Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
The subjects included in the first two semesters of the BDes in Communication Design are meant to create a foundational base for the students. Further, from the third semester onwards, the institute offers specialisations in the BDes course. The names of the specialisations are- Graphics, Animation & VFX, UX/ UI, and Game design. Further, the subjects are offered as per the specialisation selected by the student from these mentioned specialisations. Check out the names of the subjects that are common for all specialisations:
- Visual Representation & Imaging 1 & 2
- Design Fundamentals 1 & 2
- Materials for Design 1 & 2
- Domain Technology (
New Question
10 months agoContributor-Level 10
The placement rate for National Institute of Design for 2025 has not been released yet. Refer to the table below to know more bout the placement highlights recorded in 2024:
Particulars | Placement Statistics (2024) |
|---|---|
No. of Students Registered | 686 |
No. of Offers | 476 |
the highest Package | INR 36 LPA |
the lowest Package | INR 3 LPA |
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is c.
The pyranose form of glucose, which has a five-membered ring structure, is found. As a result, option (c) is wrong.
Glucose is a six-carbon monosaccharide with an aldehyde group. As a result, it's referred to as an aldohexose. As a result, the statement in option (a) is correct.
When glucose is cooked with HI, n-hexane is produced. All six carbon atoms in glucose are linked together in a straight chain during this procedure. As a result, the statement in option (b) is correct.
The aldehyde group is not free in the cyclic structure. As a result,
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(b) Equanil is a tranquilizer used in controlling depression and hypertension.
New Question
10 months agoContributor-Level 10
Some of the top recruiting companies at National Institute of Design are given in the table below:
National Institute of Design Placements: Top Recruiters | |
|---|---|
Microsoft | Samsung |
Infosys | SAP |
Wipro | Cognizant |
Infoedge | Hindustan Unilever |
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
The Correct option is b.
Nucleic acids are nucleotide polymers linked together by phosphodiester linkage. DNA and RNA are two examples. Nucleotides are formed by linking of nucleosides with three phosphate groups consuming a significant amount of energy making it an endothermic reaction.
New Question
10 months agoContributor-Level 10
The National Institute of Design average package has not been released yet, Once updated it will be mentioned here. Meanwhile, students can refer to the highest and average package provided in recent years:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) | Placement Statistics (2024) |
|---|---|---|---|
the highest Package | INR 46 LPA | INR 30 LPA | INR 36 LPA |
the lowest Package | INR 4 LPA | INR 4 LPA | INR 3 LPA |
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
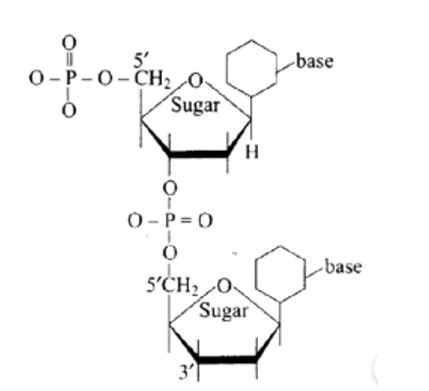
(a) 5′ and 3′ linkages are present between pentose sugars of nucleotides.
Phosphodiester links the two nucleotides together to form a dinucleotide.
Between the pentose sugars of nucleotides, there are 5′ and 3′ connections. In the process of adding phosphate to a dinucleotide, a significant quantity of energy is consumed.
New Question
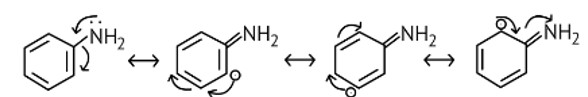
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (A)
The NH2 group is directly attached to the benzene ring in aniline and other aryl amines. As a result, the unshared electron pair on the nitrogen atom is conjugated with the benzene ring, making it less available for protonation.
New Question
10 months agoContributor-Level 10
Refer to the table below to know more about the highest package recorded during the past three years:
Particulars | Placement Data 2022 | Placement Data 2023 | Placement Data 2024 |
|---|---|---|---|
Highest Package | INR 46 LPA | INR 30 LPA | INR 36 LPA |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(d) All soaps are made by boiling fats or oils with suitable hydroxide. Variations are made by adding different raw materials. Sodium laurylsulphate and sodium dodecylbenzenesulphonate are anionic detergents.
New Question
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
Ans: The Correct option is b.
Ascorbic acid is the chemical name for vitamin C. Aspartic acid is an amino acid, adipic acid is a dicarboxylic acid with an eight-carbon chain, and saccharic acid is a dicarboxylic acid made by oxidising glucose with HNO3 Vitamin C is a water-soluble vitamin that serves a variety of activities in the human body.
New Question
10 months agoContributor-Level 10
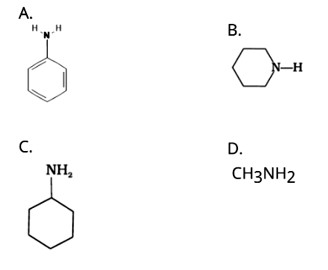
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (C)
Because of the electron-releasing nature of the alkyl group, it (R) pushes electrons towards nitrogen, making the unshared electron pair more available for sharing with the acid's proton. Furthermore, the +I effect of the alkyl group stabilizes the substituted ammonium ion formed from the amine by dispersing the positive charge.
As a result, alkylamines are more powerful bases than ammonia.
As a result, the basic nature of aliphatic amines should increase as the number of alkyl groups increases.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct answer: (D)
New Question
10 months agoBeginner-Level 5
The tiebreakers are resolved in the UP CPET exam based on the below-given methodology.
- Higher marks in Biology (Botany & Zoology)/Mathematics/Computer Science/Electronics/Statistics/Psychology in the Test
- Higher marks in Chemistry in the Test
- Higher marks in Physics in the Test
- Higher marks in English and General Knowledge in the Test
- Older age
- Higher class 10th marks percentage
- Higher class 12th marks percentage
New Question
10 months agoContributor-Level 10
APU Bangalore BSc admissions are based on JEE Main, CUET, SAT India and Azim Premji University National Entrance Test (university-in-house-entrance exam), followed by PI. The university also offers admission to BSc courses with the scores of candidates and availability of seats for the chosen specialisation. To be eligible for a BSc at Azim Premji University, candidates must complete their Class 12 in the PCM/ PCB stream with at least an aggregate of 50%. Additionally, candidates must ensure that the upper age limit to apply for our undergraduate programmes is 21 years as of Jul, 31 2025.&nbs
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(b) The compound that causes general antidepressant action on the central nervous system belongs to the class of tranquilizers.
New Question
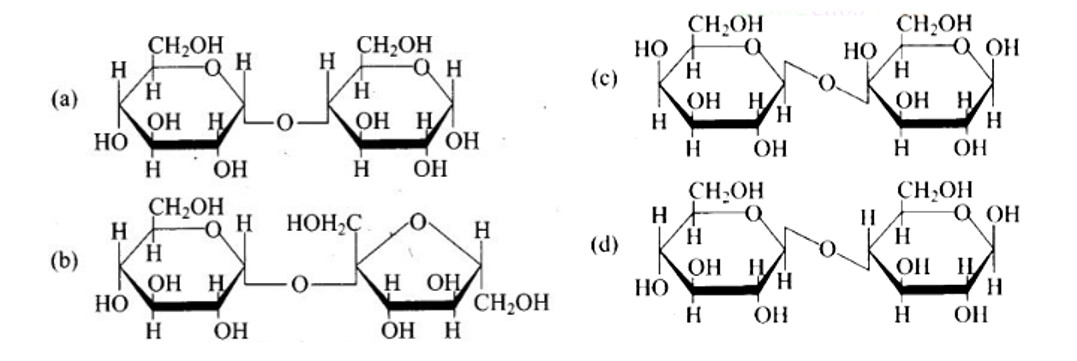
10 months agoContributor-Level 10
This is a Multiple Answer Type Questions as classified in NCERT Exemplar
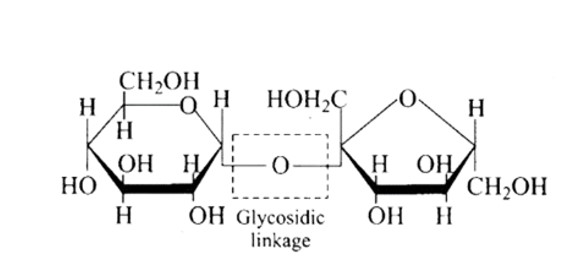
Ans: The Correct option is b.
The structure described in option (b) reflects sucrose, which has a C1−C2 glycosidic bond between α−D− glucose and β−D− fructose.
This is a non-reducing sugar since the reducing groups of glucose and fructose are involved in the creation of glycosidic linkages.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts