Ask & Answer: India's Largest Education Community
All Questions
New Question
6 months agoContributor-Level 10
Correct order of van there Waals constant b for gases is CO2 > N2 > O2 > H2 > He
New Question
6 months agoContributor-Level 6
No. The Commission will release the UGC NET notification 2026 for the June session in the form of PDF tentatively in April at ugcnet.nta.nic.in. The registration will also start for the June sesison along with the notification.
New Question
6 months agoContributor-Level 10
Kp = Kc (RT)Dng
36 * 10–2 = Kc (0.0821 * 300)–1
Kc = 0.36 * 0.0821 * 300 = 8.86 » 9
New Question
6 months agoNew Question
6 months agoContributor-Level 10
Charge passed = It
= 0.015 × 15 × 60 C
Moles of electrons passed
Moles of Zn deposited =
= 0.00007
Mass of Zn deposited = 0.00007 × 65.4 g = 4.58mg
New Question
6 months agoNew Question
6 months agoContributor-Level 10
B2 have bond order equal to 1 and also paramagnetic.
have bond order equal to 1 but are diamagnetic.
have bond order equal to 3.
New Question
6 months agoContributor-Level 10
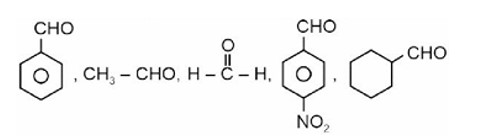
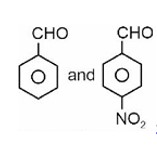
Fehling solution test can be given by aldehyde except aromatic aldehyde
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts