Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
Tl belongs to group 13 and shows both the oxidation state +1 and +3 due to inert pair effect. Tl forms basic oxide like group I elements. TlO2 is strongly basic.
New Question
11 months agoContributor-Level 10
2.9 Dielectric constant of the mica sheet, k =6
While the voltage supply remained connected :
V = 100 V
Initial capacitance, C = 17.708 F
New capacitance, = kC = 6 17.708 = 106.25 F
= 106.25 pF
New charge, = = 106.25 C = 10.62 C
If the supply voltage is removed, then there will be constant amount of charge in the plates.
Charge, q = CV = 17.708 C = 1.7708 C
Potential across plates, = = = 16.66 V
New Question
11 months agoContributor-Level 10
1.14. S.I. unit of mass is kilogram (kg).
It is defined as the mass of platinum-iridium block stored at international bureau of weights and measures in France.
New Question
11 months agoContributor-Level 10
(a) Neutral — CO
Acidic — B2O3, SiO2, CO2
Basic — Tl2O3
Amphoteric — Al2O3, PbO2
(b)-CO does not react with acid as well as base at room temperature.
Being acidic B2O3, SiO2 and CO2 react with alkalis to form salts.
B2O3 + 2NaOH à 2NaBO2 + H2O
SiO2 + NaOH à 2Na2SiO3 + H2O
CO2 + 2NaOH à Na2CO3 + H2O
Being Amphoteric, Al2O3, PbO2 react with acids and bases.
Al2O3 + 2NaOH à 2NaAlO2 + H2O
Al2O3 + 3H2SO4à (Al2SO4)3+ 3H2O
PbO2 + 2 NaOH à Na2PbO3 + H2O
2PbO2 + 2 H2SO4à 2PbSO4 + 2H2O + O2
Being basic Tl2O3 dissolves in acids
Tl2O3 + 6HCl à 2TlCl3 + 3H2O
New Question
11 months agoContributor-Level 10
1.13. Pressure= Force/Area
But weight = m x g, where m = mass (in kg) and g = 9.8 m/s2
Therefore, Pressure = 1034 g cm-2 x 9.8 m/s2
= 1034 x 10-3 kg x (100 m)2 x 9.8 m/s2
= 101332 Pa
= 1.01332 x 105 Pa
New Question
11 months agoContributor-Level 10
2.8 Area of each plate, A = 6
Distance between plates, d = 3 mm = 3 m
Supply voltage, V = 100 V
Capacitance C of the parallel plate is given by C =
In case of air, dielectric constant k = 1 and
= permittivity of free space = 8.854
Hence C = F = 17.708 F = 17.71 pF
Charge on each plate of the capacitor is given by q = VC = 100 C
= 1.771 C
Therefore, capacitance of the capacitor is 17.71 pF and charge on each plate is 1.77 C
New Question
11 months agoContributor-Level 10
Candidates wishing to apply for courses at Justice KS Hegde Institute of Management are requried to fill out the application form by uploading the scanned copies of the following documents:
- Marksheets of Class 10 and Class 12
- Graduation marksheet
- Government identity such as the Aadhaar Card
Candidates must apply to the courses through the application form. Candidates are also required to pay the application fee to successfully apply for admission to Justice KS Hegde Institute of Management.
New Question
11 months agoBeginner-Level 5
Some of the notable colleges and universities offering lateral entry admissions include
Anna University, Chennai
Jadavpur University, Kolkata
Thapar Institute Of Engineering & Technology, Punjab
PSG College Of Technology, Coimbatore
Amity University, Uttar Pradesh
Manipal Institute Of Technology, Manipal
Delhi Technological University [DTU], Delhi
College of Engineering [COEP], Pune.
These are top colleges that accept lateral entry
New Question
11 months agoNew Question
11 months agoContributor-Level 10
1.12. Density of methanol = 0.793 kg/L, molar mass of methanol (CH3OH) = 32g/mol = 0.032 kg/mol
V1 =? , V2 = 2.5 L, M2 = 0.25 M
We can apply the formula of
M1V1 = M2V2
Or V1 = M2V2/M1
Substituting M1 = density / molar mass, we get
M1 = 0.793/0.032 = 24.78
V1 = 0.25 x 2.5 / 24.78 = 0.02522 L = 25.22 mL
New Question
11 months agoBeginner-Level 1
Yes definitely, 12th marks is not related to any admission in any colleges and it is just a qualification that any student needed to get admission in BTech. You might get scholarship and all with the good marks in 12th but getting admission just need passing marks in 12th board.
New Question
11 months agoContributor-Level 10
1.11. Molar mass of sugar = (12 x 12) + (22 x 1) + (11 x 16) =342 g/mol
No. of moles in 352 g of sugar = 1 mol
No. of moles in 20 g = 20 x 1/352 = 0.0585 mol
Therefore, molar concentration = moles of solute / volume of solution in L = 0.0585 / 2 = 0.0293 mol/L
New Question
11 months agoGuide-Level 12
Allotropes: Allotropes are the different forms of an element which are having same chemical properties but different physical properties due to their structures.
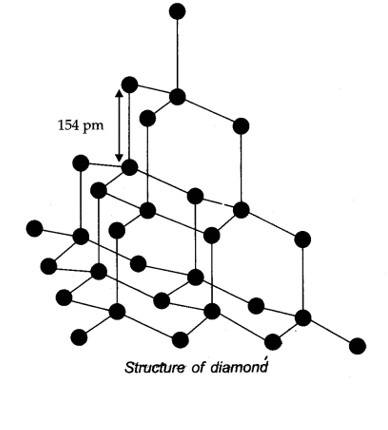
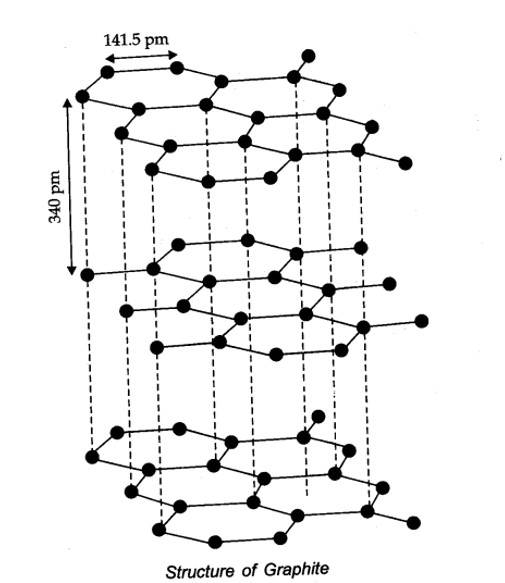
New Question
11 months agoContributor-Level 10
Due to poor shielding effect of d-electrons in Ga, the electrons in gallium experience great force of attraction by nucleus as compared to Al. That is why Ga has lower atomic radius as compared to Al.
New Question
11 months agoContributor-Level 10
Because there is increase in atomic size on moving from carbon to silicon, the screening effect increases. Thus, the force of attraction of nucleus for the valence electron in silicon decreases as compared to carbon. Therefore, the ionization enthalpy decreases from carbon to silicon.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
DMLT (Diploma in Medical Laboratory Technology) and GNM (General Nursing and Midwifery) are diploma-level programs, and both are great options in their own. It depends on your future goals and interests. But, I would suggest going for a UG-degree course like BSc Nursing after these courses to get better job and salary options.
New Question
11 months agoContributor-Level 10
(i) Al reacts with conc. HNO3 to form a very thin film of aluminium oxide on its surface which protects it from further reaction. That is why conc. HNO3 can be transported in aluminium container.
2Al (s) + 6HNO3 (conc.) → Al2O3 (s) + 6NO2 (g) + 3H2O (l)
(ii) NaOH reacts with Al to evolve H2 gas. Thus, the pressure of the gas produced can be used for opening or cleaning clogged drains.
2Al (s) + 2NaOH (aq) + 2H2O (l) → 2NaAlO2 (aq) + 3H2 (g)
(iii) Graphite has layered structure which are held by weak van there Waals forces. Thus, graphite cleaves easily between the layers, therefore it is very soft and slippery. That
New Question
11 months agoContributor-Level 10
1.10. (i) 1 mole of C2H6 contains 2 moles of carbon atoms
Therefore, no of moles of C atoms in 3 moles of C2H6 = 6 moles
(ii) 1 mole of C2H6 contains 6 moles of hydrogen atoms
Therefore, no of moles of H atoms in 3 moles of C2H6 = 18 moles
(iii) 1 mole of C2H6 contains 6.02 x 1023 molecules
Therefore, 3 moles of C2H6 will contain ethane molecules = 3 x 6.02 x 1023 molecules= 18.06 x 1023 molecules
New Question
11 months agoContributor-Level 10
Magadh University grants admission to BA Hons courses based on scores obtained in the last qualifying examination. Thus, in order to take admission, students are not required to appear for any entrance examination. However, it is to be noted that the admission criteria may vary depending on the choice of institute.
Note: The above information has been taken from official sources. In order to get detailed information, students are advised to contact the university directly.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
