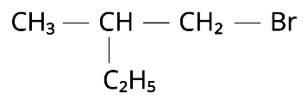Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
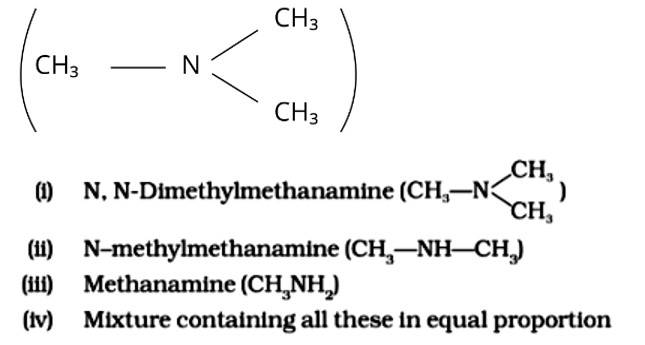
The Correct Answer is Option (iii).
Because it possesses unpaired electrons, the ammonia molecule is a nucleophile in nature. By nucleophilic substitution, this nucleophile attacks the chloromethane molecule and forms methyl amine or methanamine. The carbon atom in the molecule is partially positive due to the electronegativity of the linked halide, which is partially negative. The positive ion is attacked by the electron-rich nucleophile, causing the halide ion to be detached from the molecule. The proper response is (iii).
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
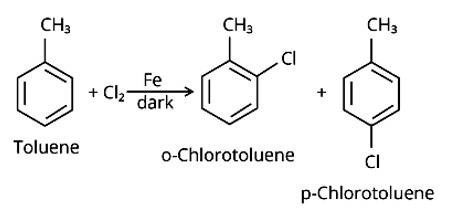
The Correct Answer is Option (iv).
By electrophilic substitution, aromatic arenes react with chlorine in the presence of Lewis acid catalysts such as iron (III) chloride, yielding ortho and para isomers of haloarenes. (ii) and (iii) are both products of the reaction. Cl2 forms a coordination complex with FeCl3, creating the Cl + FeCl4 - complex, which has a small positive charge on Cl and a negative charge on FeCl4 - . This Cl+ then interacts with the aromatic double bonds of the toluene molecule to create an addition product, which is su
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (ii).
We may conclude from the given compound's common name that there are two -C2H5 groups and one -Br group linked to a CH4 molecule. Bromomethane would be represented by the symbol CH3-Br. Two ethyl groups can be replaced for the hydrogens in the methyl molecule to make diethylbromomethane (C2H5)CH (C2H5)Br. The longest parent chain must be identified in order to offer an IUPAC name. The parent chain is CH3-CH2-CH (Br)-CH2-CH3, with the -Br group from either side connected to the third carbon atom. Because the parent chain has five carbons,
New Question
10 months agoContributor-Level 10
As per students' reviews accessed from Shiksha, the average package during SICSR Pune placements for BBA students tends to range between INR 3 LPA and INR 6 LPA. The highest package for BBA was INR 22 LPA, indicating good placement possibilities. However, the latest SICSR Pune placements for BBA have not been released yet. The aforementioned data is as per the students' review. It is still subject to change.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Option (iii)
The correct answer is option iii.
First, we must determine which carbon chain is the longest. After that, we should have CH3-CH2-CH (CH3)-CH2-Br as the real structure. -Br, because the functional halide group is linked to the first carbon atom, we begin numbering there. The methyl group branch is joined to the chain's second carbon atom. Butane is named from the number of carbons in the unbranched parent chain, which is four. 1-Bromo-2-methylbutane is the name of the molecule. Option (iii) is the right answer.
New Question
10 months agoContributor-Level 10
Yes, KK University, offers a three-year BSc through its School of Applied Sciences. The university also offers a four-year BSc (Hons). These courses are offered in five specialisations, including Chemistry, Botany, and Zoology among others. Further, a four-year BSc (Hons) in Agriculture is also available at the university. All these courses are offered in the full-time mode. The BSc curriculum at the university follows the semester pattern. For more information, interested candidates can visit the university's official website or contact the admission team directly.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (iv).
SN1 reactions occur mostly in polar protic solvents such as H2O and follow first-order kinetics. This indicates that the reaction rate is solely determined by one reactant. Because of the great stability of the generated carbocation, this reaction favours tertiary alkyl halides. When a molecule is polarised in water, it generates a carbocation as well as a halide ion. The halides' reactivity is R-I > R-Br > R-Cl > > R-F. As a result, (CH3)3C-I will be the most likely to undergo the reaction.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (ii).
When an alkyl halide with -hydrogen atoms reacts with a base or a nucleophile, depending on the nature of the alkyl halide, the strength of the base or nucleophile, the size of the molecules, and the reaction circumstances, it can undergo a specific kind of reaction. A substitution or elimination process can be followed by an alkyl halide, as well as the two kinds of substitution, SN1 and SN2. Because the entering nucleophile cannot engage with the bulky substituents on or near the carbon atom, the main alkyl halide prefers the SN2reaction
New Question
10 months agoContributor-Level 10
Students wishing to study a BBA course can consider criteria to assess which college is better than the Symbiosis Institute of Computer Studies and Research. Some of the key criteria include specialisations offered, student ratings, fees charged, placement percentage, return on investment, faculty credentials, and several other factors. Once these criteria have been reviewed, students can choose their intended course of study.
New Question
10 months agoContributor-Level 10
Yes, the last date to apply for BSc in Yoga is revised by Alagappa University. Eligible candidates can now apply till Jun 25, 2025. The application should be filled out carefully and as per the official documents.
New Question
10 months agoContributor-Level 10
Start by focusing only on the subjects you failed in and understand where you went wrong. Revise important topics, practice past papers, and take help from teachers or online tutorials. Make a daily timetable and stick to it, even if you have less time. Use your previous question papers to understand the exam pattern. Confidence and focused revision can help you clear the exam with better marks.
New Question
10 months agoContributor-Level 10
The University of California Los Angeles and University of California Berkeley are some of the most reputed universities in USA. Both universities offer something unique for its international students. When choosing a university student must check all the crucial factors about the university. Some crucial factors include fee, location, scholarship offered, acceptance rate, placement rate, and more.
UCLA placement rate stands at 98%, and the graduates are employed within 6 months of graduation. This means out of every 100 students, around 98 students secured employment. Whereas, the University of California Berkley placement rate stands
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
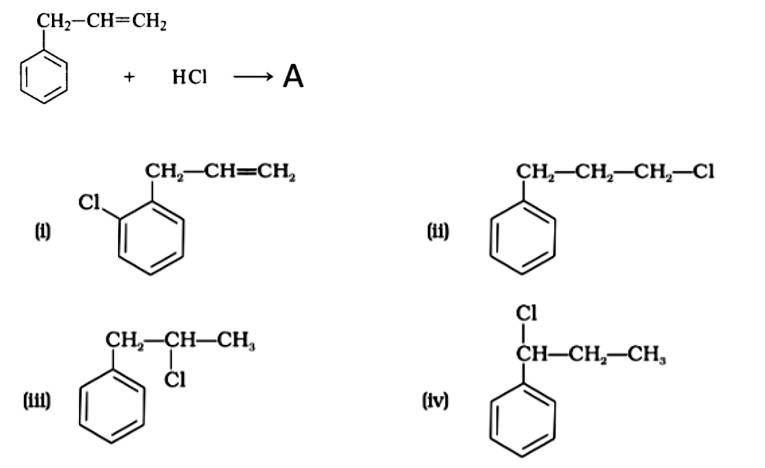
The correct answer is Option (iii).
Markonikov's rule can be used to explain the results. The hydrogen from HCl is added to the carbon immediately bonded to the most hydrogen atoms, whereas the -Cl is attached to the carbon directly bonded to the least hydrogens, according to the rule. As a result, the right response is (iii).
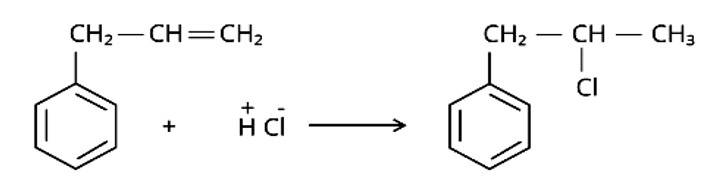
New Question
10 months agoContributor-Level 10
If you fail the compartment exam, you may need to repeat the academic year or wait for the next chance (if offered). The JAC usually allows only one compartment attempt per subject in a year. So, it is important to take this exam seriously and prepare well. You can also explore open schooling options or skill-based courses in the meantime. Talk to your school or counselors for guidance on your next steps.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (ii).
Dihaloalkanes with two halogen atoms of the same kind bonded to the same carbon atom are known as gem-dihalides. Alkylidene dihalides is the most frequent naming scheme for gem-dihalides. As a result, ethylidene dichloride is a gem-dihalide. The best choice is (ii).
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The correct answer is option ii.
Aluminum chloride is a Lewis acid catalyst that functions similarly to FeCl3. By chlorinating benzene in the presence of AlCl3, benzene is transformed to chlorobenzene. Electrophilic substitution is used to carry out the reaction. Cl2 forms a coordination complex with AlCl3 called Cl + AlCl4- , which has a small positive charge on Cl and is negatively charged on AlCl4- . This Cl+ then interacts with the benzene ring's aromatic double bond to generate an addition product, followed by deprotonation to produce chlorobe
New Question
10 months agoContributor-Level 10
The Jharkhand Board class 10 compartment result is for students who reappear in failed subjects, while the regular result is for those who appeared for all subjects in the main exam. Both results are declared online and contain the same format, showing marks and pass/fail status. The final mark sheet after the compartment exam will replace your failed marks with the new ones. There is no separate mention of it being a compartment result. So, once you pass, your mark sheet looks just like any other student's.
New Question
10 months agoContributor-Level 9
The leadership development programme at the MIT College of management develops future ready leaders. It consists of classroom learning, conferences and talks of entrepreneurs in industry. Students are involved with simulations and conferences as well as with peer learning experiences. The aim is to get proud, ethical, and visionary practitioners.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The correct answer is Option (i).
When the halogen atom is linked to an sp3-hybridised carbon atom next to an allylic carbon, a carbon-carbon double bond is formed.
Aryl halides are produced when a halogen atom is directly linked to an aromatic ring's sp2-hybridised carbon atom.
When a halogen atom is linked to a sp2-hybridised carbon atom in a carbon-carbon double bond, vinylic bonds are produced. Simply defined, it's an ethylene molecule with one hydrogen atom substituted by another -R group.
When the halogen is attached to a secondary carbon atom in the molecule, secondary
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts