Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoBeginner-Level 5
Both Him and His are pronouns for male people or living beings. His is a Possessive Pronoun. It indicates ownership or belonging. On the other hand, 'Him' is an objective case of pronoun. Him is at the receiving end and answers who is being acted upon. Example of His: This is his book. Example of Him: The book belongs to him. Example of usage of Him and His in the same sentence: The doctor called him to share his medical test reports.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iii) and (iv)
The formula of bond order is:
Bond order = ( Nb- Na)
Number of bonding and antibonding electrons can be found from the molecular electronic configuration of the species.
(i) Total number of electrons in N2 is 14. So, its molecular electronic configuration will be
σ1s2 σ∗1s2 σ2s2 σ∗2s2, π2p2x , π2p2y, s2pz2
The bond order will be:
( 10-4)= 3
(ii) The total number of electrons in N2 - is 15. So, its molecular electronic configuration will be:
σ1s2 σ∗1s2 σ2s2 σ∗2s2, &
New Question
10 months agoContributor-Level 9
Yes, Sant Baba Bhag Singh University offers scholarship to students enrolled in the four-year long BTech courses. It is provided based on merit in the last qualifying examination, i.e., Class 12. The tuition fee concession is hence, based on the aggregate in the last qualifying exam. Mentioned below is tuition fee concession details along with the required minimum aggregate:
- Above 95%: 100% concession
- 90.1% TO 95%: 50% concession
- 80.1% TO 90%: 35% concession
- 70.1% TO 80%: 25% concession
New Question
10 months agoBeginner-Level 5
Both Reflexive Pronoun and Intensive Pronoun end with 'Self' or 'Selves' such as 'Myself', 'Himself', 'Herself', etc. Reflexive pronouns are the object or indirect object of the main verb. On the other hand, the Intensive Pronouns emphasise a noun or a pronoun. Such sentences do not add value to the sentence.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i) and (iv)
(i) N2 will have 14 electrons in its structure. So, its electronic configuration will be:
σ1s2 σ∗1s2 σ2s2 σ∗2s2, π2p2x , π2p2y, s2pz2
According to the molecular electronic configuration of N2 it does not contain any unpaired electrons, it will be diamagnetic in nature.
(ii) N22– will have 16 electrons in its structure. So, its molecular electronic configuration will be:
σ1s2 σ∗1s2 σ2s2 σ∗2s2, π2p2x , π2p2y, s2pz2, , π*2p1y
According to the molecular elec
New Question
10 months agoContributor-Level 10
To get into Creative Techno College for the BSc course, candidates must meet the eligibility criteria set by the institute. Students must pass Class 12 in Science stream. Creative Techno College offers full-time BSc courses of three years' duration. Students can get into this course based on merit.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iii) and (iv)
(i) The hybridization of the central carbon atom in CO3 2- is 2 sp as the carbon is bonded with three oxygen atoms. So, the given statement is incorrect.
(ii) The resonance structure of CO32- has one C= O double bonds and two C- O single bonds. So, the statement is incorrect.
(iii) The net charge on three oxygen atoms is -2, hence the average formal charge on each oxygen atom is 0.67 units. So, the given statement is correct.
(iv) As it can be seen from the resonating structures that the structures of the bonds are not fixed. Hence, all the C-
New Question
10 months agoContributor-Level 10
This is a Mathematical Reasoning Solutions Type Questions as classified in NCERT Exemplar
(i) Chennai is not the capital of Tamil Nadu.
(ii) √2 is a complex number.
(iii) All triangles are equilateral triangles.
(iv) The number 2 is not greater than 7.
(v) Every natural number is not an integer.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (iii) and (iv)
(i) The number of valence electrons in CO2 is 14. The oxygen atom attached to the carbon atom forms a double bond. Therefore, the hybridization of the structure is sp2. Thus, carbon dioxide is linear in shape.
(ii) The number of valence electrons in CCl4 is 32. The structure of CCl4 is arranged in a tetrahedral manner. The chlorine atoms are arranged such that the hybridization is sp3.
(iii) The number of valence electrons in O3 is 24. The hybridization of the molecule is sp2 and due to the presence of lone pair of electrons on the central oxy
New Question
10 months agoContributor-Level 10
The final step of the MA admission process at the School of Government, MIT WPU includes document verification and fee payment. After being shortlisted through the MIT WPU CET and Personal Interaction, students must verify their academic records and submit the initial tuition fee amount to confirm their admission into the programme.
New Question
10 months agoContributor-Level 10
This is a Mathematical Reasoning Solutions Type Questions as classified in NCERT Exemplar
The three examples of sentences which are not statements are:
(1) He is an engineer.
Here, in this sentence, it is not evident as to whom 'he' is referred to. Hence, it is not a statement.
(2) Painting is easy for some people, painting can be easy and for some others, it can be difficult. Hence, it is not a statement.
(3) Where is my hat?
This sentence is a question which is not considered as a statement in mathematical language. Hence, it is not a statement.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i) and (ii)
The species having the same number of electrons are known as isoelectronic species.
The number of electrons in CO species is 14; 8 electrons from oxygen atom and 6 electrons from carbon atom.
The number of electrons in NO+ is 14; 8 electrons from oxygen atom, 7 electrons from nitrogen atom and 1 electron will be subtracted due to the positive charge on the species.
The number of electrons in N2 is 14 ; 7 from each nitrogen atom.
The number of electrons in SnCl2 is 84 ;50 from the tin atom and 17 from each chlorine atom.
The number of electrons in NO2
New Question
10 months agoContributor-Level 10
For admission to the BSc course at Creative Techno College, students need to fulfil the minimum eligibility criteria in the first place. The selection criteria for this course is merit must pass Class 12. The duration of the course is three years.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i) and (iv)
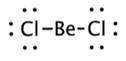
The structures of NO2 and NCO+ has a lone pair of electron on nitrogen atom thus it will form a bent shape rather than a linear shape.
New Question
10 months agoContributor-Level 10
For admission to the MA programme at the School of Government, MIT WPU, candidates are required to submit key documents such as their graduation mark sheets, ID proof, recent passport-size photographs, category certificate (if applicable), and other academic records. These documents are verified during the final admission step after shortlisting.
New Question
10 months agoContributor-Level 10
Candidates willing to pursue a BSc at Rajagiri College of Social Sciences must undergo a multi-step process. They must begin with the online application process conducted on the college's official website. The college evaluates the received applications and shortlists eligible candidates based on their merit in the qualifying exam. The next step is to appear for the personal interview (PI) as per the schedule shared by the college. Shortlisted candidates must submit the applicable fees to confirm their admission.
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (i) and option (ii)
Bond order depends on the electronic configuration of the molecular species, and the electronic configuration depends on the number of electrons a particular atom is contributing in its molecular structure.
The CN- species has a total of 14 electrons; 6 electrons from carbon atom, 7 electrons from nitrogen atom and 1 electron for the negative charge.
The NO+ species has a total of 14 electrons; 8 from oxygen atoms, 7 from nitrogen atom and 1 electron will be subtracted due to the positive charge on the species.
The O2 - species has a total
New Question
10 months agoBeginner-Level 5
There are over 10 types of pronouns. The major pronoun types are as follows:
- Personal Pronoun
- Subject Pronoun
- Object Pronoun
- Relative Pronoun
- Reflexive Pronoun
- Intensive Pronoun
- Interrogative Pronoun
- Indefinite Pronoun
- Demonstrative Pronoun
- Possessive Pronoun.
New Question
10 months agoContributor-Level 10
This is a Mathematical Reasoning Solutions Type Questions as classified in NCERT Exemplar
(i) This sentence is false because there are less than 35 days in a month. Hence it is a statement.
(ii) This sentence differs to individual, as some may find it difficult and some may find it easy. This means that it is not always true. Hence it is not a statement.
(iii) This sentence is true as the sum of 5 and 7 is 12 which is greater than 10. Hence it is a statement.
(iv) This sentence can be correct and incorrect sometimes as the square of 2 is even but the square of 3 is an odd. Hence it is not a statement.
(v) This sentence is no
New Question
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (ii)
Aluminium has [Ne]3s2 3p1 electronic configuration. Phosphorus has [Ne]3s2 3p3. The electronic configuration of silicon is [Ne]3s2 3p2 and the electronic configuration of arsenic is [Ar]3d104s24p3.
Ionisation energy increases as the atomic number in a period increases and decreases on moving down the group. It is also known that the ionisation enthalpy of group 15 elements is more than group 16 elements as group 15 has half-filled p-subshells which gives extra stability.Thus, the electronic configuration having the highest ionization entha
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
