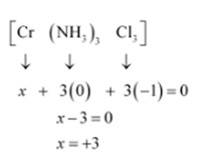Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
4.8 Given-
Initial temperature, T1 = 298 K
Final temperature, T2 = 298 K + 10 K = 308 K
Knowing that the rate constant of a chemical reaction normally increases with increase in temperature, we assume that,
Initial value of rate constant, k1 = k
Final value of rate constant, k2 = 2k
Using Arrhenius equation,

New Question
11 months agoContributor-Level 10
To enroll in the certificate course at Dfly International Aviation Institute, candidates must meet the eligibility criteria set by the institute. The institute offers certificate courses in various specialisations. Students are selected based on merit. In addition to the selection criteria, students must pass Class 12 (common eligibility).
New Question
11 months agoContributor-Level 10
(i) Starting with cation, the complex ion contains six ammonia molecules with cobalt in + 3 oxidation The name of compound: [Co (NH3)6]Cl3 is hexaamminecobalt (III) chloride.
(ii) The complex ion is cation, so there are 2 ammonia molecules, one chloride ion and methyammine molecule qith platinum in + 2 state. Going in alphabetical order, the name of compound: [Pt (NH3)2Cl (NH2CH3)]Cl is diamminechloridomethylammineplatinum (II)
(iii) It is a complex cation with six water molecules and titanium atom in + 3 state. The name of the compound: [Ti (H2O)6]3+ is hexaaquatitanium (III) ion
(iv) The complex ion is cation with
New Question
11 months agoContributor-Level 10
4.7 The rate constant of a chemical reaction normally increases with increase in temperature. It is observed that for a chemical reaction with rise in temperature by 10°C, the rate constant is nearly doubled and this temperature dependence of the rate of a chemical reaction can be accurately explained by Arrhenius equation,
k = A
Thus, the rate constant of a chemical reaction normally increases with increase in temperature.
New Question
11 months ago
Contributor-Level 10
BPharma is one of the most popular courses of Guru Nanak College Dehradun. When it comes to the placements, the institute offers decent placements annually. According to the official stats, 17 BPharma students were placed during the 2024 placement drive at Guru Nanak College Dehradun. All the important details related to Guru Nanak College Dehradun placements are mentioned below:
| Particulars | Placement Statistics (2024) |
|---|---|
| the highest package | INR 1.01 crore per annum |
| Companies visited | 100+ |
| Students placed | 17 (BPharma) |
| Top recruiters | NECLIFE, Windlas Biotech Limited, Swarnimaksh Life Sciences |
NOTE: The above details are fetched from the official website, however, the year of placement was not mentioned. Hence, considering it for 2024.
New Question
11 months agoContributor-Level 10
In the first semester of the MBA at Nagaland University - Kohima Campus, candidates are introduced to foundational subjects that build essential managerial skills. The curriculum includes:
Management Principles and Organisational Behaviour
Business Environment and Indian Economy
Managerial Economics
Accounting for Managers
Statistical and Operations Research Techniques
Managerial Skill Development
Computer Applications and MIS
New Question
11 months agoContributor-Level 10
Institute conducts NIFTEE for NIFT Mumbai BDes exam. This exam is divided into General Ability Test (GAT) and Creative Ability Test (CAT). After qualifying, appear for a subsequent Situation Test. Based on NIFTEE scores, students are ranked and allocated a seat.
New Question
11 months agoContributor-Level 10
(i) Tetrahydroxidozincate (II) = [Zn (OH)4]-2
Tetrahydroxi means 4 hydroxide ions with zinc in + 2 oxidation state. Hydroxide ions have a negative charge of -1, so balancing the overall charge of the coordination compound to be zero we get the formula as : [Zn (OH)4]-2
(ii) Potassium tetrachloridopalladate (II) = K3 [PdCl4].
Tetrachlorido means 3 Chloride ions each having a negative charge. Platinum is in + 2 state. Balance overall charge as 0, no. Of potassium ions are 3. Formula: K3 [PdCl4].
(iii) Diamminedichloridoplatinum (II) = [Pt (NH3)Cl2]2+
Diammine means 2 ammonia molecules, dichlorido means 2 chloride ions, platinum
New Question
11 months agoNew Question
11 months agoContributor-Level 10
4.6 Given-
t1/2 = 60 mins
Using the formula for half life, t1/2 = 0.693/k, we get,
k= 0.693 / t1/2
k= 0.693 / 60
∴k = 1.155 × 10-2 min-1
The rate constant of the reaction, k is 1.155 × 10-2 min-1
New Question
11 months agoContributor-Level 10
The students who want to take admission in the M.Arch college at GIATP need to first apply online for the admission. After filling up the form, the college may give the dates for Interview process. The students are required to appear for the interview. Selected students are given admission.
New Question
11 months agoContributor-Level 10
(i) Let Oxidation of Co be x and charge on the complex is given as + 2
H2O has Oxidation Number: 0
CN has Oxidation Number: -1
en has Oxidation Number :0
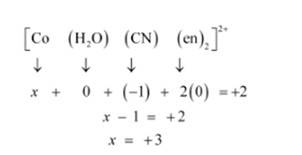
(ii) Let Oxidation number of Co be x and charge on the complex is given as + 1
Br has Oxidation number: 1
en has oxidation number : 0
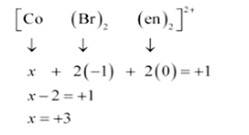
(iii) Let Oxidation number of Pt be x and charge on the complex is given as -2
Cl has oxidation number : -1
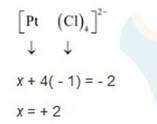
(iv) This complex can also be seen as [Fe (CN)6]3-
Let Oxidation number of Fe be x and charge given on the complex is given as -3
CN has oxidation number : -1
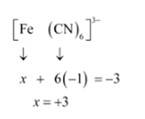
(v) Let Oxidation number of Cr be x and charge given on the complex is given
New Question
11 months agoContributor-Level 10
Candidates wishing to get admission to the courses at St. Xavier's College Goa are required to fill out the application form and appear for the next rounds of admission to secure their seat. There is no such special provision for candidates to get direct admission. Candidates are required to get admission by following the merit based admission process.
New Question
11 months agoContributor-Level 10
4.5 Given-
Rate constant, k = 1.15 * 10-3 s-1
innitial quantity R0 = 5g
Final quantity, R = 3 g
According to the formula of first order reaction
K = 2.303/ t log R0 / R
1.15 X 10-3 = 2.303 /t log 5/3
t = 2.303 / 1.15 X 10-3 log 5/3
t = 443.8 s
t = 4.438 * 102 s
The time taken for 5g of the reactant to reduce to 3 g is 4.438 * 102 s
New Question
11 months agoContributor-Level 10
4.4 Let the reaction be X→Y
As this reaction follows second order kinetics, the rate of the reaction will be,
Rate = k (X)2 → Equation 1
Since the concentration of X is increased three times, Equation 1 will become,
Rate = k (3X)2
= k × 9 (X)2
∴ The rate of formation of Y will become 9 times.
Thus, the rate of formation of Y will become 9 times
New Question
11 months ago
Contributor-Level 10
University of Michigan has received top rankings for courses such as Engineering, Supply Chain Management, Sociology, Nursing, Information Technology, Business and Management, etc. Some of the course rankings of the university are given in the table below:
| Course Name | Shiksha Popularity Ranking | US News Ranking |
|---|---|---|
| MS | #8 | #5 |
| MBA/PGDM | #7 | #6 |
| BE/BTech | #5 | #5 |
| Information Technology and Systems | #21 | #13 |
| Mechanical Engineering | #5 | #6 |
New Question
11 months agoContributor-Level 10
Pursuing a BDes from NIFT Mumbai helps students develop a foundation in the field of Design. However, in the time of increasing competition in the job market, having only theoretical knowledge might not be enough to get the dream job. Students who wish to get a dream job must gain vital skills that will set them apart from the rest. Mentioned below are some of the skills demanded in today's corporate world:
- GIS Software Proficiency
- Environmental Awareness
- Data Interpretation
- CAD (Computer Aided Design) Proficiency
- Spatial Analysis
- Problem-Solving
- Design Thinking
New Question
11 months agoContributor-Level 10
Ligands are the neutral or negatively charged entities surrounding the central metal atom of the coordination complex which possesses at least one unshared pair of electrons.
Based on the number of donor sites of these ligands, Ligands are classified as:
Unidentate ligands: These Ligands which have only one donor site are called unidentate ligands.
Example: F-, Cl – etc.
Didentate ligands: These Ligands which have only two donor site are called didentate ligands.
Example: Ethane-1,2-diamine, Oxalate ion etc.
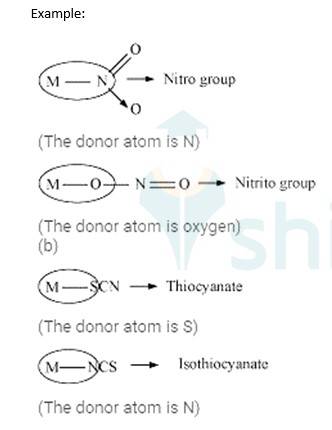
Ambidentate ligands: These ligands which can attach them with the central metal atom by two different atoms are called as ambidentate
New Question
11 months agoContributor-Level 10
14.33
The different types of RNA found in the cell are listed below:-
(i) Messenger RNA (m-RNA)
It carries the genetic message code from the DNA to ribosomes. It is produced by the DNA; m-RNA is also single stranded and constitutes about 15% of total RNA.
(ii) Ribosomal RNA (r-RNA)
It is found in the ribosomes and it is usually associated with protein to form the ribosomes. It is synthesised in the nucleus by DNA. It is single stranded, comprising about 80% of total RNA. It is metabolically stable.
(iii) Transfer RNA (t-RNA)
It is synthesised in nucleus by DNA. It is also called soluble RNA. It is single stranded. There are 20
New Question
11 months agoContributor-Level 10
(1) Coordination Entity: Coordination entity is a charged entity having positive or negative charge in which the central atom is surrounded by molecules which may be neutral/negatively charged called Ligands
Examples:
i. Cationic Complexes: [Cu (H2O)6]2+, [Al (H2O)6]3+
ii. Anionic Complexes: [CuCl4]2-, [Al (H2O)2 (OH)4]-
iii. Neutral Complexes: [Co (NH3)4 Cl2], [Ni (CO)4]
(2) Ligands: Ligands are the neutral or negatively charged entities surrounding the central metal atom of the coordination complex which possesses at least one unshared pair of electrons
Example: F-, Cl-, Br-, I-, H20, and NH3
(3) Coordination Number: Coordina
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts