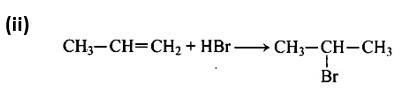Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
The placement percentage for the 2025 drive is not out yet. Earlier, the Institute of Management Nirma University placement rate recorded in 2024 was 100%. Check out the table below to know the placement rate for the past three years:
Particulars | Placement Statistics (2022) | Placement Statistics (2023) | Placement Statistics (2024) |
|---|---|---|---|
Placement rate | 100% | 100% | 100% |
New Question
10 months agoNew Question
10 months agoContributor-Level 10
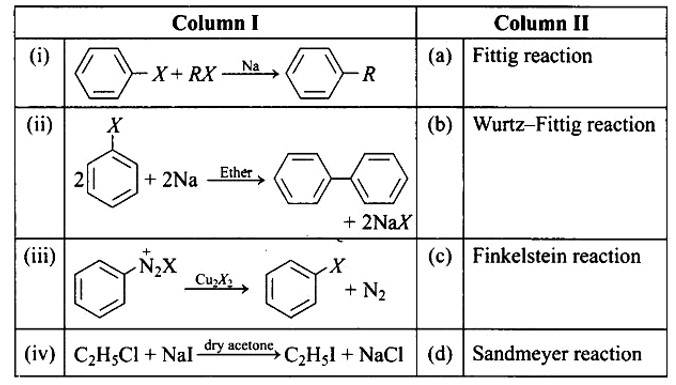
This is a matching answer type question as classified in NCERT Exemplar
In the presence of Na metal in dry ether, an alkyl halide and an aryl halide combine to create alkyl arene in the Wurtz-Fittig reaction.
The Fittig reaction, which involves a coupling reaction between two aryl halides in dry ether in the presence of Na metal, yielding diaryl compounds, is the second reaction.
The Sandmeyer reaction produces aryl halides from primary amines by converting them to diazonium salts and treating them with cuprous bromide.
The Finkelstein reaction, which produces alkyl iodides by reacting alkyl chlorides or bromides with NaI in dry acetone, i
New Question
10 months agoContributor-Level 10
Institute of Management Nirma University provides good placements to its graduating students. The highest package offered in 2025 was the same as in 2024, i.e INR 70 LPA. The average package in 2025 was INR 11.10 LPA and in 2024 it stood at INR 11.22 LPA. Check out the table below to know the key highlights of the Institute of Management Nirma University placements for MBA during 2023-2025:
Particulars | Placement Statistics (2023) | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|---|
the highest package | INR 30 LPA | INR 70 LPA | INR 70 LPA |
Average package | INR 13 LPA | INR 11.22 LPA | INR 11.10 LPA |
Median package | INR 11 LPA | INR 10.5 LPA | NA |
Placement Rate | 100% | 100% | NA |
Total companies | 87 | 85 | NA |
Entrepreneurial stand | 20 | 29 | NA |
Students placed overseas | NA | 3 | NA |
Average package of top 50 | INR 17.50 LPA | INR 16.29 LPA | INR 17.08 LPA |
Average package of top 100 | INR 15 LPA | INR 13.58 LPA | INR 14.74 LPA |
New Question
10 months agoContributor-Level 10
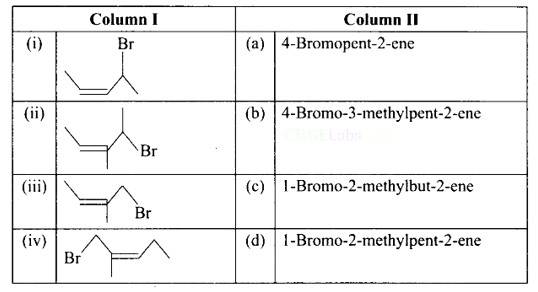
This is a matching answer type question as classified in NCERT Exemplar
(i) A: 4-bromopent-2-ene.
(ii) B: 4-bromo-3-methylpent-2-ene.
(iii) C: 1-bromo-2-methylbut-2-ene.
(iv) D: 1 -bromo-2-methylpent-2-ene.
Correct Answer: (i) → (a) (ii) → (c) (iii) → (b) (iv) → (d)
New Question
10 months agoContributor-Level 10
It is recommended that you should study at least 3-4 hours daily to score high marks in the UK 10th exam. The duration of learning varies for students according to their ability. Students should not study continuously for long hours. They should take enough breaks in between. They are advised to solve and practice previous year question papers and sample papers.
New Question
10 months agoContributor-Level 9
The placements 2025 concluded with the highest package INR 70 LPA. The average package for the 2024-25 academic year was INR 11.10 LPA. In addition to this, the average package for top 50 and top 100 during Institute of Management Nirma University placements 2025 was INR 17.08 LPA and INR 14.74 LPA, respectively.
New Question
10 months agoContributor-Level 10
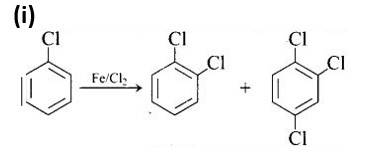
This is a matching answer type question as classified in NCERT Exemplar
The first reaction is an electrophilic substitution process in which Cl+ from the Cl2 and FeCl3 combination attacks and substitutes the benzene ring.
Following Markownikoff's rule, the second reaction is an electrophilic addition reaction in which HBr is added across the double bond.
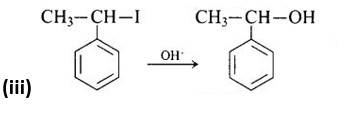
The third reaction is a nucleophilic substitution process that uses the SN1 mechanism to replace the -I group with -OH.
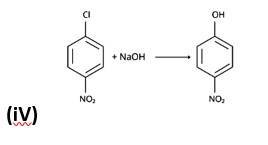
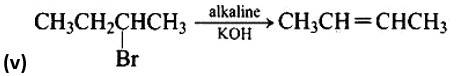
The nucleophilic aromatic substitution process, in which the -Cl group is replaced by -OH, is the fourth reaction.The fifth reaction is a dehydrohalogenation reacti
New Question
10 months agoContributor-Level 10
The total tuition fee for the BCom course at Maharaja Agrasen Institute of Management Studies is INR 4 lakh. On the other hand, the tuition fee for the BCom course at JIMS Greater Noida is INR 2.9 lakh. Hence, it can be explicitly seen that the tuition fee is more affordable at JIMS Greater Noida compared to Maharaja Agrasen Institute of Management Studies. However, there might be different factors that candidates can consider while conducting a comparison of these two institutions, such as ROI, seats, placement packages, etc.
The fee amount mentioned is sourced from the official website/sanctioning body and is subject to ch
New Question
10 months agoContributor-Level 10
UCLA graduates are highly skilled in Marketing, Strategic Planning, Strategy, Marketing Strategy, Business Development, Business Strategy, and others. University of California Los Angeles graduates work in top sector such as:
- Business Development
- Education
- Operations
- Engineering
- Research
- Media and Communication
- Arts and Design
- Community and Social Services
- Sales
- Finance
- Information Technology
- Marketing
- Human Resources
- Consulting
- Entrepreneurship
New Question
10 months agoContributor-Level 10
Ganga Institute of Technology and Management MBA programme is offered for a duration of two years. The course is offered across several specialisations. The admission criteria to the course and its specialisations is generally the same. Find below the list of MBA courses offered at the institute:
- Human Resource Management
- Marketing
- Finance
- International Business
- Information Technology
- Business Analytics
- Agri-Business
- Operation Management
New Question
10 months agoContributor-Level 10
This is a matching answer type question as classified in NCERT Exemplar
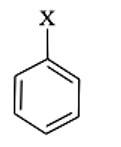
Because it includes a halogen atom bound to an sp3 hybridised carbon atom, which is subsequently linked to additional alkyl groups, the first molecule is an alkyl halide.
An allyl halide is a compound in which the halogen atom is linked to a carbon atom that is bound to an sp3 hybridised carbon atom that is attached to a carbon-carbon double bond.
The third molecule is an aryl halide, which is a chemical in which the halogen atom is sp2 hybridised with an aromatic ring carbon atom.
In a carbon-carbon double bond, the halogen atom is linked to a sp2 hybridised carbon at
New Question
10 months agoContributor-Level 10
The equation can be written as-
Ap+x + Bq- y ? xAp+ (aq) + yBq-
S moles of A and B dissolves to give x S moles of Ap+ and y S moles of Bq-.
Ksp = [Ap+]x [Bq-]y = [x5]x [y5]y
= xx y y 5 x+y
New Question
10 months agoContributor-Level 10
We cannot predict the difficulty level of the UK 10th Science paper 2026. UK 10th Board exams are usually on the easier side of the spectrum but it totally depends on your preparation level and how the board is going to set the paper. It also depends on the checking. You have to prepare well in your board exams for scoring well. It is advised that you complete the UK 10th Science syllabus 2026 and know the exam pattern well. You must also solve the previous years' question papers for better preparation of the exam.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a matching answer type question as classified in NCERT Exemplar
Racemization is the process of converting enantiomers into a racemic mixture, which is a mixture comprising two optically active enantiomers in equal quantities, from the results of the SN1 reaction. Chemical fire extinguishers are chlorobromocarbons.
Bromination of alkenes produces vic-dibromides, which are formed when halogen atoms of the same kind are linked to nearby carbon atoms of the molecule. Alkylidene dihalides, sometimes called gem-dihalides, are dihalides that have two halogen atoms bonded to the same carbon atom. The Saytzeff rule governs the removal of&
New Question
10 months agoContributor-Level 10
This is a matching answer type question as classified in NCERT Exemplar
The medicinal uses are linked to the halogen derivatives. Chloramphenicol is an antibiotic used to treat typhoid fever, thyroxine is a hormone whose dysregulation causes goitre, chloroquine is a malaria medication, and chloroform is often used as an anaesthetic.
Correct Answer: (i) → (c) (ii) → (d) (iii) → (a) (iv) → (b)
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Heat an alkyl chloride/bromide in the presence of a metallic fluoride such as AgF, Hg2F2, CoF2, or SbF3 to produce alkyl fluorides. Swarts reaction is the name given to this reaction. CoF2 and Hg2F2 are the only choices that work as reagents. NaF is not suitable because the end products of NaF + RCl→ RF + NaCl are soluble in the solvent, i.e. water, making separation difficult. As a result, the right alternatives are (ii) and (iii).
Correct Answer: Option (ii) and (iii)
New Question
10 months agoContributor-Level 10
Yes, MKGVU has an impressive placement record. Tabulated below is the placement data for the past three years as per the NIRF Report 2025:
Particulars | 2022 | 2023 | 2024 |
|---|---|---|---|
Total Students | 1,241 (UG 3-year) 33 (UG 4-year) 23 (PG 1-year) 1,417 (PG 2-year) | 1,429 (UG 3-year) 35 (UG 4-year) 29 (PG 1-year) 1,374 (PG 2-year) | 1,236 (UG 3-year) 33 (UG 4-year) 54 (UG 5-year) 35 (PG 1-year) 1,326 (PG 2-year) |
No. of Students Placed | 434 (UG 3-year) 18 (UG 4-year) 12 (PG 1-year) 354 (PG 2-year) | 569 (UG 3-year) 22 (UG 4-year) 17 (PG 1-year) 57 (PG 2-year) | 510 (UG 3-year) 19 (UG 4-year) 50 (UG 5-year) 21 (PG 1-year) 455 (PG 2-year) |
Median Salary | INR 2.40 LPA (UG 3-year) INR 5.50 LPA (UG 4-year) INR 6.20 LPA (PG 1-year) INR 4.20 LPA (PG 2-year) | INR 3.37 LPA (UG 3-year) INR 6 LPA (UG 4-year) INR 6.50 LPA (PG 1-year) INR 3.37 LPA (PG 2-year) | INR 3.50 LPA (UG 3-year) INR 4.50 LPA (UG 4-year) INR 5.55 LPA (UG 5-year) INR 6 LPA (PG 1-year) INR 3.50 LPA (PG 2-year) |
NOTE: The above data is obtained from the NIRF report 2025
New Question
10 months agoContributor-Level 10
Yes, NIIT University provides scholarships to MBA students based on academic merit and financial need. These scholarships help ease the financial burden for deserving candidates. Applicants are encouraged to visit the university's official website to check the detailed scholarship criteria and application process.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts