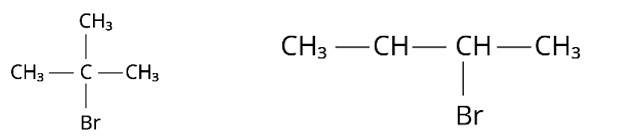Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Michigan Tech has four accommodations which include Douglass Houghton Hall, McNair Hall, Wadsworth Hall and Hillside Place Apartments. These provide facilities such as study lounge, kitchen, sauna, laundry service, a reception, fitness area and many more. It has been declared as the safest public college in Michigan. Currently there are 2265 students living in these resident halls.
New Question
10 months agoContributor-Level 10
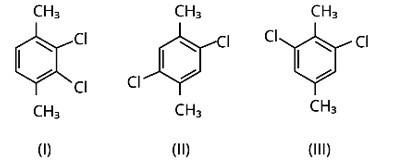
This is a short answer type question as classified in NCERT Exemplar
Because both methyl groups and chlorine atoms are symmetrically positioned at para-positions in compound (II), these molecules fit better in the crystal lattice than other isomers, and hence have the greatest melting point.
New Question
10 months agoContributor-Level 7
Swami Vivekanand Institute of Pharmacy and Research fee for DPharma course comprises different components such as Tuition Fees, Caution Fees, etc. Find below the total tution fees for both the years of the programme:
- Tuition Fees (First & Second year): INR 52,500
- Caution Deposit (First & Second year): INR 750
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The addition reaction of the alkene produces two products, as indicated below.
CH3−CH2−CH=CH−CH3 + HCl→ A + B
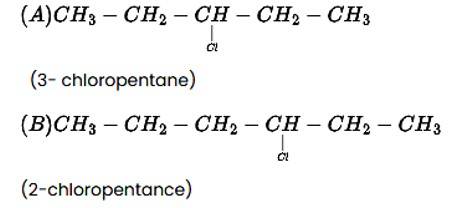
New Question
10 months agoNew Question
10 months agoContributor-Level 10
NIT Nagaland M.Tech admission in the OBC category requires the student to achieve 450 or higher \during the Round 1 cutoff list. Considering the competitive scores, the most competitive M.Tech specialisations during Round 1 admission was Computer Science and Engineering (450) for the OBC category students.
Check the specialisation-wise cutoff information below:
| Course | GATE CCMT Round 1 Cutoff 2025 |
|---|---|
| M.Tech. in Communication Engineering | 327 |
| M.Tech. in Computer Science and Engineering | 450 |
| M.Tech. in Power Systems Engineering | 322 |
| M.Tech. in VLSI Systems | 362 |
New Question
10 months agoContributor-Level 7
Swami Vivekanand Institute of Pharmacy and Research admission to DPharm is completely entrance-based. Check below the detailed admission criteria for the programme:
| Course Name | Eligibility | Selection Criteria |
|---|---|---|
| DPharma | Pass Class 12 with minimum 45% aggregate in Physics, Chemistry and Mathematics or Biology as subjects | PPHT |
New Question
10 months agoContributor-Level 10
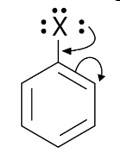
This is a short answer type question as classified in NCERT Exemplar
When an arene is treated with Cl2 in the presence of FeCl3, the electrophilic addition reaction chlorinates the arene to produce aryl chloride. As a result, the chemical provided is C6H5-CH3, often known as toluene. o-chlorotoluene and p-chlorotoluene are the results of electrophilic substitution chlorination.
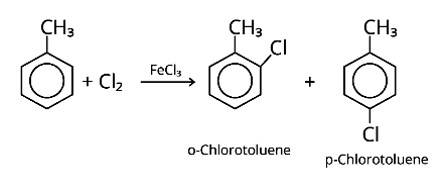
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
(i) Because the rate of reaction of compound ‘A' with aqueous KOH is solely dependent on the concentration of ‘A, ' the reaction mechanism is SN1and ‘A' is 2-Bromo-2-methylpropane (tertiary bromide).
‘B', on the other hand, is an isomer of ‘A' and is optically active. As a result, ‘B' has to be 2-Bromobutane. Because the rate of reaction of ‘B' with aqueous KOH is determined by its concentration, the reaction mechanism is SN2.
(ii) Compound ‘B' will have an inverted conformation as a result of the SN2 reaction,
New Question
10 months agoContributor-Level 10
NIT Nagaland accepts the GATE entrance exam, followed by the CCMT counselling process for admission to the M.Tech programme. For 2025, the Round 1 cutoff has been released, wherein the scores were in the range of 350 and 486 for the students belonging to the General AI quota. The cutoff scores vary for different rounds, courses, and categories. Candidates can refer to the table below to view the category-wise GATE CCMT Round 1 cutoff scores for M.Tech admission:
| Category | GATE CCMT Round 1 Cutoff 2025 |
|---|---|
| General | 350 - 486 |
| OBC | 322 - 450 |
| SC | 231 - 348 |
| ST | 252 - 341 |
| EWS | 355 - 452 |
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
(i) 1-Bromobut-2-ene: It is a primary halide.
(ii) 4-Bromopent-2-ene: Here Bromine is attached to the secondary carbon. Hence, it is a secondary halide.
(iii) 2-Bromo-2-methylpropane: Here Bromine is attached to the tertiary carbon. Hence, it is a tertiary halide.
New Question
10 months agoContributor-Level 7
Swami Vivekanand Institute of Pharmacy and Research fee structure for BPharm comprises various components. The fees is to be paid semester-wise. Find below the fees for all eight semesters of the four-year BPharm programme at Swami Vivekanand Institute of Pharmacy and Research:
- Sem 1: INR 32,950
- Sem 2: INR 32,950
- Sem 3: INR 33,700
- Sem 4: INR 33,700
- Sem 5: INR 32,950
- Sem 6: INR 32,950
- Sem 7: INR 32,950
- Sem 8: INR 32,950
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
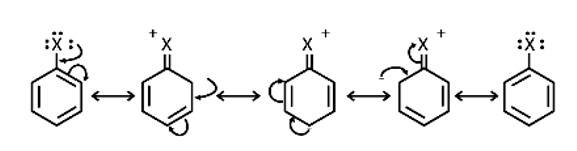
Because electron density is higher at ortho and para locations, the functional groups contained in these compounds are ortho-para directed.
New Question
10 months agoContributor-Level 10
The NCERT Exemplar questions go beyond the textbook questions as they are designed to be more challenging and conceptually advanced. Practicing the NCERT exemplar provides a better understanding of complex topics like conductance and electrochemical cells. It also helps in better preparation for the entrance exams. The exemplar often involves multi-concept, application-based, and analytical problems.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Because energy is required to overcome the attractions between the haloalkane molecules as well as to break the hydrogen bonds between water molecules in order to dissolve a haloalkane in water, haloalkanes are only weakly soluble in water. New attractions between the haloalkane and the water molecules, on the other hand, release less energy since they are weaker than the water's initial hydrogen bonds.
New Question
10 months agoContributor-Level 7
Swami Vivekanand Institute of Pharmacy and Research offers courses at UG and Diploma levels in the field of Pharmacy. These are BPharma and DPharma. Students can also enroll for direct admission in the second year of BPharm through Lateral Entry. Admission to both the programmes at Swami Vivekanand Institute of Pharmacy and Research is entrance-based.
New Question
10 months agoBeginner-Level 1
Yes, you can pursue B.Tech in biotechnology with PCB in many colleges like Brainware university, University of Engineering and Management, Kolkata, and many more.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
This addition reaction is carried out in accordance with Markovnikoff's rule, which states that in a double bond, the hydrogen from the hydrogen halide is added to the carbon atom with the most hydrogen atoms attached to it, while the halogen atom is attached to the carbon atom with the fewest hydrogens attached to it. The main product in the combination will be the molecule that follows this guideline. As a result, the molecule (B) will be the reaction's main product.
New Question
10 months agoContributor-Level 10
A salt bridge in a galvanic cell maintains electrical neutrality by allowing the ions to flow between the two half-cells. It ensures the continuous flow without participating in the reaction. The salt bridge prevents the charges accumulation as such an accumulation can halt the electrochemical reaction by stopping the flow of electrons.
New Question
10 months agoBeginner-Level 4
To get admission in a Diploma in Electrical Engineering program, you'll typically need:
- 10th pass certificate from a recognised board
- Minimum 35-40% marks in 10th standard
- Some institutions may require entrance exams or interviews
- Age limit varies, but usually, it's between 15-25 years
Documents required:
- 10th mark sheet and pass certificate
- Transfer certificate
- Caste certificate (if applicable)
- Migration certificate (if applicable)
Admission process:
- Online/offline application
- Merit-based selection or entrance exam scores
- Counseling or interview (in some cases)
It's best to check with the institution directly for specific requ
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts