Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: We have V = 109677 [1/ni 2 - 1/nf 2 ] cm-1
Given, ni = 2, nf=4
V =109677 [1/4 - 1/16] = 20564.44 cm-1
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: As per the Hund's rule the half filled and fully filled orbital leads to the extra stability due to the symmetry thus fully filled 3d and half filled 4s is preferred.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans:

We know that energy is inversely proportional to the wavelength

Thus, the increasing order of energy is B
New Question
10 months agoContributor-Level 10
The National Institute of Public Cooperation and Child Development in India several universities and institutions offer their Ph.D. programs in the specialisation of Home Science.
Some of the best options include - Banaras Hindu University, Delhi University, Calcutta University, Aligarh Muslim University, Banasthali University etc.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The mass number (A) is defined as the sum of the number of protons and neutrons present in the nucleus and the atomic number is defined as the number of protons or electrons present in an atom. Thus A=13, the number of neutrons is 7 so the number of protons is 6. Thus, the atomic number is 6.
New Question
10 months agoContributor-Level 10
Mohamed Sathak A.J. Academy of Architecture offers separate, well-furnished hostels for boys and girls with modern amenities and hygienic dining facilities. The girls' hostel, located 5 km away in a prime, secure area, accommodates up to 213 students. The campus also promotes holistic development through a variety of sports facilities for BArch students, including basketball, football, cricket, indoor games, and more, encouraging both physical fitness and team spirit.
New Question
10 months agoContributor-Level 8
The K-LAT Exam Date 2026 has been announced on the official website. The K-LAT 2026 exam is on January 18, 2026. Candidates who wish to appear in the KIITEE Law Exam must first fill the application form. The KIITEE Law Application Form is avaiable only in online mode. Candidates who will complete the application process on or before the deadline will be allowed to appear in the K-LAT Exam.
New Question
10 months agoContributor-Level 10
As such, there is no particular age limit prescribed for enrolling into BA Geography course. Aspiring candidates are required to secure atleast 50% marks in their Class 12 in any stream from a recognised board of India. Admissions to BA Geography courses are both merit and entrance based.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The charged particles get deflected by the electric field. As among the given option only neutron is the particle that is neutral thus it does not get deflected by the electric field.
New Question
10 months agoContributor-Level 8
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Based upon the above information, arrange the following orbitals in the increasing order of energy.
(a) 1s, 2s, 3s, 2p
(b) 4s, 3s, 3p, 4d
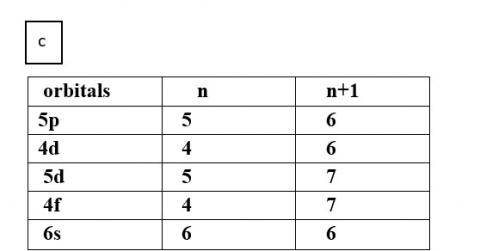
(c) 5p, 4d, 5d, 4f, 6s
(d) 5f, 6d, 7s, 7p

Thus, the increasing order of energy is 3s<3p<4s<4d
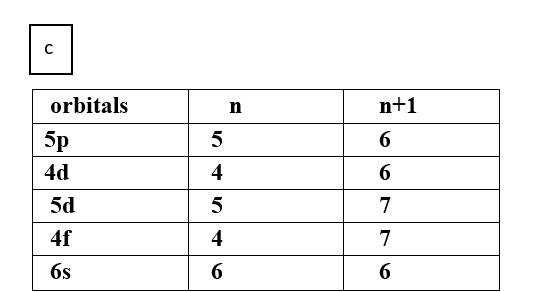
Thus, the increasing order of energy is 4d<5p<6s<4f<5d
Thus, the increasing order of energy is 7s<5f<6d<7p
(II) Based upon the above information, solve the questions given below :
(a) Which of the following orbitals has the lowest energy?
4d, 4f, 5s, 5p
Ans -
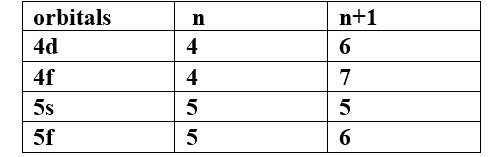
The lowest
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The number of radial nodes is given by n-l-1, where n is principal quantum number, l is azimuthal quantum number. The number of angular nodes is given by n-l, where n is principal quantum number, l is azimuthal quantum number. Here n =3 and l =1 Thus, angular nodes = 3-1 = 2 and radial node = 3-1-1 = 1.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: In a multielectron atomic system the energy of an electron depends not only on its principal quantum number (shell), but also on its azimuthal quantum number (subshell). Electrons having the same shells and same subshells have the same energy and they are known as degenerate orbitals.
Thus 3dxy ,3dz2 , 3dyz and 4dxy, 4dyz, 4dz2 are degenerate.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The electronic configuration of nickel is 1s2 2s2 2p6 3s2 3p6 3d8 4s2 , thus it loses electrons from its 4s orbital as its energy is higher compared to the other orbital.
New Question
10 months agoContributor-Level 10
As mentioned in the National Institute of Public Cooperation and Child Development, in India, several universities and institutions offer their Ph.D. programs in the specialisation of Home Science. Some of the best options include Banaras Hindu University, Delhi University, Calcutta University, Aligarh Muslim University, Banasthali University, etc. These universities are renowned for their top-notch infrastructure, faculty and curriculum.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The electronic configuration of oxygen atom is 1s2 2s2 2p4 and its orbital diagram is given by

New Question
10 months agoContributor-Level 10
For students applying to MSAJAA BArch programme, key NATA 2025 dates include exam slots from 13–14 June, 20–21 June, and 27–28 June. An additional round is tentatively scheduled for August 2025. Candidates should regularly check the official NATA website for updates and register early to secure their preferred dates. A valid NATA score is mandatory for admission.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The effective nuclear charge (Zeff) is defined as the net positive charge experienced by the outermost electrons in the atom. With the increase of Azimuthal quantum number (l) the Zeff experienced by the electron decreases. Hence the arrangement of subshells in the increasing order of Zeff is : d
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts