Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
RBSE sometimes updates its class 10 syllabus to match current educational standards. For the academic year 2025-26, the board has not made any changes in the syllabus. However, students should always check the official RBSE website or ask their teachers to confirm the latest RBSE 10th syllabus 2025-26. Even small changes, like a few removed or added topics, can impact exam preparation. Knowing these changes early helps avoid wasting time on outdated topics. Always study from updated textbooks and official materials.
New Question
10 months agoContributor-Level 10
The University of Otago is one of the most prominent universities in the New Zealand. The University of Otago placement rate is 95%, and the students are employed within 6 months of graduation. This figure states that out of every 100 students, around 95 students secured employment. The University of Otago placement rate attracts international students the most. Otago University graduates work in top companies all over the world such as ANZ, Deloitte, Fonterra, University of Canterbury, Victoria University of Wellington, ASB Bank, and others. The university is known for having the highest placement rate all over the world. The Universi
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
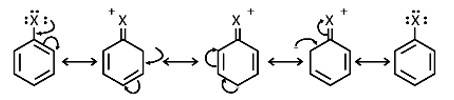
Resonance effect: Because the C – X bond has the characteristics of a partial double bond, it is extremely difficult to break.
(i) The C - X bond in haloarenes is extremely less reactive to nucleophilic groups
(ii) The C atom linked to the halogen is sp2 hybridised in the C – X bond. Carbon that has been sp2 hybridised with a higher s-character is more electronegative in nature and can retain the electron pair of the C - X bond more tightly than carbon that has been sp3 hybridised with a lower s -character in haloalkanes.
New Question
10 months agoContributor-Level 10
Students need not worry about the accuracy of the UK 10th result released online. UK 10th result 2026 will be released after various checking process. There is very less chance for the UK 10th result released online to be incorrect. Students are advised to collect the mark list from the school after the UK 10th result is announced.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The reaction of HCl and isobutylene is given as:
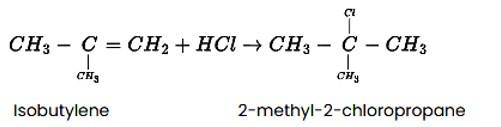
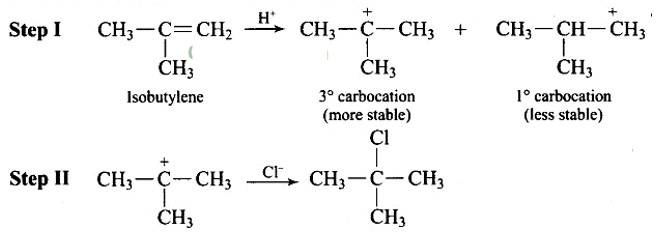
Steps in reaction involves are:
New Question
10 months agoContributor-Level 10
Linguistics is a vast field covering many topics. The fundamentals of phonetics and phonology are also included in his study. students analyze speech sounds and the organisation of language. There are many reasons why students should pursue this course. Some of these reasons are:
Linguistics encompasses a broad range of topics that delve into the structure, function, and expansion of human communication. The linguistics courses provide a thorough investigation of the Science of language.
Linguistics is a comprehensive field that provides an intellectually stimulating and useful way to explore the complexities of human language. Lin
New Question
10 months agoContributor-Level 10
Follow the tips below to manage stress level during UK 10th board exams.
- Prepare a time table
- Study daily as per syllabus
- Practice sample papers and previous question papers.
- Revise important notes
- Take enough breaks
- Stay healthy
- Practice yoga
New Question
10 months agoContributor-Level 10
A Linguistics course can be pursued at the degree, certificate, and Diploma levels. The course duration of a linguistics degree depends on the level at which it is being pursued. Some of the popular Linguistics courses and their course duration have been mentioned in the table below:
Linguistics Courses | Course Duration |
BA in Linguistics | 3 years |
MA in Linguistics | 2 years |
PhD in Linguistics | 2 years |
Diploma in Linguistics | 1 year |
PG Diploma in Linguistics | 1 year |
Certificate in Linguistics | 6 months to 1 year |
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
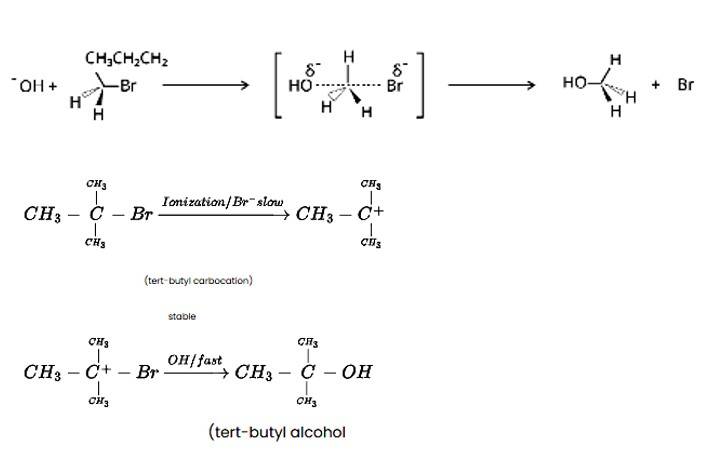
Tert-butyl bromide is substituted via the SN1 process because it may produce a stable carbocation in the first step after the halide group is cleaved. The nucleophile OH - interacts with the carbocation next. The primary halide n-butylbromide, on the other hand, is unable to create a stable carbocation, thus it undergoes the SN2 process, which is a one-step substitution involving OH - attack and concomitant X - leaving to generate n-butyl alcohol.
New Question
10 months agoContributor-Level 10
Once you finish learning according to UK 10th syllabus, you can start solving the UK 10th sample papers and previous year question papers. You can solve the question papers and understand your weaknesses. Thus you can concentrate more on the topics on which you are weak.
New Question
10 months agoContributor-Level 10
There are different factors that determine the seat intake of the BBA at SICSR, Pune. Additionally, there are more than 100 seats for the BBA programme provided by the institute. The following is a list of the main factors that candidates can consider for the SICSR Pune BBA seat intake:
- Availability of Seats
- Number of students who applied
- Number of shortlisted students
- Academic record
- Scores obtained in the entrance exam
- Performance in the PI and GE round
New Question
10 months agoContributor-Level 10
The University of Otago is among the top universities for higher education in USA. The University of Otago placement rate is 95%, and the students secured employment within 6 months of graduation. This figure shows that out of 100 students, 95 students secured employment. Otago University graduates work for top companies such as EY, Westpac New Zealand, Air New Zealand, University of Melbourne, New Zealand Rugby, Microsoft, etc. Moreover, Otago University graduates work for top sectors such Operations, Education, Research, Sales, Finance, Information Technology, etc. Otago University placement rate varies from one programme to anothe
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
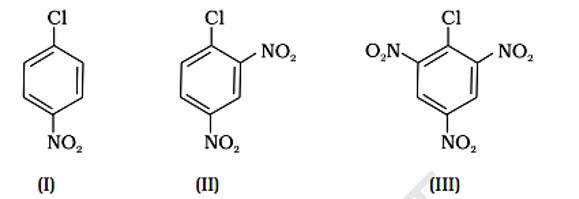
Because of resonance stabilisation, aryl halides are less reactive to nucleophilic substitution. The inclusion of a -NO2, an electron-withdrawing group in the ortho or para position, enhances the aryl halide's sensitivity to substitution. The more electron withdrawing groups there are in the aryl halide, the more reactive it is. The decreasing sequence of reactivity, according to this theory, is III > II > I.
New Question
10 months agoContributor-Level 10
Students are advised to correct the errors even if it as a minor mistake. They must inform the school authorities about the error in the UK 10th hall ticket. School authorities will help you in getting the new hall ticket without any mistakes.
New Question
10 months agoContributor-Level 9
Along with the global rankings and reputation, the University of Alabama at Birmingham is popular and best for various reasons. Some of the crucial reasons to attend the University of Alabama at Birmingham are discussed below:
- Excellent programs: The university offers diverse popular programs in fields like Health Professions, Business, Management, Marketing, Biological and Biomedical Science, Psychology, and more. The university also ranked #125 for providing the best undergraduate engineering programs
- Research opportunities: Students willing to grab research opportunities must come to UAB, as it is the largest research institution in t
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Nucleophilic substitution and elimination (β-elimination) reactions are both possible with alkyl halides.
Although, with the appropriate reaction conditions and reagent selection, a specific product can be produced. In most cases, the elimination reaction is best suited to strong and bigger bases, as well as high temperatures. The substitution reaction, on the other hand, is best for weaker and smaller bases at lower temperatures.
CH3CH2Br+alc.KOH![]() KCH2=CH2 + HBr
KCH2=CH2 + HBr
CH3CH2Br + aqKOH→CH3CH2OH + KBr
New Question
10 months agoContributor-Level 10
The students should divide the topics and set daily study hours for each subject. They can give more sessions to difficult subjects and less time to easy papers.
New Question
10 months agoContributor-Level 9
International applicants must complete their high school degree and a bachelor's degree from a recognised university or equivalent, and submit the required admission documents to get into the university on time. The admission requirements to get into the undergraduate and graduate programs at the University of Alabama at Birmingham are given below:
Undergraduate requirements:
- High school degree from a recognised institute
- Official high school record
- LOR
- Essay
- English language requirements
- Additional test scores of the ACT/SAT are optional
Graduate requirements:
- Undergraduate GPA of 3.0 on a 4.0 scale
- Academic transcripts
- GMAT/GRE score (optional)
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The IUPAC names for DDT and benzene hexachloride are 2, 2-bis (4-chlorophenyl)-1, 1-trichloroethane and 1, 2, 3, 4, 5, 6-hexachlorocyclohexane, respectively.
They are poisonous and non-bio digestible at the same time. They are fat soluble, and their concentration in the food chain continues to rise. As a result, they are prohibited in India and other nations.
New Question
10 months agoContributor-Level 10
It is mandatory to have 75% attendance to get hall ticket for the board examinations. However, you may submit a medical certificate in case you were not well or due to any other medical condition to get a waiver.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts