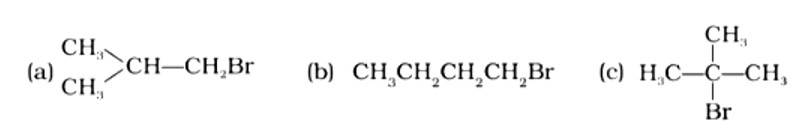Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
The admissions at Ambedkar University Delhi, Karampura are held on the basis of entrance-test basis. As such, there is no cutoff criteria for a specific course at AUD Karampura. Students seeking enrollment at the university can register for the Common University Entrance Test (CUET) and specify their choice as AUD Karampura. The admissions are conducted on the basis of the combinations of sections mapped to the course-choice in the CUET exam.
New Question
10 months agoContributor-Level 10
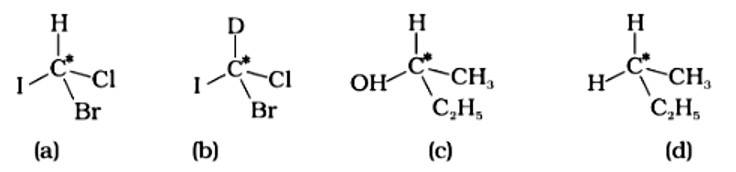
This is a multiple choice answer as classified in NCERT Exemplar
The correct answer is (ii).The mirror copies of the molecules (a), (b), and (c) are non-superimposable. Because all four species linked to the carbon atom are identical, molecule (d) cannot have an asymmetric carbon atom, thus the mirror image of (d) when rotated 180? is superimposable on the original picture.
New Question
10 months agoContributor-Level 10
To get admission at Ganga Institute of Technology and Management, aspiring candidates need to ensure that they satisfy the admission criteria of the desired course. Before starting the admission process for any of the courses, candidates need to check the admission criteria for the same. Eligible students can then apply online. The institute may ask students to appear for instituteThose who get selected need to pay the admission fees to secure their spots at the institute.
New Question
10 months agoContributor-Level 10
The various centers of study at AUD Karampura provide specialised learning experiences to students. The following are the Ambedkar University Delhi, Karampura centers:
School of Public Policy and Governance | School of Human Studies |
School of Heritage Research and Management | School of Letters |
School of Education Studies | School of Management |
School of Design | School of Undergraduate Studies |
School of Culture and Creative Expressions | School of Legal and Socio-Political Studies |
School of Human Ecology | School of Global Affairs |
School of Development Studies | School of Vocational Studies |
School of Liberal Studies |
|
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (iii).
Isomeric alkyl halides' boiling points are related to their branching, with a reduction in B.P. as branching increases. As a result, the boiling point of the tertiary isomer is the lowest, while that of the primary isomer is the highest. Thus, the order of lowering boiling points is (b) > (a) > (c).
New Question
10 months agoContributor-Level 10
DR BR Ambedkar University Delhi, Karampura is currently accepting the Common University Entrance Test (CUET) for admissions to its various courses. Students can visit the official website to learn more regarding their choice of programs and the specific eligibility criteria that is prescribed for the same. Students seeking admissions can register for the CUET test and signify their preferences for AUD Karampura.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (i).
Alkyl halides have a higher density than water. Their densities are determined by the masses of the halogen atoms, the number of halogen atoms, and the number of carbon atoms. Simply expressed, Br has an atomic mass of 79 while Cl has an atomic mass of 35. (d) will be the heaviest of the molecules, followed by (c), (b), and (a). Because density is exactly proportional to mass, the order of decreasing densities will be the same. The right answer is (i).
New Question
10 months agoNew Question
10 months agoContributor-Level 10
Students seeking to enroll into AUD Karampura can choose to avail the accommodation benefit provided by the university. The hostel fees at Ambedkar University Delhi, Karampura is INR 51,000, which includes the one-time security fees of INR 10,000. Students choosing to receive the hostel mess facility would be required to pay an amount of 19,200 per semester (plus INR 1,000 maintenance fee).
New Question
10 months agoContributor-Level 10
Dr BR Ambedkar University is located in the well-connected and serene neighborhood of Karampura, Moti Nagar in West Delhi. From the Indira Gandhi International (IGI) Airport, students can take a drive of around 30 minutes to reach the university campus.
From the New Delhi Railway Station (NDLS), students can arrive at the AUD Karampura campus by taking a 30-minute drive. The nearest metro stations to the university campus are the Kirti Nagar and Moti Nagar metro stations on the Delhi Metro Blue Line.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
The correct Answer is Option (i).
Chlorination in the presence of UV light can be used to produce alkyl chlorides from alkanes. Under the action of UV radiation, the chlorine molecule generates free radicals, which react with alkanes to create a mixture of isomeric mono- and poly haloalkanes.
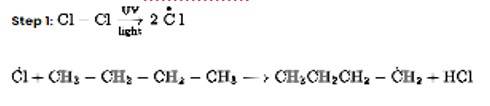
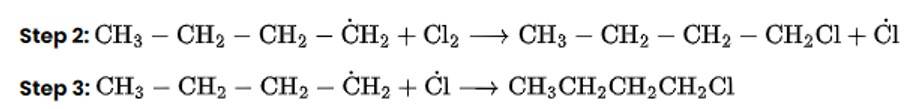
New Question
10 months agoContributor-Level 10
DR BR Ambedkar University Delhi, Karampura is one of the popular and consistently recognised universities in the Delhi region. Students at the AUD Karampur are taught according to a strong humanistic and socially-driven curriculum.
This is designed to instill within students values consistent with the namesake of the university, Dr BR Ambedkar. Students get the opportunity to grow holistically with plenty of courses and specializations to choose from, with a heavy focus on the Humanities.
New Question
10 months agoContributor-Level 10
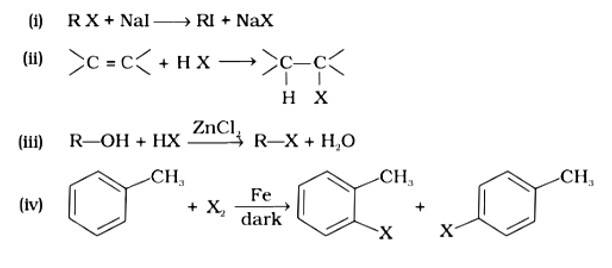
This is a multiple choice answer as classified in NCERT Exemplar
Option (i)
The correct answer is option (i). Option (i) is an example of a Finklestein reaction, which is a halogen exchange reaction that produces alkyl iodide by treating alkyl halides with NaI in dry acetone. Option (ii) is an addition reaction in which an alkene is transformed to the equivalent alkyl halide. Option (iii) is a substitution reaction, whereas option (iv) is an electrophilic substitution reaction.
New Question
10 months agoContributor-Level 10
Hall ticket for the UK 10th board exam is issued by the UBSE to which you are enrolled via your school. So your school cannot deny you the admit card even if you have failed to pass in their pre-boards exam. If they still goes on to deny you the hall ticket you can straight away write it to board asking them to issue your 10th hall ticket in duplicate as your school has failed to give you.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
No, it is not mandatory to pass the model exam to get the UK 10th hall ticket. Model exams are meant for students to be familiar with the board exam. Clearing the model exams is not a criteria for obtaining the UK 10th hall ticket.
New Question
10 months agoContributor-Level 10
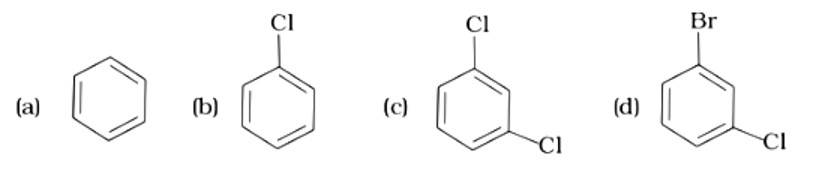
This is a multiple choice answer as classified in NCERT Exemplar
The Correct Answer is Option (ii). Toluene is an aromatic hydrocarbon (C6H5-CH3) that may be treated with halogens and utilised in an electrophilic substitution process to create aryl halides in the presence of the Lewis acid catalyst iron (III) chloride. In the absence of light, the reaction is carried out using chlorine or bromine, and the products are o- and p- haloarenes.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
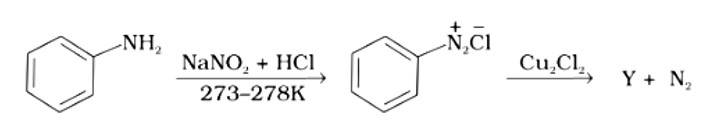
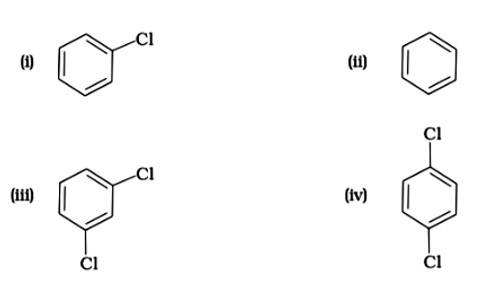
The correct answer is (i). Sandmeyer's reaction can be used to synthesise haloarenes from amines. A primary aromatic amine that has been dissolved or suspended in cold aqueous mineral acid is treated with sodium nitrite to create a diazonium salt in this procedure. When this freshly produced salt is combined with cuprous chloride, the diazonium group is replaced with - Cl, resulting in aryl chloride. Option I is the chemical Y, which is an aryl chloride.
New Question
10 months agoContributor-Level 10
For 2025, GATE CCMT has been released across different categories under the AI quota. For the General AI category students, the 2025 Round 1 cutoff score for CSE admission stood at 407. Please note that the cutoff scores vary for different rounds, specialisations and categories. Candidates seeking admission can refer to the table below to view the Guru Ghasidas Vishwavidyalaya CSE cutoff 2025 via GATE CCMT across various category:
| Category | GATE CCMT Round 1 Cutoff 2025 |
|---|---|
| General | 407 |
| OBC | 367 |
| SC | 331 |
| ST | 264 |
| EWS | 354 |
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
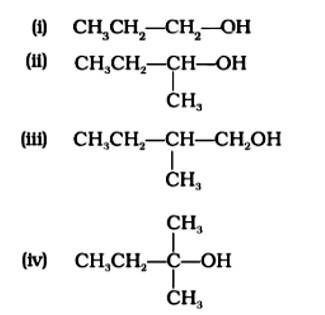
The Correct Answer is Option (iv) Because the tertiary carbocation is the most stable, it interacts the most with concentrated HCl. As a result, for tertiary alcohol, room temperature is sufficient for the reaction. However, for primary and secondary alcohols, the presence of a catalyst (ZnCl2) is required.
As a result, option (iv) is accurate.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts