Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
CII Institute of Hospitality - ITC Gardenia, Bangalore admissions for all courses are currently ongoing. Interested candidates can apply in online mode through the official website. The online application process is explained in the following steps:
Step 1: Visit the official website of CII Institute of Hospitality - ITC Gardenia, Bangalore (ciiih.com/bangalore).
Step 2: Fill out the form.
Step 3: Complete the form and submit it.
New Question
11 months agoContributor-Level 9
Hello, GNM is a 3.5-year diploma-level programme according to the Indian Nursing Council (INC). This includes 3 years of classroom study with 6 months of mandatory internship in a healthcare facility.
New Question
11 months agoContributor-Level 10
Yes, applications are open at CII Institute of Hospitality - ITC Gardenia, Bangalore for various courses. The mode of application is online. Aspirants need to apply at the official website for admission to the various programme. Candidates can fill out the application form on the institute's official website.
New Question
11 months agoContributor-Level 10
For admission to Magadh University's BA (H) courses, students must possess the minimum qualification as prescribed by the university. As per the eligibility criteria, students who have completed Class 12 from a recognised board of India are eligible for the programme. Students note that the eligibility criteria may differ from college to college.
New Question
11 months agoContributor-Level 10
(a) Silicon is heated with methyl chloride at high temperature in the presence of copper catalyst at 537 K, methyl substituted chlorosilanes MeSiCl3, Me2→SiCl2→, Me3→SiCl and Me4→Si are formed.
(b) When silicon dioxide is treated with hydrogen fluoride, first SiF4→ is formed and then hydro fluorosilicic acid is obtained.
SiO2 →+ 4HF → SiF4 →+ 2H2→O
SiF4+2HF → H2SiF6
(c) When CO is heated with ZnO, ZnO is reduced to Zn metal.
CO + ZnO →CO2 →+ Zn
(d) When hydrated alumina is treated with aqueous NaOH solution, it dissolves to form sodium meta al
New Question
11 months agoContributor-Level 10
1.9. Average atomic mass = (Fractional abundance of 35Cl x molar mass of 35Cl) + (Fractional abundance of 37Cl x molar mass of 37Cl)
= (75.77/100 x 34.9689) + (24.23/100 x 36.9659)
= 26.4959 + 8.9568
= 35.4527
New Question
11 months agoContributor-Level 10
(a) When borax is heated strongly, it loses water and swells into the white mass, which on further heating melts to form a transparent glassy solid called borax glass and borax bead.
Na2B4O710H2O→ Na2B4O7+ 10H2O
Na2B4O7 → 2NaBO2+ B2O3
(b) When boric acid is added to water, it accepts electrons from –OH ion. Boric acid is sparingly soluble in cold water however fairly soluble in hot water.
B (OH)3→ + 2H2→ O→ [B (OH)4→ ]→ + H3→ O+
(c) Al reacts with dilute NaOH to form sodium tetrahydroxoaluminate (III). Hydrogen gas is liberated in the process.
2Al +
New Question
11 months ago
Contributor-Level 10
In order to get admission at CII Institute of Hospitality - ITC Gardenia, Bangalore, candidates must keep certain documents ready. Student can check the list of documents below:
- Scanned Class 10, Class 12 and graduation marksheet
- Leaving Certificate
- Aadhaar Card
- Passport-size photograph
- Signature
Note: The list of documents mentioned above are the general documents. For more details visit the official website.
New Question
11 months agoContributor-Level 10
2.7 Let the three capacitors be
= 2 pF, = 3 pF and = 4 pF
The equivalent capacitance, is given by
= 2 + 3 + 4 = 9 pF
When the combination is connected to a 100 V supply, the voltage in all three capacitors = 100 V
charge in each capacitor is given by the relation, q = VC
Hence for , = 100 = 200 pC = 2 C,
for , = 100 = 300 pC = 3 C,
for , = 100 = 400 pC = 4 C
New Question
11 months agoContributor-Level 9
Among paramedical degree holders, Nurse Anesthetist is one of the popular profiles. The highest salary offered to the CRNAs in India range from INR 10 LPA to INR 20 LPA. Impact Paramedical and Health Institute BSc graduates can also apply for the job profile. However, for selection, the degree holders will have to pass the selection rounds of the employer.
New Question
11 months ago
Contributor-Level 10
Pursuing a PGPEX course form IIM Calcutta is worth it if considering the placement opportunities. IIM Calcutta has released the placement report for its PGPEX batch 2024. According to the report, a total of 67 students participated in the placement process and secured 68 job offers from the participating recruiters. The the highest and average packages offered during IIM Calcutta placements for its PGPEX Class of 2024 stood at INR 52 LPA and INR 32.68 LPA, respectively. The key highlights of the IIM Calcutta placements for its PGPEX Class of 2023 and 2024 are pr
New Question
11 months agoContributor-Level 10
Boric acid contains planar BO33- ions which are linked together through hydrogen bonding shown in the fig.
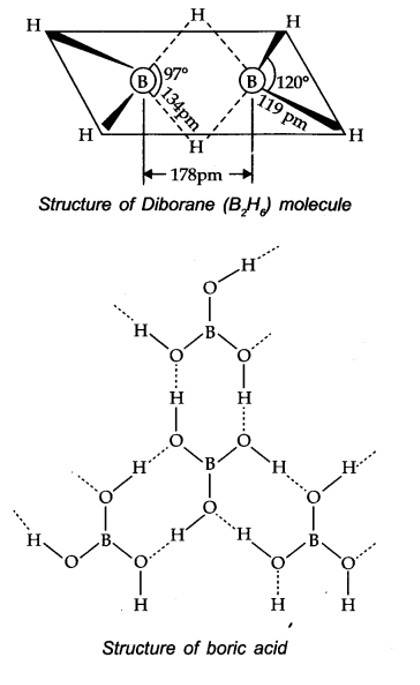
New Question
11 months agoContributor-Level 10
The total tuition fee to pursue a BA (H) at Magadh University ranges from INR 504 to INR 6,200. Other than this, the fee structure includes other charges such as admission fees, hostel fees, development & training fees, etc. Candidates who wish to know the complete fee structure can visit the official website of the university.
Note: The above-mentioned fees is as per the official admission brochure. However, it is subject to change.
New Question
11 months agoContributor-Level 9
Hello, I believe you'll not face many difficulties, though having a PCB in Class 12 has its own advantages, but the GNM course syllabus is not that complex and provides foundational knowledge in Biology. If you are willing to put some extra effort, then you'll complete the course without any delays.
New Question
11 months agoContributor-Level 10
2.6 Capacitance of each of the three capacitors, C = 9 pF
The equivalent capacitance when the capacitors are connected in series is given by
= + = = =
Hence, = 3 pF
Supply voltage, V = 120 V
Potential difference ( ) across each capacitor is given by = = = 40 V
New Question
11 months agoContributor-Level 10
Excess of CO2 absorbs heat radiated by the earth. Some of it is dissipated into the atmosphere while the remaining part is radiated back to the earth. As a result, temperature of the earth increases. This is the cause of global warming.
New Question
11 months agoContributor-Level 10
1.7. 1 mole of CuSO4 contains 1 mole (1 g atom) of Cu
Molar mass of CuSO4= 63.5 + 32 + (4 x 16) = 159.5 g mol-1
Thus, Cu that can be obtained from 159.5 g of CuSO4 = 63.5 g
∴ Cu that can be obtained from 100 g of CuSO4 =63.5/159.5 * 100 = 39.81g
New Question
11 months agoContributor-Level 10
The highly poisonous nature of CO arises because of its ability to form a complex with haemoglobin, which is about 300 times more stable than the oxygen-haemoglobin complex. This prevents haemoglobin in the red blood corpuscles from carrying oxygen round the body and ultimately resulting in death.
New Question
11 months agoContributor-Level 10
Since, anhydrous HF is a covalent compound and weak acid due to high bond dissociation energy. AlF3does not dissolve in HF.
Whereas NaF is ionic compound.
3NaF + AlF3 → Na3 [AlF6]
Na3 [AlF6] + 3BF3 (g)→ AlF3 + 3Na+ [BF]–
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts

