Note: Candidates are advised not to base their decisions solely on this placement data, as it includes only one student and alumni reviews and feedback.
Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
BBA admission at VIT Vellore is entirely merit-based. There is no entrance exam requirement. Selection is determined by a candidate's performance in Class 12, where a minimum of 60% aggregate marks from a recognised board is mandatory for eligibility.
New Question
10 months agoContributor-Level 10
For 2025, GATE CCMT has been released across different categories under the AI quota. For the General AI category students, the 2025 Round 1 cutoff score for Computer Engineering admission stood at 681. Candidates seeking admission can refer to the table below to view the MANIT Jaipur cutoff 2025 for Computer Engineering through GATE CCMT scores across all the student categories:
| Category | GATE CCMT Round 1 Cutoff 2025 |
|---|---|
| General | 681 |
| OBC | 651 |
| SC | 506 |
| ST | 389 |
| EWS | 660 |
New Question
10 months agoContributor-Level 10
VIT Vellore BBA Applicants must upload scanned copies of their Class 12 marksheet, passport-sized photograph, signature, and other documents as specified in the application form. All documents must be in the correct format as per the guidelines provided by VIT Vellore during the admission process.
New Question
10 months agoContributor-Level 10
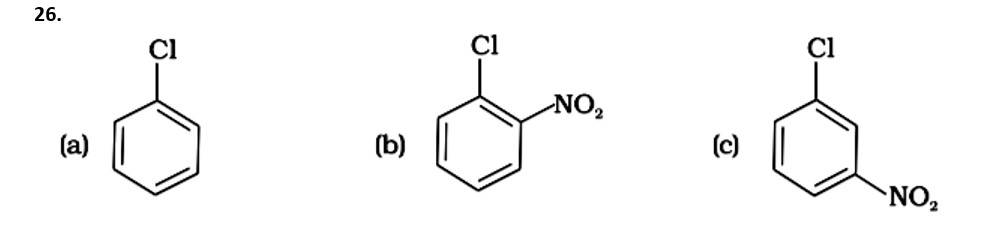
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (iii).
Due to the resonance stabilisation of the benzene ring, the reactivity of aryl halides to nucleophilic substitution is exceedingly low. Because of resonance, the -Cl bond gains a partial double bond. The presence of an electron withdrawing -NO2 group at ortho or para positions on the ring enhances the reactivity of aryl halides. The presence of -NO2 near the C-Cl makes the molecule more reactive. The order of reactivity should be (b) > (c) > (a). The right option is (iii).
New Question
10 months agoContributor-Level 10
The final step in securing admission to the BBA course at VIT Vellore is fee payment. Once selected, students must pay the course fee, which ranges from INR 1.1 Lacs to INR 5.67 Lakh, to confirm their seat in the university's BBA programme.
New Question
10 months agoContributor-Level 10
VIT Vellore BBA admission is merit-based. Students must secure 60% marks in Class 12 from the recognised board.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
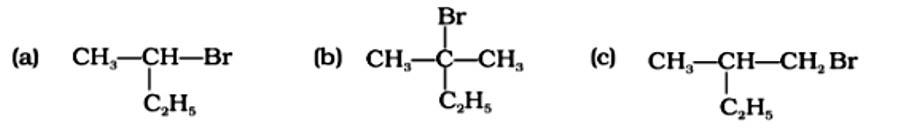
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (i).
A racemic mixture is one that contains two enantiomers in equal quantities but has no optical activity because the opposing optical rotations of the two enantiomers cancel each other out. The optically active reactant undergoes the SN1 reaction in order for a racemic mixture to form following nucleophilic substitution. Option (a) is a chiral carbon atom that will undergo the SN1 process, resulting in a racemic mixture. Option (b) does not include an asymmetric carbon, but option (c) has a secondary carbon asymmetric atom that is less reacti
New Question
10 months agoContributor-Level 10
The KK University BSc fee structure includes tuition fees, exam fees, and admission fees. Additional hostel and mess charges are payable by candidates seeking accommodation at the university. The fee varies across the BSc specialisations offered at the university. The following table presents the total KK University BSc tuition fee for various specialisations:
| Fee Component | Tuition Fee |
|---|---|
| BSc (Hons) in Agriculture | INR 2.4 Lakh |
| BSc (Hons) (Chemistry/ Zoology/ Botany/ Physics/ Mathematics) | INR 16,000 |
| BSc (Chemistry/ Zoology/ Botany/ Physics/ Mathematics) | INR 12,000 |
NOTE: The mentioned fee is sourced from the official website of the university/ sanctioning body. It is still subject to change and hence, is indicative.
New Question
10 months agoContributor-Level 10
Ganga Institute of Technology and Management offers Golden Eagle Awards to meritorious students on the basis of marks obtained by them in the qualifying exam. Students must satisfy the eligibility criteria and provide the required documents to be eligible for this award. The institute provides the following amount to students who meet the eligibility:
| Marks | BTech | BBA/ BCA | MBA | MCA |
|---|---|---|---|---|
| 90 & above | INR 50,000 | INR 20,000 | INR 40,000 | INR 30,000 |
| 85 - 89.99 | INR 25,000 | INR 10,000 | INR 20,000 | INR 15,000 |
| 80- 84.99 | INR 10,000 | INR 5,000 | INR 10,000 | INR 7,500 |
Note: This information is taken from official sources and is subject to change.
New Question
10 months agoContributor-Level 10
The highest placements package offered to BCom graduates at JIMS Greater Noida has not been released yet. However, as one of the verified Shiksha reviews of the student from the batch of 2024, claimed the highest package offered to BCom graduates was INR 5.9 lakhs.
New Question
10 months agoContributor-Level 10
There were many top recruiters who participated in the SICSR placements for the BBA programme. However, the latest placement statistics have not been released yet. As per the students' reviews on Shiksha, the top recruiters of the Symbiosis Institute of Computer Studies and Research BBA placement are mentioned below:
- Amazon
- Infosys
- Deloitte
- Tata Consultancy Services
- DE Shaw India Pvt. Ltd
- Cognizant
- ZS Associates, etc.
New Question
10 months agoNew Question
10 months agoNew Question
10 months agoContributor-Level 10
After the completion of a BCom from JIMS Greater Noida, candidates can apply for different government jobs. Graduates tend to seek employment in public administration, teaching, and social services. Candidates can apply for competitive exams such as UPSC, SSC, and State Public Service Commissions for the positions of IAS, IPS, or other government departments. Besides, there are opportunities in banking, railways, and other PSUs, where a BCom degree is a qualifying criterion. Hence, with appropriate preparation and focus, graduates in BCom can get employment in the government sector.
New Question
10 months agoContributor-Level 7
Anna University accepts TNEA scores for admission to the B.E and B.E Integrated courses. According to TNEA last-round Seat allotment results, the CSE cutoff ranged between 44 and 194103 for the General AI category candidates for B.Tech. in Computer Science and Engineering. For OBC AI category, the CSE cutoff ranged between 83 to 67513 for the affiliated colleges under Anna University via TNEA. Similarly, for ECE, the cutoff ranged between 107 to 194579 for the affiliated colleges under the Anna University for General AI quota.
New Question
10 months agoContributor-Level 10
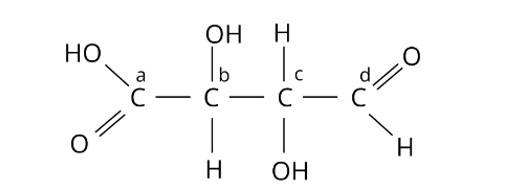
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (ii).
Chiral molecules are made up of one carbon atom surrounded by four different species. Because of the presence of two or more identical groups, such as hydrogens, all straight chain molecules cannot be chiral. Even carbons with double or triple bonds to a group are not considered chiral. Through covalent connections, an asymmetric carbon must be surrounded by four distinct species. As a result, atoms b and c are asymmetric. The correct answer is option (ii).
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (i).
When benzyl chloride is treated with aqueous sodium hydroxide, where -OH is the nucleophile, a nucleophilic substitution reaction occurs, resulting in the formation of benzyl alcohol. The benzene ring is resonance stabilised here, and this stability is extended to the connected methylene group, giving a positive charge to -CH2, making the whole carbocation stable when the link between benzyl and bromide is broken. This is an SN1 reaction with two stages that is followed due to the stability of the carbocation. This is a coordinated reaction
New Question
10 months agoContributor-Level 10
BSc admission at KK University is subject to the fulfilment of the eligibility criteria specified by the university. Further, candidates are required to present all the necessary documents establishing their eligibility at the time of admission. The KK University BSc eligibility criteria are given below:
- To be eligible for admission to a BSc (Hons) in Agriculture, candidates must have passed Class 12 with PCM/ PCB/ Agriculture with 40%
- To be eligible for all other BSc and BSc (Hons) specialisations, candidates must have passed Class 12 in the Science stream with at least 45% in the respective subject
New Question
10 months agoContributor-Level 10
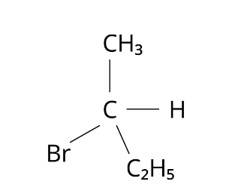
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (i).
The structure of 2-Bromobutane is depicted in the diagram below. Because all of the groups connected to the central carbon atom differ, the mirror image of the molecule cannot be superimposed on the original molecule.
The structure of 1-Bromobutane is shown in the diagram below. Because there are just three groups that vary from one another, the molecule is not chiral in nature.
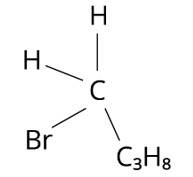
The structure of 2-Bromopropan-2-ol, shown below, has two identical species and hence cannot be chiral.

Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts