Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Assertion Type Questions as classified in NCERT Exemplar
64. Option (iii) i.e., Both A and R are not correct is the answer since sulphur is estimated by Carius method in the form of white precipitate of BaSO4 on heating with fuming and BaCl2. If light yellow solid is obtained means impurities are present. It is filtered, washed and then dried to get pure BaSO4 .
New Question
10 months agoContributor-Level 10
(i) Let A =
Then, A’ =
Let P = (A + A’) =
=
Then, P’ = = P.
∴ P = (A + A’) is symmetric matrix
Let Q = (A + A’) =
=
Then Q.’ = = (-1) = (-1) Q.
Q.’ = Q,
∴ Q = (A - A’) is a symmetric matrix
Now, P + Q = (A + A’) + (A - A’)
P + Q = = A.
This A is represented as a sun of symmetric and skew symmetric matrix
Let A =
Then A’ =
Now, A + A’ =
= =
Let P = (A + A’) =
Then, P’ =&n
New Question
10 months agoContributor-Level 8
For admission to BCA at Babasaheb Bhimrao Ambedkar University (via the Amethi satellite centre), CUET 2022–23 cut offs in the General/EWS category ranged roughly from 162 to 221 marks. Actual eligibility changes yearly based on competition and available seats, so final cut offs for BBAU’s main campus will reflect that variation.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The lewis structure of O2– ion is,
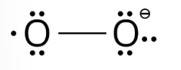
Oxygen atom having no charge has 6 electrons, so its oxidation number is zero. Oxygen atoms containing −1 charge have 7 electrons, so its oxidation number is −1. The average oxidation state of oxygen in this ion is, =1/2
New Question
10 months agoContributor-Level 10
Candidates seeking admission to Maya Somaiya School of Music and Performing Arts MPA courses must fulfil the eligibility criteria set by the institute. Check below to know the course-specific eligibility requirements:
Vocal, Swar Vadya, Taal Vadya:
- Completed a three-year BPA (Music) or equivalent examination with 45% or more, or
- Passed an undergraduate degree and must have passed Music for not less than five years in a recognised institution, or
- An undergraduate degree in any subject recognised by the UGC and having learnt Music for not less than five years from a well-known teacher/guru
Bharata Natyam & Kathak:
- Completed
New Question
10 months agoContributor-Level 10
Some MMS courses in Mumbai are relatively more affordable than MBA programmes in the city. At Shiksha, you can find 10+ MMS colleges in Mumbai that cost INR 1 to 3 Lakh for tuition only. Around 2 institutes offer the same under INR 1 Lakh. See the most affordable options with good quality programmes.
| Affordable MMS Courses in Mumbai | Tuition Fee |
|---|---|
| SIMSREE MMS course | INR 69,000 |
| XIBMS MMS course | INR 45,000 |
| VIVA Institute of Management and Research MMS Course | INR 1.62 Lakh |
| Shivajirao S. Jondhle Institute of Management Science and Research MMS Course | INR 1.24 Lakh |
| S.A.V Acharya Institute of Management Studies MMS Course | INR 1 Lakh |
Note: All info. is from official sites and can change.
New Question
10 months agoContributor-Level 10
Somaiya Vidyavihar University provides financial aid to candidates enrolled in the MLISc course at Department of Library and Information Science. The financial aid is provided in the form of tuition fee concessions. Candidates are required to fulfil the specified eligibility criteria to avail of the financial aid. Listed below are the criteria for various the available financial aid:
- EWS/BPL: Applicants with family income up to INR 8 LPA are eligible for financial aid worth up to 100% on the tuition fees
- PWD: Applicants with benchmark disabilities (40% or more) are eligible for a concession of 50% on the tuition fees
- Defence Personnel: Wa
New Question
10 months agoContributor-Level 10
This is a Assertion Type Questions as classified in NCERT Exemplar
63. Option (iv) i.e., A is not correct, but R is correct is the answer since hybridization of C can be found out by counting σ bonds and π bonds present on C atom. This can be shown as-

If C has 3σ bonds then it is sp2 hybridized. If C has 2σ bonds then it is sp hybridized.
New Question
10 months agoContributor-Level 10
Given, A =
Then, A’ =
So, A + A’ =
(A + A’) =
And A - A’ =
=
(A - A’) = =
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
In the Solvay process, carbon dioxide is transferred through a concentrated solution of ammoniacontaining sodium chloride, which forms ammonium carbonate followed by ammonium hydrogen carbonate. The chemicals in ammonium hydrogen carbonate are different and are heated to form sodium carbonate. NH3 is found in a solution containing NH4Cl that is heated and treated with Ca (OH)2? The reaction of (NH4)2CO3 with NaCl provides two products, Na2CO3 and NH4Cl both soluble in water which do not shift to the right balance.
New Question
10 months agoContributor-Level 10
Given, A =
Then, A’ =
Let P = A + A’ =
So, P’ = = P
i e, ( A + A’ )’ = A + A’.
Hence, A + A’ is symmetric matrix.
Let Q = A A’ =
So,Q1 = = (1) = (1) Q.
Q1 = Q.
i e, (A A’)’ = -(A - A’).
Have, A - A’ is a show symmetric matrix
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Ionic compounds are formed from the alkali metals due to their large ionic size and low ionization enthalpy. Thus, they are soluble in water. But, due to the small ionic size, high ionization enthalpy, and high electronegativity of lithium, it forms compounds of covalent nature and thus, are soluble in organic solvents.
New Question
10 months agoContributor-Level 10
This is a Assertion Type Questions as classified in NCERT Exemplar
62. Option (i) i.e.,
Both A and R are correct, and R is the correct explanation of A is the answer as when two or more compounds differ in the position of substituent atom or functional group on the carbon skeleton, they are known as position isomers and this phenomenon is termed as position isomerism. Pent-2-ene and pent-l-ene are position isomers because they differ in the position of the double bond.
New Question
10 months agoContributor-Level 10
(i) Given A =
Then, A’ =
∴A’ = A.
Here, A is symmetric matrix
(ii) Given, A =
Then, A’ =
A’ = (1) A.
A’ = A.
Hers A is a show symmetric matrix.
New Question
10 months agoContributor-Level 10
In recent years, Mohamed Sathak College of Arts and Science has welcomed many well-known companies for BSc placements. During the 2024 placement season, firms like HCL, First Source Solution, Foxconn, and EOS Globe Solutions recruited students. The following year, 2025, companies such as Hitachi, India Filings, and Prudent took part, providing a variety of job opportunities across multiple sectors.
New Question
10 months agoContributor-Level 10
This is a Assertion Type Questions as classified in NCERT Exemplar
61. Option (iv)
i.e., A is not correct, but R is correct is the answer. This is because canonical structures always have more energy than the resonance hybrid. Resonance hybrids are always found to be more stable than any of the canonical structures since the delocalization of electrons lowers the orbitals energy and gives stability.

New Question
10 months agoContributor-Level 9
SRM School of Management offers eleven specialsiations in the MBA course, from which the school does not offer the highest number of seats in any specialisation. However, it offers the highest number of seats in the general MBA course. Check out the below table to get an exact number of seats offered in the general MBA course:
| Courses | Seats |
|---|---|
| Master of Business Administration (MBA) | 450 |
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
BeSO4 and MgSO4 readily soluble in water while CaSO4, SrSO4 and BaSO4 are insoluble The greater hydration enthalpy of Be2+ and Mg2+ ions overcome the lattice enthalpy factor and therefore, their sulfates are soluble.
New Question
10 months agoContributor-Level 10
(i) Given, A =
Then, A’ =
∴A’ A =
=
=
=
= A ’ A = 1.
Given,
(ii) 1 A =
Then, A’ =
∴A’ A =
=
=
=
A’ A. = I
New Question
10 months agoContributor-Level 10
A Master of Management Studies (MMS) degree can be quite beneficial for students who wish to build their careers in the fields of business administration and management. Students will develop theoretical knowledge and learn through practical internship experiences if they choose to pursue an MMS course from Mumbai colleges.
Students can evaluate the ROI of MMS colleges in Mumbai by comparing the fees and salary given in the table below:
College Name | Average Fees | Average Package |
|---|---|---|
INR 6.01 Lakh- 7 Lakh | INR 26.4 LPA | |
Prin. L. N. Welingkar Institute of Management Development MMS | INR 6.7 Lakh | INR 10 LPA |
INR 4.52 Lakh | INR 7.5 LPA | |
INR 2.79 Lakh | INR 5 LPA | |
INR 80,000 | INR 6 LPA |
Disclaimer: This information is sourced from the official website and may vary.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
