Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
The highest salary offered to a BA Geography candidate can vary significantly depending on the job profile which the candidate opts for as well as experience. Job profiles such as Urban Planner, Environmental Consultant, GIS Specialist, etc. are among the highest paying job roles in this field.
Experienced professionals in these job profiles with relevant skills can earn around INR 8-10 LPA.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Molality is the number of moles of substance (also known as the solute) found in a given mass of solvent (in Kg ) in which it is dissolved. Molality is calculated by using the formula.
Molality =
So, temperature has no effect on the molality of the solution because molality is expressed in mass.
New Question
10 months agoTotal number of orbitals associated with third shell will be __________.
(i) 2
(ii) 4
(iii) 9
(iv) 3
Contributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (iii) 9
The total number of orbitals is given by n2, where n is the principal quantum number or the principal shell.
Thus for the third shell number of orbitals is 32 i.e. 9
New Question
10 months agoContributor-Level 10
Following are some tips to improve the writing and presentation skills for the Andhra Pradesh SSC board exams.
- Students must write the answers neatly. The hand writing should be legible.
- Write the answers with proper heading and subheading.
- Highlight your important points.
- Cross out the errors neatly with single line.
- Divide the time for each question to avoid rushing.
- Practice writing the previous year question papers.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Mass of NaoH = 40 g
Mass of solvent =1000 g
Mass of solution = 40 x 3+1000
Density =
=
= = 1009.0 mL
Molarity = = 2.97M
1009.00 mL= 1.009 L
Hence, the molarity of the solution is 2.97M
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: (ii) Heisenberg's uncertainty principle.
According to Heisenberg's uncertainty principle it is impossible to determine simultaneously, the exact position and exact momentum (or velocity) of an electron. Thus it implies that determining the trajectory of an electron is impossible as it requires exact position and velocity which is not possible as per the uncertainty principle.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
65.3 g of Zinc gives 22.7 litres of Hydrogen gas
32.65 g Zinc gives = 32.65g x 22.7 litres/65.3 = 11.35 L
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (iii) 2
The number of angular nodes is given by n-l
where n is principal quantum number, l is azimuthal quantum number
For 4d orbital, n=4 and l=2
Thus, the number of angular nodes
= n-l
= 4-2
= 2
New Question
10 months agoContributor-Level 10
Students who have done a BCom from Takshashila University can pursue jobs offered by the government sector of India. The public sector has many such jobs that require the knowledge and skills gained during a BCom programme. Tabulated below are some of the prospective government jobs along with their average salaries:
Job Profile | Average Salary |
|---|---|
Bank Probationary Officer (PO) | INR 5 LPA to INR 7 LPA |
IBPS PO | INR 3 LPA to INR 4 LPA |
IBPS Clerk | INR 2 LPA to INR 3 LPA |
RBI Assistant/Grade B Officer | INR 6 LPA to INR 8 LPA |
Note: The above-mentioned are industry-based average payouts. The actual salary may differ from student to student based on their skills and competency.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: option (iv)
The number of radial nodes is given by n-l-1
where n is principal quantum number, l is azimuthal quantum number
For 3p orbital, n=3 and l=1
Thus, the number of radial nodes
= n-l-1
= 3-1-1
= 1
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
(Natural abundance of 1H x molar mass ) + (Natural abundance of 2H x molar mass of 2H)
Natural abundance of 1H = 99.985
Natural abundance of 2H = 0.015
Average atomic mass =
= 1.00015u
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iv)
sum of the number of protons and neutrons is same but the number of protons is different
Isobars are the atoms which have same mass number (sum of number of neutrons and protons) but different atomic number (i.e. different proton number)
For example, 146C and 147N.
New Question
10 months agoContributor-Level 10
Some widely opted job profiles by the BA Geography course graduates are mentioned in the table given below:
- Cartographer
- GIS Technologist
- Urban Planner
- Climatologist
- Environmental Consultant
- Geographer
- Hydrologist
Read more:
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i)
Overall neutrality of atom.
According to the J.J Thomson model the positive charge is uniformly distributed and the electrons are embedded into it in such a manner as to give the most stable electrostatic arrangement just as watermelon of positive charge with plums or seeds (electrons) embedded into it. Thus this model is able to explain the overall neutrality of the atom
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
(a) Is this statement true?
Ans: Yes the given statement is true
(b) If yes, according to which law?
Ans: Multiple law of proportions: According to the Law of Multiple proportions, when two elements combine to generate more than one compound, the weights of one element that combine with a fixed weight of the other are in a ratio of tiny whole numbers.
(c) Give one example related to this law
Ans:
C (g) + O (g) -> CO (g)
12 g 16 g 28 g
C (g) + O2 (g) -> CO2 (g)
12 g 32 g &n
New Question
10 months ago
Contributor-Level 10
United School of Business Management fee for UG and PG courses vary. Candidates need to pay a certain amount of fee at the time of admission to the respective courses. Check below the course-wise total fee:
- MBA - INR 3.05 Lakh
- MCA - INR 2.30 Lakh
- Executive MBA - INR 1.50 lakh
- BBA - INR 1.80 Lakh
- BCA - INR 1.80 Lakh
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
The mass of an electron is equal to the mass of a neutron.
The neutron is heavier than the electron as
Mass of the neutron = 1.67 x 10-27 kg
Mass of the electron = 9.11 x 10-31kg
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iv)
Characteristics of cathode rays depend upon the nature of gas present in the cathode ray tube. As per the result obtained from the cathode ray discharge tube experiment the characteristics of cathode rays (electrons) does not depend upon the material of electrodes and the nature of the gas present in the cathode ray tube. Which concludes that electrons are the basic constituent of all the atoms
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
If all gases are at the same temperature and pressure, Gay lussac's law of gaseous volumes states that gases combine or are created in a chemical reaction in a simple volume ratio.
H2 (g) + Cl2 (g) → 2HCL (g)
1 volume 1 volume 2 volume
22.4 litre 22.4 litre 44.8 litre
2N2 (g) +
New Question
10 months agoContributor-Level 10
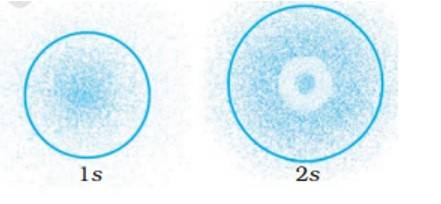
This is a Multiple Choice Questions as classified in NCERT Exemplar
ANS – Option (iv)
The probability density of electrons for 2s orbital decreases uniformly as distance from the nucleus increases. Electrons at the 1s orbital decreases as we move far from the nucleus, however in case of 2s the probability decreases initially then it increases with the distance and thereafter at a certain point it starts decreasing with the distance.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts