Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
The selection process for the MA course at the School of Government, MIT WPU involves a written entrance test conducted by MIT-WPU followed by a Personal Interaction (PI). Based on performance in both rounds, a merit list is released. Shortlisted candidates are then called for document verification and payment of fees to confirm their admission.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i)
The most electronegative element in the periodic table is fluorine having an atomic number 9 . The electronic configuration of fluorine is 1s2 2s2 2p5. Hence, the electronic configuration of the outermost shell of fluorine is 2s2 2p5 .
New Question
10 months agoNew Question
10 months agoBeginner-Level 5
Pronoun is one of the important aspects of English grammar. It is one of the eight traditional parts of speech. The significance of pronoun lies in the fact that it provides an alternate or substitute for nouns. This helps in breaking the monotony and avoiding repetition, hence bringing variation in reading or conversation.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
The electronic configurations and the bond orders of the given species are given below:
Bond order of O2 - : σ1s2 σ∗1s2 σ2s2 σ∗2s2 σ2p2z (π2p2x = π2p2y) (π∗2p1x= π∗2p1y)
Bond order = ( Nb- Na)
BO= (10-7)= = 1.5
Bond order of O2 + : σ1s2 σ∗1s2 σ2s2 σ∗2s2 σ2p2z (π2p2x = π2p2y) (π∗2p1x= π∗2p0y)
Bond order = (10-5)= =2.5
Bond order of O2 : σ1s2 σ∗1s2 σ2s2 σ∗2s2 σ2p2z (π2
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iv)
(a) A molecule which has a zero bond order never exists and a molecule having a non-zero bond order either exists or is expected to exist.
(b) If the value of bond order is high then the bond strength will also be higher. Electrons present in the bonding molecular orbital are known as the bonding molecular orbital ( Nb ) and the electrons present in the anti-bonding molecular orbital are known as anti-bonding electrons ( Na ). The half of the difference of bonding and anti-bonding electrons is known as the bond order.
The bond order of Be2 : Be2=s 1s2, s
New Question
10 months agoContributor-Level 10
Document verification is a crucial step in the admission process of Gujarat Technological University. The list of required documents that need to verified are mentioned below:
- Marksheet of Class 10
- Marksheet of Class 12
- Caste Cetificate
- Valid NCL certificate (if applicable
- Certificate of Ex-Serviceman (if applicable)
- Certificate of Disability (if applicable)
- EWS Certificate (if applicable)
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iv)
In the molecules of boron ( B2 ), carbon ( C2 ) and nitrogen ( N2 ) the energy of s2pz molecular orbital is greater than the energy of P2px and P2py molecular orbital.
So, the correct order of energies of molecular orbital of N2 is
(π2py ) > (σ2pz ) < (π* 2px ) ≈ (π*2py )
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
The element B represents the electronic configuration of phosphorus atom and element C represents the electronic configuration of chlorine atom. Both phosphorus and chlorine are non-metals hence the bonding forming between them will be a covalent bond.
New Question
10 months agoContributor-Level 10
Studying for a BCA Degree in India is a good choice from Creative Techno College because it allows students from all streams to pursue a career in computer science. Automation is now prevalent in every business, involving computer professionals in every aspect of businesses, including operations, backend, etc. - thus, allowing BCA course graduates to pursue jobs in various sectors. Also, BCA course fees are comparatively much less than BTech course fees, making it pocket-friendly for students from different backgrounds to study the course.
- Financial institutions like Banks, NBFC and Accounting f
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iv)
The electronic configuration of B given as 1S2 2S2 2P6 3S2 3P3 which represents phosphorous atom. The electronic configuration of C is given as 1S2 2S2 2P6 3S2 3P5 which represents chlorine molecule.
So, the molecular formula representing B and C will be phosphorous trichloride ( PCl3 ) which corresponds to BC3 .
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
The electronic configuration of C is given 1S2 2S2 2P6 3S2 3P5 which the electronic configuration of chlorine atom ( Cl ). It is unstable due to one unpaired electron in the p-subshell. Hence, the C2 will be the stable form of C as it will form a dichloride molecule ( Cl2 ).
New Question
10 months agoContributor-Level 10
Yes, Ganpat University BSc syllabus includes various elective subjects for students to choose from. Some of the elective subjects included in the curriculum of the BSc in Biotechnology are listed below:
- Environmental Science
- Computer Fundamentals and Applications
- Basic Statistics
- Disaster Management
- Basic Probability Theory
New Question
10 months agoContributor-Level 10
The seat intake of Veer Madho Singh Bhandari Uttarakhand Technical University for the BPharm programmes depends on multiple aspects. The university has provisioned 1,402 seats for its BPharm programmes. The key factors upon which VMSB Uttarakhand Technical University BPharm seat intake depends, are:
- Availability of Seats
- Total number of applications
- Total number of shortlisted students
- Academic credentials
- Scores in entrance exam
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i)
The electronic configuration of A 1S2 2S2 2P6 is having no unpaired electrons in the outer most shells. So, an atom having no unpaired electron in its valence shell is said to be stable.
New Question
10 months agoContributor-Level 10
The total tuition fee amount varies at both the BTech offering institutions. Sant Baba Bhag Singh University BTech tuition fees is INR 3.19 lakh. Additionally, STC BTech tuition fees is INR 2.4 lakh. Hence, in terms of fees, Sukhjinder Group of Institutes offers BTech at more affordable price. However, for a fair college comparison, students can consider other parameters as well. It must also be noted that the fee amount mentioned here is as per the official website/ sanctioning body. It is still subject to changes and hence, is indicative.
New Question
10 months agoContributor-Level 10
The key highlights of IIT Guwahati placements 2024 and 2025 for BDes are presented below:
Particulars | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|
| the highest package | INR 95 LPA | INR 52 LPA |
| Average Package | INR 36.17 LPA | INR 28.42 LPA |
| Median Package | INR 30.75 LPA | INR 23.28 LPA |
| the lowest Package | INR 8.16 LPA | INR 10 LPA |
| Students Placed | 22 | 26 |
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
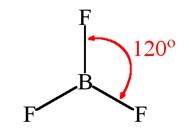
For a molecule having a hybridization of sp2, its geometry in plane is trigonal planar. For a molecule having a trigonal planar geometry, its bond angle will be 120°
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
