Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
Because it is a tertiary alkyl halide, the described reaction is a nucleophilic substitution that follows the SN1 mechanism. It's a two-step process. The first step is the gradual breaking of a polarised C-Br bond, which results in the formation of a carbocation and a bromide ion. To complete the substitution, the stable intermediate carbocation is attacked by the nucleophile OH−. (i) and (iv) are the right statements.
Correct Answer: option (A) and (iv)
New Question
10 months agoBeginner-Level 5
City allotment list for AIAPGET 2025 was released on June 24, 2025 online. Candidates will have to enter application number, date of birt and security pin to download AIAPGEt 2025 city allotment slip. The city allotment list will make candidates know about the particular city where his/she centre will be there. Name and address of exact centre for AIAPGET 2025 will be informed through AIAPGET 2025 admit card. NTA has uploaded AIAPGET 2025 admit card on June 30, 2025.
New Question
10 months agoContributor-Level 10
The Mahatma Gandhi Kashi Vidyapith University placement report for 2024 is not out yet. However the NIRF report for 2025 has been released.Check out the tabulated data given below to know more about Mahatma Gandhi Kashi Vidyapith University placement 2024:
Course | Total Students | Students Placed | Median Salary |
|---|---|---|---|
UG 3-year | 1,236 | 510 | INR 3.50 LPA |
UG 4-year | 33 | 19 | INR 4.50 LPA |
UG 5-year | 54 | 50 | INR 5.50 LPA |
PG 1-year | 35 | 21 | INR 6 LPA |
PG 2-year | 1,326 | 455 | INR 3.50 LPA |
NOTE: The above data is obtained from the NIRF report 2025
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
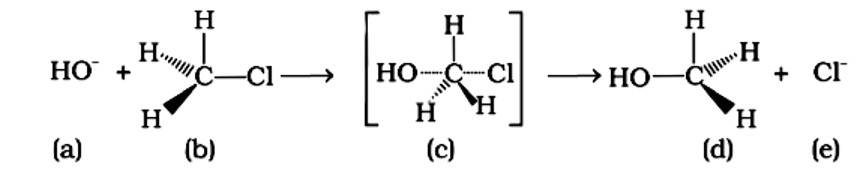
Because the carbon atom is transiently linked to five atoms, the intermediate (iii) is less stable than reactant (ii), which is coupled to four groups.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
An SN2 reaction, also known as a nucleophilic substitution, is the reaction in question. The entering nucleophile causes the groups surrounding the carbon atom to migrate in the opposite direction of the nucleophile, causing the configuration of alkyl halides to invert. As a result, (b) and (d) will be in the opposite order. This reaction is also SN2 since there is just one phase in which OH- is added and Cl- is removed at the same time. (i) and (ii) are the right answers.
Correct Answer: Option (A) and (B)
New Question
10 months agoContributor-Level 10
According to the currently available information, there is no provision for direct admission to the BSc course of KK University. Admission is granted to applicants based on their performance in the selection rounds conducted by the university. These rounds include University Entrance Test, GD and/or PI. Additionally, applicants are required to fulfil the course-specific eligibility criteria specified by the university. Hence, getting direct admission to the BSc programme at KK University is not possible.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
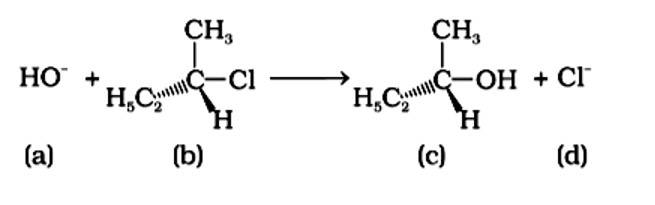
An SN2 nucleophilic substitution reaction occurs when CH3Cl interacts with the hydroxide ion to produce CH3OH. Both OH - and Cl - are nucleophiles with an excess of electrons in this reaction. Because the binding of OH - and the leaving of Cl - occurs simultaneously in the transition state (c), the carbon atom is linked to just three hydrogens. As a result, the carbon atom becomes sp2 hybridised. Aand (C) are the right statements.
Correct Answer: Option (A) and (C)
New Question
10 months agoNew Question
10 months agoContributor-Level 10
MNNIT Allahabad GATE cutoff 2025 has been released for Round 1 via CCMT counselling for MTech admission. As per the Round 1 cutoff, the closing scores were in the range of 353 and 711 for the students belonging to the General AI quota. Please note that the cutoff scores vary for different rounds, specialisations, and categories for MTech admission across multiple specialsiations. Candidates seeking admission can refer to the table below to view the MNNIT Allahabad MTech cutoff 2025 for General category students.
| Course | GATE CCMT Round 1 Cutoff 2025 |
|---|---|
| M.Tech. in Fluids Engineering | 353 |
| M.Tech. in Biomedical Engineering | 358 |
| M.Tech. in Engineering Mechanics and Design | 362 |
| M.Tech. in Thermal Engineering | 366 |
| M.Tech. in Production Engineering | 375 |
| M.Tech. in Chemical Engineering | 382 |
| M.Tech. in Geotechnical Engineering | 389 |
| M.Tech. in Environmental Engineering | 393 |
| M.Tech. in Power System | 401 |
| M.Tech. in Transportation Engineering | 408 |
| M.Tech. in Control and Instrumentation | 410 |
| M.Tech. in Design Engineering | 428 |
| M.Tech in Geoinformatics | 442 |
| M.Tech. in Power Electronics and Drives | 460 |
| M.Tech. in Structural Engineering | 468 |
| M.Tech. in Communication Systems | 475 |
| M.Tech. in Signal Processing | 499 |
| M.Tech. in Biotechnology | 513 |
| M.Tech. in Microelectronics and VLSI Design | 631 |
| M.Tech. in Information Security | 661 |
| M.Tech. in Computer Science and Engineering (AI and Data Science) | 681 |
| M.Tech. in Computer Science and Engineering | 711 |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (iv).
If the halogen atom in all of the supplied alkyl halides is the same kind, the boiling point of a compound rises as the number of alkyl groups added increases. As the quantity and size of molecules and electrons grow, so do their attractions. As a result, the greater the boiling point, the larger the molecular weight of the alkyl halide. The boiling points of the following compounds are listed in ascending order: 1-Bromoethane 1-Bromopropane 1-Bromobutane Bromobenzene The right answer is (iv).
New Question
10 months agoContributor-Level 10
Mahatma Gandhi Kashi Vidyapith University provides good placements to its graduating students. The students are given excellent placement support every year placing them in top companies. Check out the table below to know the key highlights of theMahatma Gandhi Kashi Vidyapith University Placement 2024:
Course | Total Students | Students Placed | Median Salary |
|---|---|---|---|
UG 3-year | 1,236 | 510 | INR 3.50 LPA |
UG 4-year | 33 | 19 | INR 4.50 LPA |
UG 5-year | 54 | 50 | INR 5.50 LPA |
PG 1-year | 35 | 21 | INR 6 LPA |
PG 2-year | 1,326 | 455 | INR 3.50 LPA |
NOTE: The above data is obtained from the NIRF report 2025
New Question
10 months agoContributor-Level 10
Arka Jain University offers a vast range of courses in diverse disciplines. Students can choose a course from Humanities, Law, Management, Science, etc. The Master of Business Administration (MBA) programme offers a good Management foundation to students. The total tuition fees for Arka Jain University MBA is INR 4.50 lakhs, with an additional one-time payment of INR 12,000.
The total amount adds up to INR 4.62 lakhs for the course. Students requiring financial assistance can reach out for the various scholarship schemes available at Arka Jain University.
New Question
10 months agoContributor-Level 10
Mahatma Gandhi Kashi Vidyapith University has been providing successful placements over the years. The students are given excellent placement support every year placing them in top companies. Check out the table below to know the key highlights of the Mahatma Gandhi Kashi Vidyapith University Placement 2024:
Course | Total Students | Students Placed | Median Salary |
|---|---|---|---|
UG 3-year | 1,236 | 510 | INR 3.50 LPA |
UG 4-year | 33 | 19 | INR 4.50 LPA |
UG 5-year | 54 | 50 | INR 5.50 LPA |
PG 1-year | 35 | 21 | INR 6 LPA |
PG 2-year | 1,326 | 455 | INR 3.50 LPA |
NOTE: The above data is obtained from the NIRF report 2025
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (i).
The intermolecular force of attraction will grow as the surface area increases. As a result, the boiling point rises as well. The boiling point of a comparable alkyl halide rises with increasing molecular mass. Iodine has the greatest atomic mass. As a result, 1-iodobutane has the greatest boiling point.
New Question
10 months agoContributor-Level 10
Mahatma Gandhi Kashi Vidyapith University provides good placements to its graduating students. The students are given excellent placement support every year placing them in top companies. Check out the table below to know the key highlights of the Mahatma Gandhi Kashi Vidyapith University placements 2024:
Course | Total Students | Students Placed | Median Salary |
|---|---|---|---|
UG 3-year | 1,236 | 510 | INR 3.50 LPA |
UG 4-year | 33 | 19 | INR 4.50 LPA |
UG 5-year | 54 | 50 | INR 5.50 LPA |
PG 1-year | 35 | 21 | INR 6 LPA |
PG 2-year | 1,326 | 455 | INR 3.50 LPA |
NOTE: The above data is obtained from the NIRF report 2025
New Question
10 months agoContributor-Level 10
Mahatma Gandhi Kashi Vidyapith University Placement is known for its excellent placement record. Check out the tabulated data given below to know more about Mahatma Gandhi Kashi Vidyapith University placements 2024:
Course | Total Students | Students Placed | Median Salary |
|---|---|---|---|
UG 3-year | 1,236 | 510 | INR 3.50 LPA |
UG 4-year | 33 | 19 | INR 4.50 LPA |
UG 5-year | 54 | 50 | INR 5.50 LPA |
PG 1-year | 35 | 21 | INR 6 LPA |
PG 2-year | 1,326 | 455 | INR 3.50 LPA |
NOTE: The above data is obtained from the NIRF report 2025
New Question
10 months agoContributor-Level 10
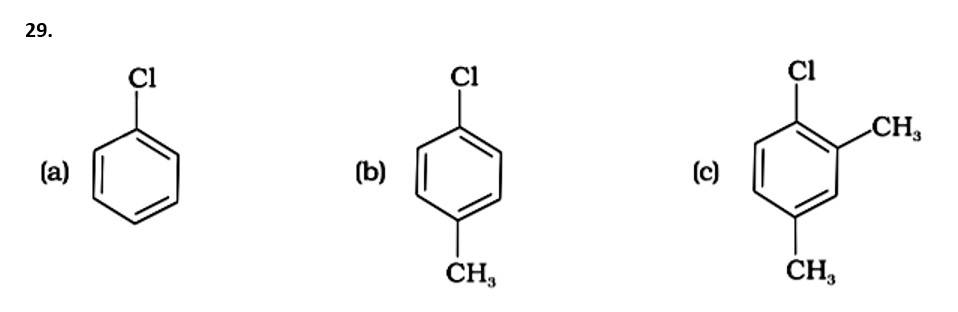
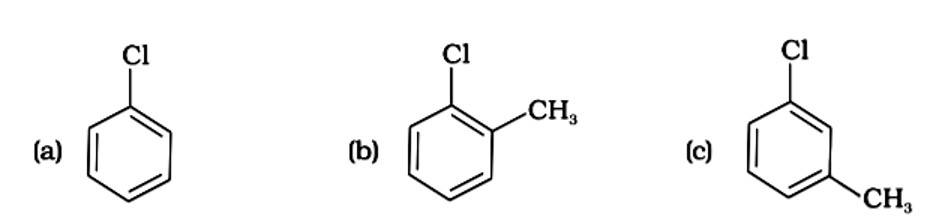
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (iii).
The presence of electron releasing groups increases the reactivity of aryl halides; the fewer the electron releasing groups, the slower the rate of nucleophilic substitution. As a result, an increase in methyl groups decreases reactivity. The sequence of reactivity is (a) > (b) > (c). The right answer is (iii).
New Question
10 months agoContributor-Level 10
Yes, the Symbiosis Institute of Computer Studies and Research does provide scholarships to meritorious BBA students. Such scholarships are offered to students on the basis of their merit, financial requirements, etc. Among the more popularly recognised scholarships is the Symbiosis Merit Scholarship, which is given to students who have shown academic excellence, the Symbiosis Need-Based Scholarship for students who have emerged from a financially challenged background, and the Symbiosis Sports Scholarship for our sportswomen/ sportsmen who have excelled in their sport. These scholarships grant financial aid to deserving students to stu
New Question
10 months agoContributor-Level 10
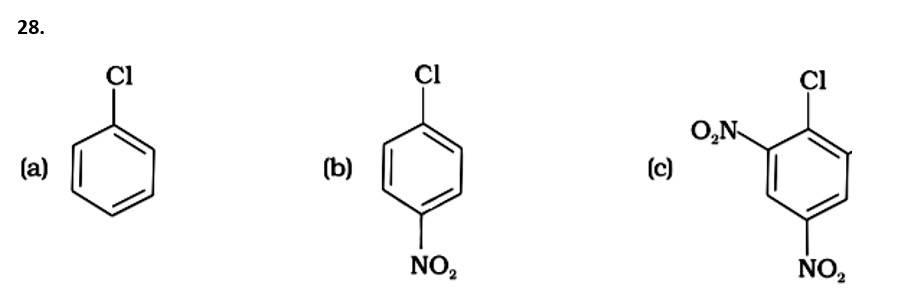
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is option (iv).
( - NO2) is an electron-withdrawing group that induces nucleophilic substitution when it is in the ortho and para positions. When this identical electron-drawing group is in the meta position, its impact is much reduced. The reactivity of aryl halides rises as the number of electron-withdrawing groups increases. As a result, the greater the number of electron-drawing groups, the greater the rate of nucleophilic substitution.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (iv).
Because the -CH3 group is an electron releasing group, it reduces the reactivity of aryl halides in the ortho and para locations. As a result, aryl halides without electron releasing groups are more reactive. As a result, the order of reactivity is (a) > (c) > (d) (b). The right answer is (iv).
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts