Ask & Answer: India's Largest Education Community
All Questions
New Question
6 months agoNew Question
6 months agoNew Question
6 months agoNew Question
6 months agoContributor-Level 10
Primary structure of protein in unaffected by physical or chemical changes.
New Question
6 months agoContributor-Level 10
For precipitation of two moles of AgCl
Two Cl- will produce as a free anion
CoCl3.4NH3 -> complex will Cl (will not give 2Cl-)
complex will be H2 [PtCl6] will not any Cl-
will produce two Cl- ion.
precipitate formation
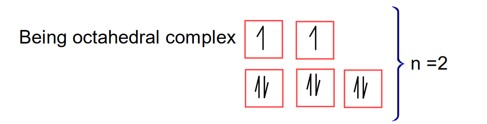
New Question
6 months agoContributor-Level 10
Most basic oxide V2O3
Here V has +3 O.S. Hence V+3 ->
two unpaired e- in d- subshell
New Question
6 months agoContributor-Level 10
Volume of H2 adsorbed =
Therefore volume of gas adsorbed per gram of the adsorbent =
New Question
6 months agoContributor-Level 9
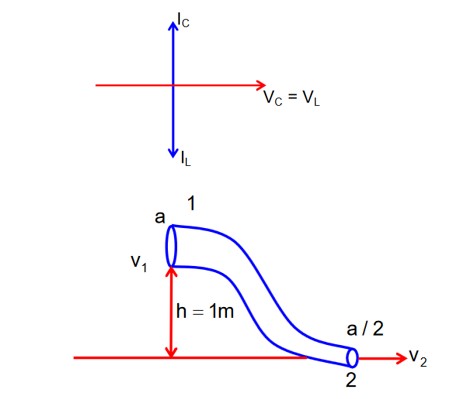
At Resonance
XL = XC
then lL = lC
Now phasor diagram
for L & C
So, Net current = zero
Therefore current through R circuit at resonance will be zero
New Question
6 months agoContributor-Level 10
Aniline show acid-base reaction with AlCl3
aniline is a Lewis base while AlCl3 acts as lewis acid.
New Question
6 months agoContributor-Level 10
Process is based upon simultaneous disintegration hence,
………….(i)
and ………….(ii)
from equation (i) and (ii)
Here; A0 = B0 and
Therefore
New Question
6 months agoNew Question
6 months agoContributor-Level 10
Here, total meq of acetic acid = 50 × 0.1 = 5
And total meq of NaOH = 25 × 0.1 = 2.5
After neutralization process
Meq of left acetic acid = 2.5
And meq of formed CH3COONa = 2.5
New Question
6 months agoContributor-Level 10
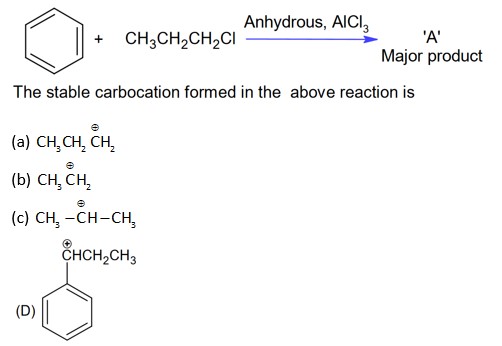
With AlCl3, alkyl halide will form cabocation which will show rearrangement.
New Question
6 months agoContributor-Level 10
Candidates looking for admission at Centre for Distance and Online Education, JIIT must complete at least a graduation degree for the MBA course. Similarly, for the BBA course, aspirants must complete Class 12.
New Question
6 months agoContributor-Level 9
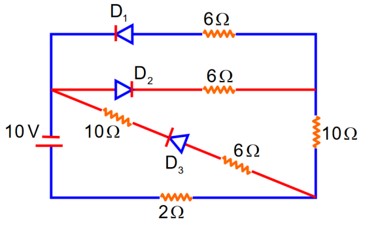
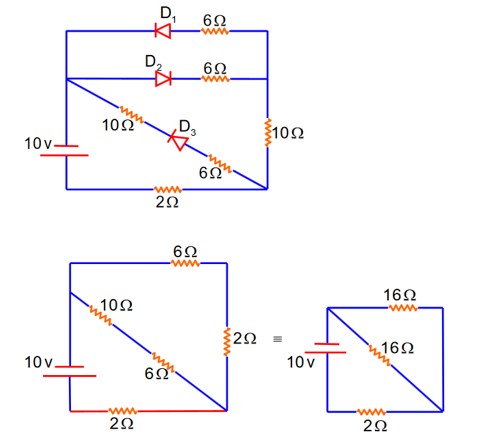
Forward biased offer zero Resistance
D2} Reversed biased offers Infinite Resistance
New Question
6 months agoContributor-Level 10
0.5 % KCl solution has molality (m) =
1 - a a a
And I =
1.976 = 1 + a
% = 97.6%
the nearest 98.
New Question
6 months agoContributor-Level 10
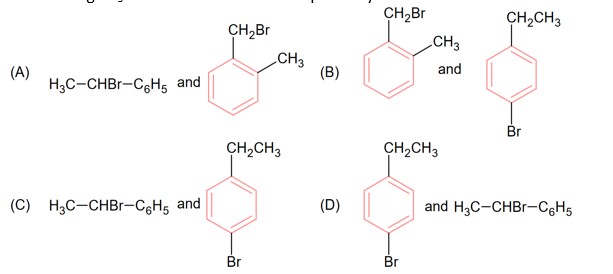
In this carbocation +M effect of -OCH3 group stabilizes the carbocation.
While in option (A) and (B), +M of -OCH3 will not work but in option (C), +M of -OCH3 works so due to more delocalization in option (D), it is more stable.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts