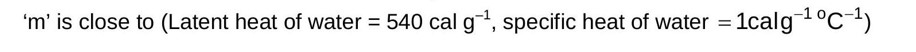The amount of heat needed to raise the temperature of 4 moles of a rigid diatomic gas from 0°C to 50°C when no work is done is___________. (R is the universal gas constant)
The amount of heat needed to raise the temperature of 4 moles of a rigid diatomic gas from 0°C to 50°C when no work is done is___________. (R is the universal gas constant)
Since process is isochoric, so
Similar Questions for you
Heat lost by steam Heat gained by water and calorimeter.
In isothermal process, temperature is constant.
In isochoric process, volume is constant.
In adiabatic process, there is no exchange of heat.
In isobaric process, pressure is constant
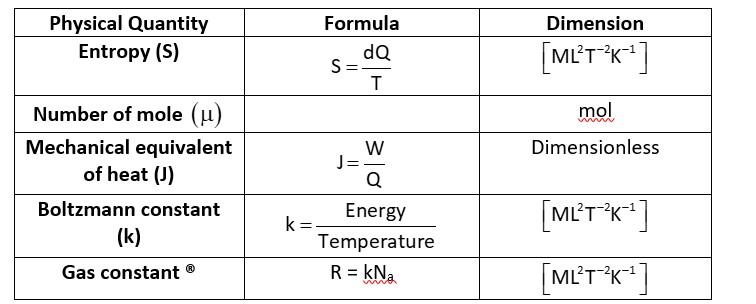
Na -> Avogadro’s Number
, and
-> Dimensionless
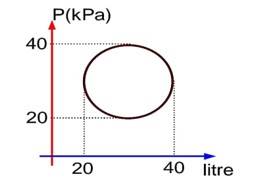
Heat absorbed in cyclic process = Work done = 100? Joule
Since process is isochoric
So
And external work
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else.
On Shiksha, get access to
Learn more about...
Didn't find the answer you were looking for?
Search from Shiksha's 1 lakh+ Topics
Ask Current Students, Alumni & our Experts
Have a question related to your career & education?
See what others like you are asking & answering