Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 7
For admission to United School of Business Management courses, students must pass Class 12 or a three-year Diploma from a state Board of Technical. There is no any particular percentage has been mentioned by the college. However, students must try to apply with a minimum 50% aggregate to seek a better chance for getting a seat.
New Question
10 months agoContributor-Level 10
The total student population at the University of Mannheim is around 11,908, per data curated from official sources, as of Fall 2023.
The University of Mannheim student population comprises of around 7,052 UG students & 3,336 PG students.
The institute welcomes students from over 100 countries.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (D)
(A) The number of moles is given by the following formula,
Moles = ….(1)
The number of moles of He is calculated by using equation (1) as follows
Moles of O2 = =1 mol
The number of atoms can be calculated as, number of moles
….(2)
On substituting the values in the above equation:
1 mol =
Number of atoms = 1 6.022 1023
(B) The number of moles of Na is calculated by using equation (1) as follows,
Moles of Na = = 2 mol
The number of atoms can be calculated by using equation (2) as follows,
2 mol =
number of atoms= 2 6.022 1023
(C) The n
New Question
10 months agoContributor-Level 10
No, the University of Mannheim is not free for international students. Students have to pay a tuition fee every semester. The per semester cost of study is very much affordable.
Students enrolled in full-time studies at the Uni Mannheim have to pay a per semester tuition fee worth EUR 1,500 (INR 1.6 L) along with a semester fee of around EUR 194 (INR 20.8 K).
1 EUR = INR 107.38
New Question
10 months agoContributor-Level 10
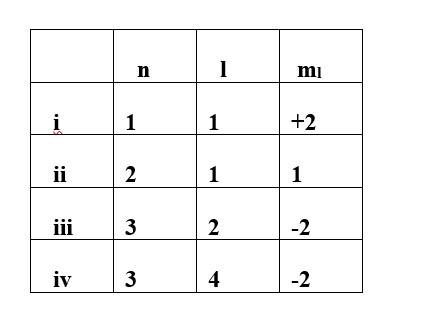
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (i)& (iv)
- Principal quantum number (n): It represents the size and energy of orbitals.
- Azimuthal quantum number (l): It represents the subshell and shape of orbitals.
- Magnetic quantum number (ml): It represents the orientation of the orbitals.
- Spin quantum number (ms): It represents the direction of spin of electrons in the orbitals.
New Question
10 months agoContributor-Level 10
The University of Mannheim acceptance rate stands somewhere between 20-35%, per data curated from unofficial sources.
This suggests the institute is only known to accept about 20-35 applications for every 100 it receives, which makes it selective to a very decent extent.
Prospective applicants, must maintain a solid academic record to have the best possible chance of admissions at the institute.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (i) & (iii)
Isoelectronic species are those species that possess the same number of electrons. Here both Na+, Mg2+ possess 10 electrons and both Na+, O2– possess 10 electrons.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (B)
The relation between molarity and volume is given as,
M1 V1= M2 V2
On substituting the value in the above equation, the political can be calculated as
5M* 500 mL = M2*1500 mL
M =1.66M
New Question
10 months agoContributor-Level 10
The University of Mannheim ranks #29 among all colleges & universities in Germany, per the latest national rankings rolled out by Shiksha.
The institute also secures a global rank #285, as part of Shiksha's the latest version of its Global University Rankings 2026.
New Question
10 months agoContributor-Level 8
An MBA degree is basically obtained by students for moving ahead in their careers or if they are looking forward to a career change. As far as a Master in Management (MIM) degree is concerned, it helps students who have just started their career gain knowledge to rekindle their business careers. MIM is mostly pursued by young students who hold zero to little work experience.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (ii) & (iii)
For the given value of n (principal quantum number) the value of l (Azimuthal quantum number) varies from 0 to n-1. However for the given value of l the ml (magnetic quantum number) varies from -1 to +1.
New Question
10 months agoContributor-Level 10
The University of Mannheim is known for offering UG, PG & doctoral courses across a number of prominent industry relevant streams, such as Business Management, Healthcare, Data Science, Law & Political Science.
Further, the institute offers a number of scholarships to international students making it easier for them to afford their studies.
The overall costs of studying & living in Mannheim is also generally lower than other major German cities such as Munich, Stuttgart, Berlin, Frankfurt & Hamburg.
New Question
10 months agoContributor-Level 7
To apply for admission to United School of Business Management, students need to fulfil the basic educational requirements of the respective courses as follows:
| Course Name | Eligibility |
|---|---|
| MBA | Graduated in Engineering/ Technology/ Pharmacy examination of minimum four/ five years duration in any discipline from any University of Odisha or from a recognised University with minimum 50% aggregate |
| MCA | Bachelor's Degree examination of minimum - Must have passed Class 12 or graduation with Mathematics as one of the subject |
| BBA | Pass Class 12 or a three-year Diploma from a state Board of Technical |
| BCA | Pass Class 12 or a three-year Diploma from a state Board of Technical |
New Question
10 months agoContributor-Level 10
The placements at Bunts Sangha's Ramanath Payyade College of Hospitality Management Studies are good. The following table presents the placement statistics for Bunts Sangha's Ramanath Payyade College of Hospitality Management Studies.
Particulars | Placement Statistics (2025) |
|---|---|
Placement Rate | 100% |
No. of students placed | 108 |
the highest package | INR 3 LPA |
Average package | INR 2.5 LPA |
No. of Companies Visited | 40 |
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (i) & (iv)
In a multielectron atomic system the energy of an electron depends not only on its principal quantum number (shell), but also on its azimuthal quantum number (subshell). Electrons having the same shells and same subshells have the same energy and they are known as degenerate orbitals.
New Question
10 months agoNew Question
10 months agoContributor-Level 8
TCG offers admission on the basis of merit. Therefore, candidates must secure a minimum aggregate of 55% in PG level courses such as MSc/ MCom/ MA/ MPhil/ M.Tech or equivalent. Candidates with 75% in 4 year UG level courses are also eligible to pursue PhD at TCG Crest. Furthermore, the Institute provides 5% marks relaxation to the SC/ ST/ OBC/ EWS, differently abled category candidates.
New Question
10 months agoContributor-Level 10
Yes, Amity School of Fashion Technology does offer an MDes course for the duration of two years, divided into four semesters, and offered in full-time mode. The institute offers an MDes course in one specialisation i.e., MDes in Fashion and Textiles. The basic eligibility criteria required by the institute to get admission into the MDes course are that candidates must complete their BDes degree with an aggregate of 50%. Additionally, the admission process is based on merit evaluated on the basis of candidates' previous academic years.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (iii) & (iv)
The isotopes are defined as atoms with identical atomic numbers but different mass numbers are known as isotopes.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (C)
No. of mole given =
On substituting the value in the above equation, the cal, can be calculated as
no, of mole = = 0.1 g
The molarity (M) is given by the formula:
M =
On substituting the values in the above equation:
Molarity =
= 0.2 mol L-1
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts