements to its graduating students. The students are given excellent placement support every year placing them in top companies. Check out the table below to know the key highlights of the Patna University placements 2024:
Course | Total Students (2024) | Students Placed (2024) | Median Salary (2024) |
|---|---|---|---|
UG 3-year | 6,156 | 59 | INR 3.70 LPA |
PG 1-year | 364 | 23 | INR 3.20 LPA |
PG 2-year | 2,124 | 112 | INR 4 LPA |
NOTE: The above data is obtained from the NIRF report 2025
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is a.
Proteins contain one or more polypeptide chains. When each polypeptide of a protein's amino acids is joined in a precise order, a proteins' main structure is referred to as its primary structure. This one is the linear structure with no bonds other than the ones between adjacent amino acids. The sequence of amino acids in the primary structure is pre-determined by the genetic code of an individual and thus is a specific sequence.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts