Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Yes, Khatima Institute of Technology and Management (KITM Group of Institutions) admissions are open. Interested students can visit the official website of the institute to start the registration process for the desired programme. The application process generally commences online. All the latest updated about the admission process of the institute can be accessed via the website itself. In case of any questions, students can connect with the admission desk of the college.
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) Tetrahedral (c) sp3
(ii) Trigonal (a) sp2
(iii) Linear (b)sp
Explanation :-
(i) A tetrahedral molecule has four electron pairs and these make sigma bonds with each other. The s and p orbitals overlap with each other thus forming a sp3 hybridized molecule.
(ii) There are two possibilities for the central atom to have sp2 Either all the bonds are in place i.e, the bond pairs are all sigma bonds or pi bonds or there are only two bonds and one lone pair of electrons.
(iii) The sp hybridization involves the mixing of the val
New Question
10 months agoContributor-Level 10
There are about 20 top LLB colleges in Delhi. Some of them are mentioned below along with their tuition fees:
| Low-cost Colleges | Tuition Fee |
|---|---|
| Chanderprabhu Jain College of Higher Studies and School of Law LLB | INR 4.44 lakh |
| Distance learning Centre-Karnataka State Open University LLB | INR 60,000 |
| Campus Law Centre, University of Delhi LLB | INR 18,030 |
| Lovely Professional University, Admission Office LLB | INR 4.8 lakh |
| Law Centre-I, University of Delhi LLB | INR 18,030 |
Disclaimer: This information is sourced from official website.
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) Hydrogen bond (d) HF
(ii) Resonance (e) O3
(iii) Ionic bond (b) LiF
(iv) Covalent solid (a) C
Explanation :-
(i) In hydrogen fluoride HF, hydrogen forms
New Question
10 months agoContributor-Level 10
The table below lists the top-ranked MBA colleges in Chennai, based on the rankings released by the NIRF for the year 2025.
College Name | NIRF 2023 | NIRF 2024 | NIRF 2025 |
|---|---|---|---|
31 | 34 | 37 | |
| Loyola Institute of Business Administration Ranking | -- | 66 | 55 |
-- | -- | 56 | |
Saveetha Institute of Medical and Technical Services Ranking | 71 | 74 | 63 |
Disclaimer: This information is sourced from the official website and may vary.
New Question
10 months agoContributor-Level 10
Yes, BBA course is available at Creative Techno College. The institute offers BBA courses at the UG level. Candidate must meet the eligibility criteria to enrol for BBA course admission set by the college. Aspirants must pass Class 12. Creative Techno College's admission process is merit-based.
New Question
10 months agoContributor-Level 10
There are 4 top MBA colleges in Chennai where students can study. These include the Great Lakes Institute of Management, the Loyola Institute of Business Administration, Saveetha Institute of Medical and Technical Sciences, and the Hindustan Institute of Technology and Science.
New Question
10 months agoContributor-Level 8
The KIITEE Law Entrance Exam or K-LAT 2026 will last for 150 minutes, or two hours and thirty minutes. Applicants must finish:
- There are 120 multiple-choice questions that span legal aptitude, logic, English, GK, and numeric aptitude.
- Allotted without any sectional time constraints, inside a rigid 150-minute time period
There is a penalty for erroneous responses, and each right answer receives +3 marks (or +4 in some patterns). Since the entire work needs to be finished in this amount of time, make sure you divide your time effectively across the sections.
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) NO (c) 2.5
(ii) CO (d) 3.0
(iii) O22- (a) 1.5
(iv) O2
New Question
10 months agoContributor-Level 10
This is a Mathematical Reasoning Solutions Type Questions as classified in NCERT Exemplar
(i) Here, 'and' is the connecting word
The component statements are–
(a) All rational numbers are real
(b) All real numbers are not complex
(ii) Here, 'or' is the connecting word
The component statements are–
(a) Square of an integer is positive
(b) Square of an integer is negative
(iii) Here, 'and' is the connecting word
The component statements are–
(a) The sand heats up quickly in the Sun
(b) The sand does not caool down fast at night
(iv) Here, 'and' is the connecting word
The component statements are–
(a) n = 2 is the root of the eq
New Question
10 months agoContributor-Level 10
The key highlights of IIT Guwahati placements 2023 and 2024 for MDes are presented below:
Particulars | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|
| the highest package | INR 52 LPA | INR 51.03 LPA |
| Average Package | INR 21.94 LPA | INR 18.96 LPA |
| Median Package | INR 17.46 LPA | INR 14.32 LPA |
| the lowest Package | INR 5.50 LPA | INR 8 LPA |
| Students Placed | 20 | 22 |
New Question
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) H3O+ (e) Pyramidal
(ii) HC? CH (a) Linear
(iii) ClO2- &nb
New Question
10 months agoContributor-Level 10
Getting a BSc seat at Ganpat University is fairly easy. To begin, students who are eligible and willing to take admission must visit the university's official website and apply for the preferred BSc/BSc Hons course. After registering, students must stay updated with the release of the merit list. For a few courses, students might be evaluated based on Class 12 marks. Those who are offered seats must complete the seat confirmation process.
New Question
10 months agoContributor-Level 10
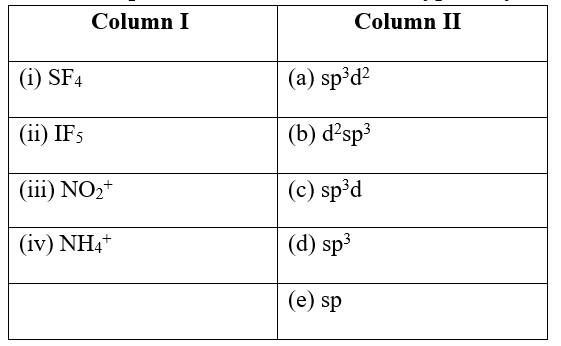
This is a Matching Type Questions as classified in NCERT Exemplar
Ans:
(i) SF4 (c) sp3d
(ii) IF5 (a) sp3d2
(iii) NO2+ (e) sp
(iv) NH4+ &nbs
New Question
10 months agoBeginner-Level 5
Pronouns for non-living objects in singular form are It and Its. In plural form, these pronouns are They, Them, These and Those. These pronouns are also applicable for living things whose gender cannot be determined such as animals, birds, etc.
New Question
10 months agoContributor-Level 10
This is a Mathematical Reasoning Solutions Type Questions as classified in NCERT Exemplar
(i) The component statements are
(a) Number 3 is prime
(b) Number 3 is odd
Here, both the statements are true.
(ii) The component statements are
(a) All integers are positive
(b) All integers are negative
Here, both the statements are false.
(iii) The component statements are
(a) 100 is divisible by 3
(b) 100 is divisible by 11
(c) 100 is divisible by 5
Here, the statements (a) and (b) are false and c) is true.
New Question
10 months agoContributor-Level 8
The general KIITEE 2026 announcement, which usually takes place in the first week of November 2025, is anticipated to include the KIITEE Law Exam 2026 notification. This timeline corresponds with the official admissions calendars for all KIITEE-based programs, including law schools.
When it is made available, the notification will contain:
- Date of application form release
- Information about the exam pattern, syllabus, and eligibility
- Deadlines (the final day to book a spot, submit, etc.)
Set the date for early November 2025 in your calendar. For the most recent bulletin, visit the official KIITEE Law website at
New Question
10 months agoContributor-Level 10
GU offers comprehensive BSc and BSc Hons courses. For admission to these courses, the university has 780 seats available. This means that 780 students can take admission into this course. Students must also note that this seat intake has been sourced from official sources. However, it is subject to change.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i) and option (ii)
(i) NaCl being an ionic compound is a good conductor of electricity in the molten state. It is not a bad conductor of electricity in solid state. So, the statement mentioned in the question is not correct. Hence, this is the correct answer.
(ii) There is no difference in the arrangement of atoms in the canonical structures. The difference is in the arrangement of electron pairs in the canonical structures. So, the statement mentioned in the question is not correct. Hence, this is the correct answer.
(iii) The hybrid orbitals have the same
New Question
10 months agoContributor-Level 10
This is a Mathematical Reasoning Solutions Type Questions as classified in NCERT Exemplar
(i) The negation of the first statement is 'the number x is a rational number'.
This is same as the second statement because if a number is not an irrational number then the number is a rational number.
Hence, the given statements are negations of each other.
(ii) The negation of the first statement is 'the number x is not a rational number. This means that the number x is an irrational number which is same as the second statement.
Hence, the given statements are negations of each other.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts