Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoBeginner-Level 5
The GAT (PGP) syllabus is important to complete to pass the exam. Pharmaceutical Analysis & Quality Assurance section includes Fundamentals of Pharmaceutical Analysis, Acid-Base Titrations, Oxidation-Reduction Titrations, Complexometric Titrations, Non-Aqueous Titrations, Potentiometry, Conductometry, Polarography, and Amperometry.
New Question
11 months agoContributor-Level 10
Yes, BA students with family income of INR 6 LPA are eligible for the Merit-cum-Need Scholarships offered at Navrachana University. However, they must have scored a minimum of 70% in Class 12. Further, they must present documents proving that their family's combined income does not exceed INR 8 LPA. The amount of scholarship offered against various income slabs are presented in the table below:
| Scholarship | Family Income Slab |
|---|---|
| 100% Tuition Fee Waiver | Up to INR 2 lakh |
| 80% Tuition Fee Waiver | Between INR 2 Lacs to INR 3 lakh |
| 60% Tuition Fee Waiver | Between INR 3 Lacs to 4 lakh |
| 40% Tuition Fee Waiver | Between INR 4 Lacs to 6 lakh |
| 20% Tuition Fee Waiver | Between INR 6 Lacs to 8 lakh |
*Scholarship will be renewed every year provided the student maintains CGPA of 7 out of 10.
New Question
11 months agoContributor-Level 10
The reaction is given below:
FeSO4 + (NH4)2SO4 + 6H2O → FeSO4 (NH4)2SO4.6H2O (Mohr Salt)
FeSO4, when reacted with (NH4)SO4, does not form any complex whereas they form a double salt, FeSO4. (NH4)2SO4.6H2O - (Mohr salt) which dissociates into ions in the solution. So, it gives the test of Fe2+ ions.
CuSO4 + 4NH3 + 5H2O→ [Cu (NH3)4SO4].5H2O
CuSO4 solution when mixed with aqueous ammonia in 1: 4 molar ratio forms a complex with formula [Cu (NH3)]SO4 in which the complex ion, [Cu (NH3)4]2+ does not dissociate to give Cu2+ ions. Therefore, it does not give the tests of the Cu2+ ion.
New Question
11 months agoContributor-Level 10
14.32
The structural difference between DNA and RNA are as follows:-

The functional difference between DNA and RNA are as follows:-

New Question
11 months agoContributor-Level 10
Bonding in coordination compounds in terms of Werner's postulates is explained as:
(a) Metals can show two types of valencies which are Primary valency and Secondary
1. Primary Valency: Primary Valency shows Oxidation Primary valencies are ionizable.
2. Secondary Valency: Secondary Valency shows coordination These are non-ionizable.
(b) Both Primary and secondary valency of the metal are to be satisfied which is done by negative ions in case of primary valency and negative or neutral species in case of secondary
(c) Metals have a fixed number of secondary valencies/ Coordination number around the central atom, these secondary
New Question
11 months agoContributor-Level 10
M.Arch at Ganga Institute of Architecture and Town Planning is offered to the interested students. The students need to initially register themselves with the college through the application from submission. The application fee is also paid alongside. Th applied students are called for personal interview and portfolio check. The college shortlists some students and offer admission. It is important for students to fulfill the eligibility criteria.
New Question
11 months agoContributor-Level 10
Yes, at NIFT Mumbai, students can withdraw their admission if they wish to. Such students are also entitled to a refund of fees. However, the refund is subject to the fulfilment of certain conditions. Listed below are the guidelines to be followed as per the fee refund policy of the institute:
| Point of time when the withdrawal request is received | Refund Amount |
|---|---|
| Before commencement of the final round of seat allocation process | Full fees except the registration fee |
| After the declaration of date decided by NIFT | Only security deposit |
New Question
11 months agoContributor-Level 10
14.31
In the helical structure of DNA, the two strands are held together by hydrogen bonds between specific pairs of bases.
Cytosine from hydrogen bond with guanine, while adenine forms hydrogen bond with thymine. As a result, the two strands are complementary to each other.
DNA consists of two strands of nucleic acid chains coiled around each other in the form of a double helix. The base of one strand of DNA is paired with bases on other strand by means of hydrogen bonding.
This hydrogen bonding is very specific as the bases can only base pair in a complementary manner.
Adenine pairs with only thymine via 2 hydrogen bonds and guanine pairs
New Question
11 months agoBeginner-Level 4
Yes, there are many online and distance education options available for professionals who want to upskill without leaving their jobs.
In India, leading platforms include IGNOU, which offers a wide range of postgraduate, undergraduate, diploma, and certificate courses via distance learning. Swayam, the Government of India's free MOOC platform, provides high-quality courses from IITs, IIMs, and central universities. NIOS caters to foundational and vocational learning, while NMIMS Global Access focuses on professional MBAs, diplomas, and certifications.
Globally, platforms like Coursera, edX, Udemy, and LinkedIn Learning offer industry-reco
New Question
11 months agoContributor-Level 10
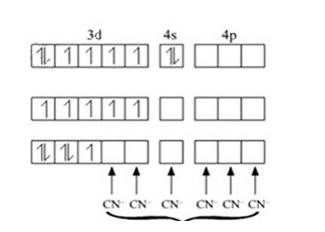
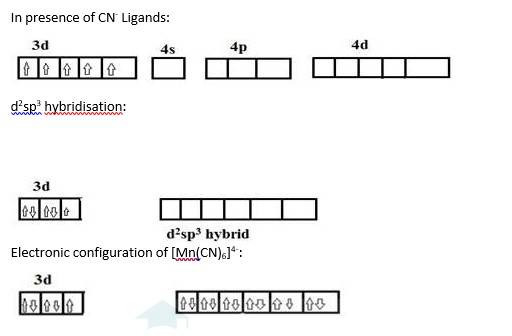
[Mn (H2O)6]+2 | [Mn (CN)6]4- |
Oxidation state of manganese: Overall charge balance: X + 6 (0) = 2 X = + 2 | Oxidation state of manganese: Overall charge balance: X + 6 (-1) = -4 X = + 2 |
Outer electronic configuration of Mn = d5 | Outer electronic configuration of Mn = d5 |
H2O is a weak field ligand so it does not cause pairing of the electron. Therefore Mn undergoes sp3d2 hybridization. Geometry is octahedral. Therefore the 5 unpaired electrons from the d orbital remain as it is. | CN is a strong field ligand so it causes pairing of the electron (5 electrons get paired to form 2 pairs and one unpaired electron). Therefore Mn undergoes d2sp3 hybridization. Geometry is octahedral. |
Mn in + 2 oxidation state:
New Question
11 months agoContributor-Level 10
In Pt [ (CN)4]2- ion:
Overall charge balance:
X + 4 (-1) = -2 X = + 2.
The oxidation state of Pt is + 2.
Since CN- is a strong field ligand, it causes pairing of the unpaired electrons.
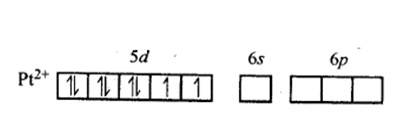
Therefore, now the 2 unpaired electrons from 5d orbital get paired and it undergoes dsp2 hybridisation. It forms square planar geometry. Since all the electrons are paired,
No. of unpaired electrons = 0.
New Question
11 months agoContributor-Level 10
St. Xavier's College Goa accepts candidates through their merit. Candidates seeking admission to BBA or other UG courses must qualify for admission by fiulling out the application form. The university does not accept scores for any other entrance exams. Also, direct admission is possible for candidates with exceptional riguor.
Candidates are required to get the admission confirmation by participating in the merit based admission process.
New Question
11 months agoContributor-Level 10
14.30
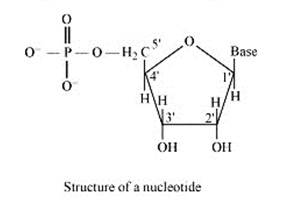
A nucleoside is formed when l-position of a pyrimidine (cytosine, thymine or uracil) or 9- position of a purine (guanine or adenine) base is attached to C-l of sugar (ribose or deoxyribose) by a linkage. Thus in general, nucleosides may be represented as: Sugar-Base.
Nucleoside = sugar + base
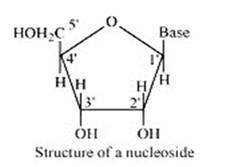
On the other hand, all the three basic components of nucleic acids (i.e., pentose sugar, phosphoric acid and base) are present in a nucleotide.These are obtained by esterification of C5' –OH group of the pentose sugar by phosphoric acid. Thus, in general, a nucleotide is represented as:-
Nucleotide= sugar + base + phosphoric acid
New Question
11 months agoContributor-Level 10
[Co (NH3)6]3+ | [Ni (NH3)6]2+ |
Oxidation state of cobalt: Overall charge balance: X + 6 (0) = 3 X = + 3 | Oxidation state of nickel: Overall charge balance: X + 6 (0) = 2 X = + 2 |
Outer electronic configuration of cobalt = d6 | Outer electronic configuration of nickel = d8 |
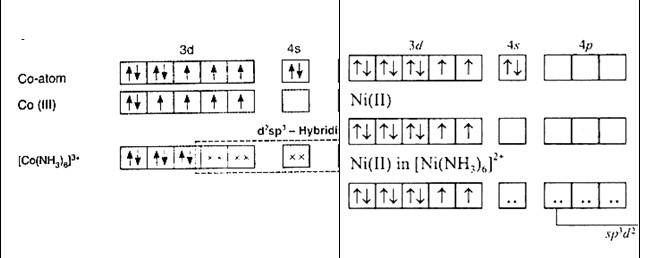
NH3 is a strong field ligand so it causes pairing of electron. Therefore cobalt undergoes d2sp3 hybridisation. As in the hybridisation d2 orbitals are used from the (n-1)d orbitals (inner orbitals as n = 4 being quantum number) . hence it is a inner orbital complex. | NH3 is a strong field ligand so it causes pairing of electron. Therefore, nickel undergoes sp3 d2 hybridisation. As in the hybridisation, d2 orbitals are used from the and orbitals (outer orbitals as n = 4 being quantum number). Hence, it is an outer orbital complex |
New Question
11 months agoContributor-Level 10
14.29
Nucleic acids are Biomolecules which are found in the nuclei of all living cells, inform of nucleoproteins or chromosomes (proteins containing nucleic acids as the prosthetic group). Nucleic acids are of two types: – deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Nucleic acids are also known as Polynucleotide as they are long- chain polymers of nucleotides.
The two important functions of nucleic acids are listed below:-
(i) DNA which is responsible for the transference of hereditary effects from one generation to another, which is due to their property of replication during cell division as a result of which two identica
New Question
11 months agoContributor-Level 10
In [Fe (H2O)6]3+
Electronic configuration of Fe is: [Ar]3d64s2
[Ar] = 1s22s22p63s23p6
Electronic configuration of Fe+3 = [Ar]3d5
Outer electronic configuration of Fe+3 = 3d5
Overall charge balance:
X + 6 (0) = 3
X = + 3
In [Fe (CN)6]3-
Overall charge balance:
X + 6 (-1) = -3
X = + 3
In both the compounds Fe is in + 3 oxidation state.
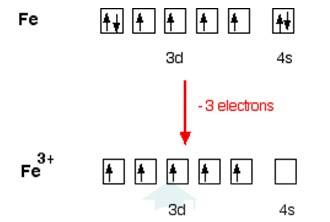
In case of [Fe (H2O)6]3+
H2O is weak field ligand so it does not pair the unpaired electron. Total no. of the unpaired electron, n =5, Spin only magnetic moment is given by:
μ = [n (n + 2)]1/2
μ = [5×7]1/2
μ = 5.916BM
In case of [Fe (CN)6]3-
CN- is a strong field ligand so it pairs up the electron.
Total no. o
New Question
11 months agoContributor-Level 10
The Master of Applied Business at Unitec Institute of Technology is a great option for international students who want to pursue their careers in business administration and management. This program is of 180 credits and includes specializations like Business Analytics, Digital Marketing, Human Resources, Leadership, or Supply Chain & Logistics. The program helps students on gaining hands-on experience through projects, research based work, and internships. Following are the requirements for admission into this program:
- Applicants would need bachelor's degree in a relevant field from a recognised university with at least a B- grade
New Question
11 months agoContributor-Level 10
14.28
The deficiency of Vitamin A leads to Xerophthalmia which is hardening of the cornea of the eye and night blindness as well. Vitamin-C is essential to us because its deficiency causes scurvy which is a phenomenon of bleeding gums; and pyorrhoea which is phenomenon of loosening-bleeding of teeth.
The sources of vitamin A are fish, cod liver oil, carrots, butter and milk.
The sources of vitamin C are citrus fruits, lemon, amla and green leafy vegetables.
New Question
11 months agoContributor-Level 10
In [Ni (Cl)4]2- ion, Cl- is a weak field ligand so it will not pair the unpaired electrons of Ni+2 ion. Electronic configuration of Ni is: [Ar]3d84s2 where [Ar] = 1s22s22p63s23p6
Electronic configuration of Ni+2 = [Ar]3d8 Outer electronic configuration of Ni+2 = 3d8 Overall charge balance:
X + 4 (-1) = -2 X = + 2.
Therefore it undergoes sp3 hybridization. So it will have tetrahedral geometry.

Since there are 2 unpaired electrons in the d orbital so it is a paramagnetic compound. In [Ni (Co)4]:
Overall charge is neutral and oxidation state of Ni can be calculated as:
X + 4 (0) = 0
x = 0
Ni is in zero oxidation state.
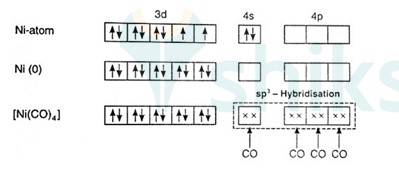
Co is a strong field ligand
New Question
11 months agoContributor-Level 10
The course admission at Dfly International Aviation Institute is not tough. The selection criteria for the course is based on merit. Aspirants looking for course admissions must meet the eligibility criteria set by the college. Candidates must pass Class 12. The course is offered in various specialisations such as Hospitality Management, Customer Service Training, etc.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts