Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months ago11.9. What are electron deficient compounds? Are BCl3 and SiCl4 electron deficient species? Explain.
Contributor-Level 10
Electron deficient compounds are those in which the central atom in their molecule has the tendency to accept one or more electron pairs. They are also known as Lewis acid. BCl3 and SiCl4 both are electron deficient species.
Since, in BCl3, B atom has only six electrons. Therefore, it is an electron deficient compound.
In SiCl4 the central atom Si has 8 electrons but it can expand its covalency beyond 4 due to the presence of d-orbitals.
New Question
11 months agoContributor-Level 10
There are various institutes in Delhi that offer BSc in paramedical science. The eligibility and selection criteria can vary for these courses from one college to another. IPHI Delhi offers BSc and UG Diploma in MLT, Radiology and Imaging Technology, and Operation Theatre Technology. Admission criteria is merit-based.
Some of the other top ranked and average ranked institutes in Delhi that offer paramedical courses are:
- AIIMS Delhi
- Jamia Hamdard
- Delhi Paramedical and Management Institute
- Vardhman Mahavir Medical College, etc.
New Question
11 months agoContributor-Level 10
Aluminium reacts with acid as well as base. This shows amphoteric nature of aluminium.
2Al (s) + 6HCl (dil.) →2AlCl3 (aq) + 3H2 (g)
2Al (s) + 2NaOH (aq) + 6H2O (l) →2Na+ [Al (OH)4]– (aq) + 3H2 (g)
New Question
11 months agoContributor-Level 10
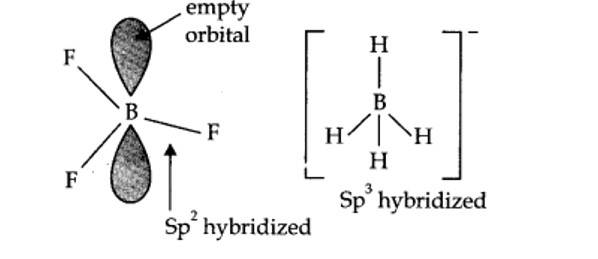
In BF3, boron is sp2 hybridized.
? shape of BF3 = planar.
In [BH4]–, boron is sp3 hybridized, thus the shape is tetrahedral.
New Question
11 months agoContributor-Level 10
1.2 Molar mass of Na2SO4= (2 x Atomic mass of Sodium) + Atomic mass of Sulphur + (4 x Atomic mass of Oxygen)
= (2x 23) + 32 + (4x 16)
= 46 + 32 + 64
= 142 g/mol

New Question
11 months agoContributor-Level 8
The average cost of studying in the UAE for international students varies greatly as per the location, college and course, potentially ranging from AED 73,500 to AED 152,000 or higher, encompassing tuition fees (AED 37,500 - AED 110,000+ per year) and living expenses (AED 3,000 - AED 6,000+ per month).
Dubai and Abu Dhabi on the other hand tend to be more expensive than the other emirates in the UAE. While the above-mentioned ones are the general costs, additional costs of studying in the UAE include visa fees, health insurance, and textbooks. Note that the scholarships to study in the UAE are available at some universities.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
On heating boric acid above 370 K, it forms metaboric acid, HBO2 which on further heating yields boric oxide B2O3.
H3B2O3 → HBO2 → B2O3
New Question
11 months agoBeginner-Level 5
A p-type semiconductor is electrically neutral despite having more holes, because the number of positively charged holes is exactly balanced/equal by the number of negatively charged acceptor ions introduced during doping. so practically untill any volatage is applied the semiconductor remains chargeless in other words doesn't produce any current even after doping.
As per the NCERT Textbooks information"Although the number of holes is more than the number of electrons in a p-type semiconductor, the material as a whole is electrically neutral because the charge of holes is balanced by the negatively charged acceptor ions.”
New Question
11 months agoContributor-Level 10
Boric acid is a Lewis acid since it accepts electrons from hydroxyl ion of H2O molecule. It is not a protic acid.
B (OH)3 + 2HOH → [B (OH)4]– + H3O+
New Question
11 months agoContributor-Level 10
CII IH, Bangalore offers UG Diploma and Degree in Tourism and Hospitality courses. The selection criteria are based on the CII Aptitude Test and Industry Panel Viva. To get admitted to the CII Institute of Hospitality - ITC Gardenia, Bangalore, candidates must meet the eligibility criteria set by the institute. Students can check the table below to know detail course-wise selection criteria at CII Institute of Hospitality - ITC Gardenia, Bangalore:
| Courses | Eligibility | Selection Criteria |
|---|---|---|
| Swiss Professional Diploma | Class 12 with 50% aggregate | CII Aptitude Test and Industry Panel Viva |
| Degree in Tourism and Hospitality | Class 12 | CII Aptitude Test and Industry Panel Viva |
New Question
11 months agoContributor-Level 10
2.1

Let the charges be
= 5 C and = -3 C
Distance between the two charges, d = 16 cm = 0.16 m
Consider a point P on the line joining the two charges
R = distance of the point P from the charge
Let the electrical potential (V) at point P is zero
Potential at point P caused by charges and respectively.
V = ………………………….(1)
Where = permittivity of free space
For V = 0, the equation (1) becomes
or = or =
= or =&nbs
New Question
11 months agoContributor-Level 8
The UAE, particularly Dubai and Abu Dhabi, is a good study destination due to its quality education, diverse environment, safety, and English-taught programs. While the cost of living can be high, it offers job opportunities and modern infrastructure. Sharjah and Ajman are also notable educational hubs.
New Question
11 months agoContributor-Level 10
In BCl3, there is only six electrons in the valence shell of B atom. Thus, the octet is incomplete and it can accept a pair of electrons from water and hence BCl3 undergoes hydrolysis. Whereas, in CCl4, C atom has 8 electrons and its octet is complete. That's why it has no tendency to react with water.
New Question
11 months agoContributor-Level 10
Impact Paramedical and Health Institute offers BSc courses in different specialisations. The admission criteria is straight forward. Candidates get selected based on performance in the last qualifying examination, i.e., Class 12. For a fair course comparison, the below-mentioned parameters can be considered:
- Fees
- Quality of faculty
- Location, etc.
New Question
11 months agoContributor-Level 10
In BF3, central atom has only six electrons after sharing with the electrons of the F
atoms. It is an electron-deficient compound and thus behaves as a Lewis acid.
New Question
11 months agoContributor-Level 10
BCl3 is quite stable. Because there is absence of d- and f-electrons in boron three valence electrons (2s2 2px1) are there for bonding with chlorine atom. In Tl the valence s-electron (6s2) are experiencing maximum inert pair effect. Thus, only 6p1 electron is available for bonding. Therefore, BCl3 is stable but TlCl3 is comparatively unstable.
New Question
11 months agoContributor-Level 10
(i) B to Tl
Common oxidation states are +1 and +3. The stability of +3 oxidation state decreases from B to Tl while +1 oxidation state increases from B to Tl.
(ii) C to Pb
The common oxidation states are +4 and +2. Stability of +4 oxidation state decreases from C to Pb.
New Question
11 months agoBeginner-Level 5
As per NCERT Textbboks"If forward current is too large, it can produce large heating and damage the junction. So, a resistor is used in series with the diode to limit the current in the circuit.”
It means when a semiconductor diode is connected to a source under high current it causes excess heat due to more electrical energy. This excess heat is responsible to damage the junction in semiconductor diode permanently. Student can check out NCERT Solutions for Semicondutor Electronics of class 12 physics.
New Question
11 months agoContributor-Level 10
Impact Paramedical and Health Institute (IPHI) Delhi admission is granted to students who meet eligibility criteria. Students who are Class 12 pass with at least 50% can apply. The admission form can be obtained and submitted in offline mode to the admission office. Applications are then evaluated and shortlisted based on performance in the qualifying examination. The selected students are then informed. These students can pay the admission fee amount to confirm their admission.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts

