Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 9
MIT College of Management presents MBA in General business, Agri and Food business, and applied data science. There is also a Technology Management, Project & Construction Management and a global MBA with international exposure. There are Executive MBA choices in such fields as HR, Marketing etc. They provide such industry-based programmes which have international alliances.
New Question
10 months agoContributor-Level 10
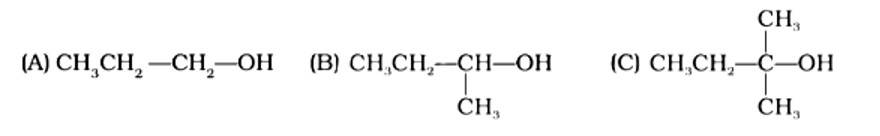
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (ii). Haloalkanes are made by combining alcohols with halogen acids, in which the hydroxyl group of the alcohol is replaced by the halogen. Primary, secondary, and tertiary alcohols are represented by options (A), (B), and (C). Tertiary alcohols are more reactive than secondary and primary alcohols, and they generate haloalkanes from haloacids without the need of catalysts at ambient temperature. Alcohols have a reactivity order of 3? >2? >1? . As a result, the proper option is (ii).
New Question
10 months agoContributor-Level 10
The University of Otago is highly regarded for offering the highest placement rates in the New Zealand. Otago University placement rate attracts international students the most. University of Otago placement rate stands at 95%, and the graduates were employed within six months of graduation. This figure states that out of every 100 students, around 95 students secured employment. The university graduates are highly skilled Strategic Planning, Team Leadership, Strategy, Marketing, Business Strategy, Marketing Strategy, Business Development, etc. The university graduates are in high demand all over the world. University of Otago graduate
New Question
10 months agoContributor-Level 10
Currently, IPU CET cutoffs for JIMS Greater Noida BCom admission for 2025 have not been released. However, the cutoff is expected to be announced soon. To assess the competition level to get admission into the BCom course at JIMS Greater Noida, candidates can check out the last round cutoff ranks for the years 2024, and 2023:
| Course | 2023 | 2024 |
|---|---|---|
| Bachelor of Commerce (BCom Hons) | 2448 | 2078 |
New Question
10 months agoContributor-Level 10
The students should break the long answer and topics into small points and then try to understand them. They should make short notes and revise them.
New Question
10 months agoContributor-Level 10
The University of Otago placement rate is 95%, and the graduates were employed within six months of graduation. University of Otago graduates are working in leading sectors such as Sales, Media and Communication, Marketing, Human Resources, Finance, Entrepreneurship, etc. Some popular companies that hire Otago Uni graduates are mentioned below.
- ANZ
- Deloitte
- Fonterra
- University of Canterbury
- Victoria University of Wellington
- ASB Bank
- EY
New Question
10 months agoNew Question
10 months agoContributor-Level 10
For candidates belonging to the General AI quota, the minimum and maximum GATE scores required were 356 and 407 during Round 1 admission to the M.Tech course. Please note that the cutoff score vary for different rounds, courses and categories. For GATE, the higher the cutoff scores, the higher is the competiton and vice versa. Candidates can refer to the table below to view the Guru Ghasidas Vishwavidyalaya GATE CCMT Round 1 cutoff scores for the students belonging to the General AI quota:
| Course | GATE CCMT Round 1 Cutoff 2025 |
|---|---|
| M.Tech. in Civil Engineering (Structural Engineering) | 356 |
| M.Tech. in Information Technology | 393 |
| M.Tech. in Computer Science & Engineering | 407 |
| M.Tech. in Electronics and Communication Engineering | 366 |
New Question
10 months agoContributor-Level 9
Apply online or submit the offline application to see admission in the MIT College of Management Pune. Attend entrance test and then there are an attitude test, a group discussion and a personal interview. The final selection is done on combination score of all assessment rounds. Candidates who have not taken their results are permitted to be under provisional admission.
New Question
10 months agoContributor-Level 10
Yes, you can complete the RBSE class 10 syllabus without coaching if you follow a smart and regular study routine. Use the official RBSE books, online resources, and video lessons available for free. Practice from sample papers and solve previous year questions to test your preparation. Ask school teachers or classmates if you have doubts. Self-study with dedication is enough to score good marks.
New Question
10 months agoContributor-Level 10
UK 10th Mathematics question paper 2026 will be available only after the conduct of exam. Once the exams are over, we will provide UK 10th Mathematics question paper 2026 on the UK 10th question paper page. You can download the question paper by clicking on the link provided here.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
The most sought job profile after graduating from a linguistic course is that of a content writer or a translator. Majority of the graduates opt for these job profiles after completing their graduation. The average salary for these profiles usually ranges between INR 3-5 LPA.
New Question
10 months agoContributor-Level 10
While all chapters are important, students should focus more on frequently asked topics like Electricity, Acids-Bases-Salts, Life Processes, and Magnetic Effects of Current. These chapters often carry more weight in exams. Go through past 5-year question papers to spot high-scoring areas. Don't skip small chapters, as they may carry easy marks. A balanced approach to all topics is best for scoring well.
New Question
10 months agoContributor-Level 10
At the time of preparing the JIMS Greater Noida BCom cutoff, there are several factors that can impact the increase and decrease of accepted ranks. Likewise, the authorities will consider examination and admission-related factors. The listed factors will be checked and considered by the authorities while determining the cutoff of JIMS Greater Noida BCom cutoff:
- Previous years' cutoff trends
- Difficulty level of exam
- Total number of qualified candidates
- Total number of candidates applying for a specific course or institute
- Number of seats available for admission, etc.
New Question
10 months agoContributor-Level 10
The University of Otago is highly regarded for offering the highest placement rates across the nation. University of Otago placement rate stands at is 95%, and the graduates were employed within 6 months of graduation. This figure states that out of 100 students, around 95 students secured employment. Otago University graduates are always in high demand all over the world. Moreover, the average salary of University of Otago graduates is NZD 87,400 (INR 44.79 LPA), per year. The university graduates work for top sectors such as Information Technology, Marketing, Media and Communication, Legal, Human Resources, Entrepreneurship, and more
New Question
10 months agoNew Question
10 months agoContributor-Level 10
There are approximately 70 colleges in India known for their specialised Linguistic programs. Out of these, around 53 are government entities and 7 are private institutions. Aspiring students can opt for either of the two colleges based on factors such as budget, distance, location, etc.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Ambident nucleophiles are groups that include two nucleophilic centres, such as cyanide and nitrile. In the cyanide group, linkage can occur from both the carbon and nitrogen ends. The carbon end functions as a greater nucleophile in an aqueous media because it leads to the creation of a C-C sbond, which is stronger than a N-C bond in the identical molecule.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
In the presence of ZnCl2, ethanol can be treated with HCl to produce chloroethane. Iodoethane is formed when chloroethane reacts with NaI.
C2H5OH+HCl→ZnCl2? C2H5Cl→ NaI ? C2H5I
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts