Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iv)
The electrons involved in the formation of any chemical bond are the valence shell electrons. In the given electronic configuration,1s2 2s2 2p6 3s2 3d2 4s2 the valence shell electrons are in d-orbital and s-orbital. So, the four electrons involved in the chemical bond formation will be 3d2 4s2.
New Question
10 months agoContributor-Level 10
Rajagiri College of Social Sciences (RCSS) offers BSc under two specialisations. The selection process for BSc admission at the college is primarily merit-based. Prospective candidates are shortlisted based on their merit in the qualifying exam. Bonus marks are awarded to NCC, NSS, & Ex-servicemen applicants, and widows & children of Jawans & Ex-servicemen. The next step in the selection process is for shortlisted candidates to appear for a Personal Interview (PI) conducted by the college. To be considered for the selection process, candidates are required to fulfil the course-specific eligibility criteria.
New Question
10 months agoContributor-Level 10
Institute of Hotel Management, Catering Technology & Applied Nutrition, Bhubaneswar BSc admission is based on candidates' performance in the NCHMCT JEE entrance exam, followed by the NCHMCT JEE counselling process. IHM Bhubaneswar NCHMCT JEE cutoff 2025 has been released for the first round, wherein the closing rank stood at 7808 for the students belonging to the General All India quota. Aspirants seeking admission can refer to the table below to view the three years of NCHMCT JEE first-round closing ranks for the general category students:
| Course | 2023 | 2024 | 2025 |
|---|---|---|---|
| B.Sc. in Hospitality and Hotel Administration | 6892 | 8821 | 7808 |
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
Hydrogen bond is the strongest in that molecules have a higher difference in electronegativity. Due to the small size of the oxygen atom, it has the highest electronegative character. So, H2O molecules will have the strongest hydrogen bonding
HCl, HI and H2Sdo not have hydrogen bonding between their atoms. So, H2O will have the strongest hydrogen bonding.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iii)
(i) The molecule XeF4 has a square planar geometry and the bond lengths of all the fluoride atoms attached to xenon atoms are equal.
(ii) The molecule BF4 - has a tetrahedral geometry and the bond lengths of all the fluoride bonds attached to boron are equal.
(iii) In the molecule C2H4 all the bonds are not equal. The bond length of the double bond between the carbon atoms is 134 pm and the bond length of CH- is 110 pm.
(iv) The molecule SiF4 has a tetrahedral geometry and the bond lengths of all the fluoride atoms attached to silicon atoms are equal.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iii)
The molecular orbital diagram of O22-, π∗2Px and π∗2Py molecular orbitals are completely filled, which are partially filled in O2. These are no unpaired electrons. So, O22-does not contain unpaired electrons.
New Question
10 months agoContributor-Level 10
The fee across specialisations for the NIFT Raebareli Certificate courses is INR 47,500.
Candidates can fill out the application form present on the official website of NIFT Raebareli to get a seat in the programme. The course duration is 3 months for various specialisations offered in the Certification programme. Candidates can get admission on the basis of their academic performance uptil Class 12.
New Question
10 months agoContributor-Level 10
If you want to do B.Tech in CSE in Bangalore, top colleges include RV College of Engineering, MS Ramaiah Institute of Technology, BMS College of Engineering, PES University (Ring Road), and UVCE. To get in, you'll need a good KCET or COMEDK rank—ideally under 4,000 for the top ones, and even better for CSE.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iii)
A double bond has one s bond and one π bond. In the above given structure 5π bonds are present. The s bonds are the additions of all the single bonds in the structure. There are 19s bonds in the above given structure.
New Question
10 months ago
Contributor-Level 10
Some of the most common Interjection words include the following:
· Wow
· Yay
· Yippie
· Ouch
· Oops
· Oh God
· Oh no
· Yikes
· Hush
· Umm
· Eh
· Argh
· Ugh
The above list is i
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i)
(i) The molecule of BH4 - has four bond pairs and zero lone pair of electrons, so it will be a tetrahedral molecule.
(ii) NH2 - has two bond pairs and two lone pairs of electrons on the nitrogen atom. So, it will have a bent geometry.
(iii) CO3 2- has three bond pairs and no lone pairs of electrons on carbon atoms. So, it will have a trigonal planar geometry.
(iv) H3O+ has three bond pairs and one lone pair of electrons on oxygen atoms. So, it has a pyramidal geometry.
New Question
10 months agoContributor-Level 10
Yes, you can change TOEFL test center, but only if another center has available seats. You may need to pay a reschedule fee. Make sure to check the new center and book early so you don’t miss your test date.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
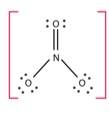
Ans: Option (iv)
The number of bond pairs on the nitrogen atom of NO3- are 4 and the structure of - NO3 does not have any lone pairs of electrons. The structure of NO3- molecule is:
New Question
10 months agoContributor-Level 10
IISER Pune accepts various national-based/ in-house entrance test scores for admissions to MSc courses. Moreover, which entrance exam is accepted by the institution depends on the specialisation candidates have selected. Check out the name of the specialisation and its accepted entrance exam in the table below:
| Names of MSc Specialisation | Accepted Entrance Exam |
|---|---|
| MSc in Chemistry | IIT JAM (Chemistry) |
| MSc in Earth and Climate Science (Geology) | IIT JAM (Geology) |
| MSc in Mathematics | The selection to the programme will be based on a two-stage written test conducted by the institute. |
| MSc in Physics (Quantum Technology) | IIT JAM 2025 exam with one of the following subject codes: MS, MA, PH. GATE exam with subject codes PH, EE, EC, IN, MA, ST, CS, DA. |
| Integrated BS + MS Course | They are required to apply for the courses by applying for the IAT (IISER Aptitude Test). |
New Question
10 months agoContributor-Level 10
You need a valid ID (mostly a passport) and an ETS account. You must enter correct name and ID details while booking.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
In a polyatomic molecule or an ion, the formal charge of an atom is defined as the difference between the valence electrons present in that atom and the number of electrons assigned to that atom in the Lewis structure.
Formal charge = No. of valence electrons - (No. of lone pair
Formal Charge = 6 -
Formal Charge = -1
New Question
10 months agoContributor-Level 10
It is better to book TOEFL exam at least 1 or 2 months early, because seats get full very fast, specially in popular TOEFL centers or busy exam season. Early booking give you enough time for TOEFL preparation and avoid last minute problem.
New Question
10 months agoBeginner-Level 5
Punctuations which are commonly put around Interjections include comma (, ), semicolon (;), and exclamation marks (!). Some of the other punctuations are hyphen (-) or full stop (.).
New Question
10 months agoContributor-Level 10
Yes, candidates can obtain a seat in the BPharm at VMSB UTU by meeting certain eligibility criteria. The selection of candidates is based on the scores obtained in the entrance exam and performance in academics. The university accepted UKSEE for BPharm admission. The university offers approximately 1,400 seats for its BPharm programmes. However, it must be noted that the mentioned seat intake is as per the official website. Hence, it may vary.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts