Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months ago
Contributor-Level 10
The Indian Institute of Management Calcutta packages offered to the PGPEX batch 2024 are presented below:
| Particulars | Statistics (2024) |
|---|---|
| the highest Salary | INR 52 LPA |
| Average Salary | INR 32.68 LPA |
| Median Salary | INR 32.27 LPA |
New Question
11 months agoContributor-Level 10
2.5 Capacitance between the parallel plates of the capacitor, C = 8 pF
Let us assume, initially, distance between the parallel plates was d and it was filled with air.
Dielectric constant of air, k = 1
Capacitance C is given by the formula,
C = = , since k = 1…………………….(1)
Where A = area of each plate
= permittivity of free space = 8.854
d = distance between two plates
If the distance between two plates is reduced to half, then distance between two plates =
Dielectric constant of a new substance, = 6
Then the resistanc
New Question
11 months agoContributor-Level 10
B-Cl bond has dipole moment because of polarity. In BCl3, since the molecule is symmetrical (planar), the polarities cancel out and hence the dipole moment is zero
New Question
11 months agoContributor-Level 10
No, candidates looking for course admission at CII Institute of Hospitality- ITC Gardenia, Bangalore cannot enrol for direct admissions. The institute admits students based on CII Aptitude Test + Industry Panel Viva. It also considers the candidates' academic performance for final selection. The final selection of the candidate will be based on the overall performance in the CII Institute of Hospitality- ITC Gardenia, Bangalore admission selection process.
New Question
11 months agoContributor-Level 10
1.6. A mass percent of 69% means that 100 g of nitric acid solution contains 69 g of nitric acid by mass.
Molar mass of nitric acid HNO3= 1 + 14 + (3x16) = 63 gmol-1
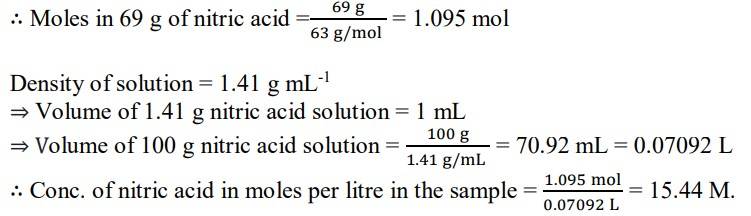
New Question
11 months agoContributor-Level 10
The difference in bond length is due to the difference in the state of hybridisation. In BF3 'B' is sp2 hybridised and in BF4– 'B' is sp3 hybridised.
New Question
11 months agoContributor-Level 10
Yes, BA (H) is available at Magadh University. This undergrad programme is offered via many affiliated colleges. Students who are willing to take admission can check the below table to know the key highlights:
Particulars | Statistics |
|---|---|
Duration | Four years |
Eligibility | Passed Class 12 from recognised board |
Selection Criteria | Merit based |
Fees | INR 504 - INR 6,200 |
Note: The above information has been sourced from various sources. Hence it is indicative.
New Question
11 months agoContributor-Level 10
2.4 Radius of the spherical conductor, r = 12 cm = 0.12 m
Uniformly distributed charge, q = 1.6 C
The electrical field inside the conductor is zero.
Electric field E just outside the conductor is given by
E = where = permittivity of free space = 8.854
E = = 99863.8 N N
At a point 18 cm from the centre of the sphere. Hence r = 18 cm = 0.18 m
From the above equation we get
E at 18 cm from the centre = = 4.438 N
New Question
11 months ago
Contributor-Level 10
The key highlights of the IIM Calcutta placements for its PGPEX Class of 2023 and 2024 are presented below:
| Particulars | Statistics (2023) | Statistics (2024) |
|---|---|---|
| the highest Salary | INR 78.17 LPA | INR 52 LPA |
| Average Salary | INR 30.99 LPA | INR 32.68 LPA |
| Median Salary | INR 30.12 LPA | INR 32.27 LPA |
| Students Registered | 71 | 67 |
| Offers Made | 79 | 68 |
New Question
11 months agoContributor-Level 10
- PbCl2 + Cl2→ PbCl4.
This is because Pb can show +2 oxidation state more easily than +4 due to inert pair effect. - PbCl4→ PbCl2 + Cl2
Because Pb2+ is more stable than Pb4+ due to inert pair effect. - PbI4does not exist because I- ion being a powerful reducing agent reduces Pb4+ ion to Pb2+ ion in solution.
Pb4+ + 2I– → Pb2+ + I2
New Question
11 months agoContributor-Level 10
1.5

Since the molar mass of sodium acetate is 82.0245 g mol-1.
⇒ 1 mol of CH3COONa has a mass = 82.0245 g
∴Mass of sodium acetate (CH3COONa) required to make 500 ml of 0.375 molar aqueous solution
= 0.375 x 0.5 mol x 82.0245 g mol-1
= 15.3795 g = 15.380 g
New Question
11 months agoContributor-Level 10
Yes, Impact Paramedical and Health Institute offers four-year long BSc paramedical courses. The available programmes as per the official website are MLT, Radiology and Imaging Technology, and Operation Theatre Technology. Admission is done on a merit-basis. Moreover, students who are Class 12 or equivalent pass with at least 50% aggregate can apply for admission.
New Question
11 months agoContributor-Level 10
Diamond has a crystalline lattice where each carbon atom undergoes sp3 hybridisation and linked to four other carbon atoms by using hybridised orbitals in tetrahedral fashion. The C–C bond length is 154 pm. The structure extends in space and produces a rigid three- dimensional network of carbon atoms. It is very difficult to break extended covalent bonding and, therefore, diamond is a the hardest substance on the earth.
Graphite has layered structure in which the layers are held by van there Waals forces and distance between two layers is 340 pm. Each layer is composed of planar hexagonal rings of carbon atoms. C—C bond length
New Question
11 months agoContributor-Level 10
2.3 The arrangement is represented in the adjoining figure.

An equipotential surface is the plane on which total potential is zero everywhere. The plane is normal to line AB. The plane is located at the midpoint of the line AB because the magnitude of the charge is same.
The direction of the electric field at every point on that surface is normal to the plane in the direction of AB.
New Question
11 months agoContributor-Level 10
1.4 In order to answer the question, we need to know the balanced equation for the combustion of carbon in dioxygen/air, which can be written as:
C (s) + O2 (g) à CO2 (g)
(i) We can see from the above equation,
1 mole of carbon reacts with 1 mole of oxygen to produce 1 mol of carbon dioxide.
In air, combustion is complete.
Therefore, CO2 produced from combustion of 1 mole of carbon= Molar mass of CO2= 44 g
(ii) As only 16 g of dioxygen is available, it can combine only with 0.5 mole of carbon, i.e., dioxygen is the limiting reactant.
Hence, CO2 produced = 22 g
Here, dioxygen acts as the limiting reagent.
(iii) Here again, dioxygen is
New Question
11 months agoContributor-Level 10
2.2

The charge at each corner, = 5 C
The sides of the hexagon, AB = BC = CD = DE = EF =FA = 10 cm = 0.1 m
Let O be the centre. The electric potential at O is given by
V = , where = permittivity of free space = 8.854
Hence V = = 2.696 V
Therefore, the potential at the centre is 2.7 V
New Question
11 months agoContributor-Level 10
1.3 We are given that the percentage of iron by mass is 69.9% and the percentage of oxygen by mass is 30.1%.
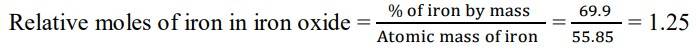
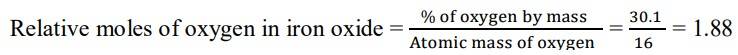
Since we have relative moles of both elements, we can calculate the simpler molar ratio of iron to oxygen
= 1.25: 1.88
Divide by the smaller value to both
= 1.25/1.25: 1.88/1.25
= 1: 1.5
= 2: 3
So, now we can write the empirical formula of iron oxide as Fe2O3.
New Question
11 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
