Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The uncertainty principle is significantly only for the microscopic particles and not for the macroscopic particles can be concluded by considering the following example. Let us consider a particle or an object of mass 1 milligram i.e. 10-6 kg Then its uncertainty can be calculated as,
? x ? v = 6.626 10-34 / 4x 3.14 106
= 10-28 m-2 s-1
Thus, the value obtained is negligible and insignificant for the uncertainty principle to be applied to this particle.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Given, m (mass) = 10g, v (speed) = 90 m/s and accuracy = 4%
Uncertainty in speed = 3.6 ms-1
Uncertainty in position = h/4 πmΔv = 6.626 × 10-34/4 × 3.14 × 10 × 3.6
=1.36 × 10-33m
New Question
10 months agoContributor-Level 10
Given below is a list of some necessary documents required to be submitted for BA Geography admission:
- Passport Size Picture
- Class 10 and 12 marksheets
- ID Proof
- Adhaar Card
- Migration Certificate (Optional)
- Entrance Exam Scorecard (Optional)
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
According to the law of multiple proportions, when two elements react to form two or more than two chemical compounds, the ratio between different masses of one of the elements combining with a fixed mass of the other is always in the ratio of tiny numbers.
Example:
1. Compounds of carbon and oxygen:
C and O react to form two different compounds CO and CO2. In CO, 12 parts by mass of C reacts with 16 parts by mass of 0 .
In CO2 ,12 parts by mass of C reacts with 32 parts by mass of O .
If the mass of C is fixed at 12 parts of mass then the ratio in the masses of oxyg
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans:


New Question
10 months agoContributor-Level 10
Yes, students enrolled in any of the BCom courses offered by Takshashila University are offered many scholarships and fee waiver schemes to choose from. BCom scholarships are distributed based on students' performance in the qualifying exam. However, these scholarships are offered on a first-come, first-served basis. Thus, students interested in availing a scholarship are encouraged to apply timely.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: We know that
λ= c/υ Given,
υ = 4.620 × 1014 Hz
Thus, λ= c/υ = (3.0 × 108 m/s)/ (4.620×1014 Hz) = 649.4nm
This frequency falls under visible range
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
The volume of HCl solution is 250 mL and its molarity is 0.76M.
The number of moles of HCl as follows,
Moles of HCl Molarity Volume (in L)
= 0.76M 0.250 L
= 0.19 mol
The molar mass of CaCO3 is 100 g / gQl and the mass of CaCO3 is given as 1000 g
The number of moles of CaCO3 is calculated as
Moles of CaCO3 = M a s / M o l a r m a s
= 1000 g / 100 g / m o l = 10 mol
According to the given reaction, 1 mole of CaCO3 requires 2 moles of HCl. So, the required number of moles of HCl for 10
New Question
10 months agoContributor-Level 10
Yes, JK Business School offers a scholarship of INR 50,000 to its BBA students who take admission into the PGDM programme. This scholarship is a financial incentive and will be evenly adjusted across all fee installments during the PGDM course duration.
New Question
10 months agoContributor-Level 10
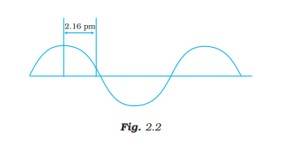
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The wavelength is defined as the distance between two consecutive crests or troughs of a wave, and it is denoted by l .
l = 4*2.16 pm = 8.64 pm
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: We have
λ = h/mv
Thus, the equation signifies that in order to have the same wavelength the electron should have higher velocity as the mass of the proton is higher than that of the electron
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The line spectrum associated with any element possesses lines corresponding to specific wavelengths and these lines are obtained as a result of electronic transitions between the energy levels. Hence, the electrons in these levels have fixed energy i.e., quantized values.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
P1=1 atm
P2= 1/2=0.5 atm
T1=273.15 K
V2=?
V1=?
32 g dioxygen occupies = 22.4 L volume at STP
∴ 1.6 g dioxygen will occupy = 22.4L x 1.6g / 32g = 1.12 L
V1=1.12 L
From Boyle's law (as temperature is constant)
p1V1=p2V2
V2=p1V1p2
= 1 atm x 1.12 l/0.6 atm g = 2.24 L
(ii) Number of molecules of dioxygen.
New Question
10 months agoContributor-Level 10
June 8 was the deadline to submit AIAPGET 2025 application form. Candidates have to register online and submit application form for exam. Registration for exam had started on May 9, 2025. Correction window to modify incorrect details uploaded with AIAPGET 2025 form was opened from June 10 to June 12, 2025. The exam was held on July 4, 2025.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: As per de Broglie, every object in motion has a wave character i.e. every object/matter have dual nature both particle and wave nature. But the wavelengths associated with ordinary objects are so short (because of their large masses) that their wave properties cannot be detected.
Given, m (mass) = 100g, v=100km/hr
We have
λ = h/mv= 238.5 × 10-34 m
As the wavelength associated with the cricket ball is so small that it can’t be detected.
New Question
10 months agoContributor-Level 6
The UPSC ESE/IES 2025 cutoff is determined based on the difficulty level of the exam (prelims and mains), the number of vacancies, previous years' trends, and the overall performance of the candidates. The cutoff marks may vary slightly across categories and branches.
New Question
10 months agoContributor-Level 10
BArch graduates from Mohamed Sathak A.J. Academy of Architecture can explore a wide range of career opportunities with competitive salary packages. Depending on the role and experience, average CTCs range from INR 7 LPA for Architecture Designers to INR 19.40 LPA for Senior Principal Architects. Other prominent roles include Design Manager, Project Manager, and Principal Architect, reflecting the strong career potential after graduating from MSAJAA.
New Question
10 months agoNew Question
10 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts