Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 6
Special ro 2nd OJEE registration form will likely be released in the second week of June 2026. The application forms will be available at ojee.nic.in after the first OJEE exam has been conducted.
New Question
10 months agoContributor-Level 8
Backstage Pass Institute of Gaming and Technology offers courses at the UG and PG levels in full-time mode for the durations varying from 2 years to 4 years. The Institute offers the respective courses on the basis of merit and institute level entrance exams. Furthermore, the fee structure of the course includes tuition fees varying from INR 2.25 Lacs to INR 6 Lakh. Considering the placement facilities, course curriculum, and exposure the Institute offers, the fees structure is self explanatory. However, in comparison to other private institutes, Backstage Pass Institute of Gaming and Technology fees will lie under the category of expe
New Question
10 months agoContributor-Level 10
Yes, Ranchi University does release a merit list for UG admissions. Once the application window is closed, the admission committee reviews all the applications received and prepares a merit list based on CUET UG results. Applicants without a CUET UG score are reviewed based on Class 12 scores. Once the list is produced, it is released on the official website of the Chancellor's Portal.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(II) Substitution reaction
Alkyl halides undergo substitution reactions to form corresponding alcohol.The halide ion, X- is substituted by OH- ion to form alcohol by nucleophilic substitution reaction.
R-X → R-OH
New Question
10 months agoContributor-Level 6
BEST Innovation University powered by Sunstone offers admission to various courses based on merit scores and in-house tests. The university conducts the Sunstone Screening Test and Interview for the shortlisted students. Students are required to submit the necessary documents. The minimum eligibility requires the candidates to pass Class 12 from a recognised board with 50% aggregate marks.
New Question
10 months agoContributor-Level 10
UPES School of Business Studies offers full-time BCom and BCom (Hons) at the UG level. Admission to these courses is subject to the fulfilment of the eligibility criteria specified by the university. As per the specified eligibility criteria, candidates seeking admission to BCom at UPES School of Business Studies are required to have passed Class 10 and Class 12 with a minimum aggregate of 50%. Candidates from all streams (Science/ Commerce/ Arts) are eligible to apply. Hence, Mathematics is not compulsory for BCom admission at UPES School of Business.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(III) Treatment with pyridinium chlorochromate
The pyridinium chlorochromate is a complex of chromium trioxide with pyridine. This reagent is preferred for mild oxidation to form aldehydes and hence the oxidation to carboxylic acid is prevented.
Primary alcohols are oxidized to form aldehyde, whereas secondary and tertiary alcohols are oxidized to form ketones. The conversion of a hydroxyl group to aldehyde can be done by the removal of hydrogen atoms (Oxidation), looking at the following options; pyridinium chlorochromate and potassium dichromate are both oxidizing a
New Question
10 months agoContributor-Level 10
The MD specialisations offered by PK DAS Institute of Medical Sciences - Hospital and Medical College are as follows:
New Question
10 months agoContributor-Level 10
The Ranchi University 2025 admission process for UG courses is still ongoing. However, the application portal has been closed. The last date to apply for UG courses offered by the university was 19 Jun, 2025. Students who require an extension of the application date are advised to reach out to the university's admission office.
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(III) 3° > 2° > 1°
Alcohols are classified as primary, secondary and tertiary by lucas reagent which is a mixture of concentrated HCl and dry anhydrous ZnCl2. The carbocation is formed as the intermediate during the lucas test of alcohol.More the stability of intermediate carbocation, more will be the reactivity of alcohol.
Tertiary carbocation is most stable then the secondary alcohol is stable due to hyperconjugation and the primary alcohol is the least stable.
Stability of intermediate carbocation:
3° > 2° > 1°
Reactivity of a
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(I) The alcohol with molecular formula C4H10O is butanol has 4 isomers, these are:
(a) CH3-CH2CH2-CH2OH (Butane-1-ol)
(b) CH3-C*H-CH2-CH3 (Butane-2-ol)
|
OH
(c) CH3-CH-CH2-OH (2-methylpropane-1-ol)
|
CH3
New Question
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(IV) Benzyl alcohol
The chlorine atom is substituted by the hydrogen atom of methyl hydrocarbon present in toluene to form benzyl chloride on monochlorination of toluene in presence of sunlight by free radical pathway.
C6H5CH3 + Cl2 ![]() C6H5CH2Cl
C6H5CH2Cl
Toluene Benzyl Chloride
Benzyl chloride (monochlorinated toluene product) on hydrolysis with aq. NaOH yields benzyl alcohol by substituting the chloride ion by OH- ion by nucleophilic substitution reaction.
C6H5CH2Cl ![]() C6H5CH2OH
C6H5CH2OH
Benzyl &
New Question
10 months agoContributor-Level 10
Ranchi University accepts CUET UG scores for admission to its BCom courses. This implies that students willing to take admission can take the CUET UG exam. For the session 2025, the exam was held from 26 May 2025 to 3 Jun 2025. Moreover, the results are expected to be announced in the last week of June 2025.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Increasing order of acidity is: ethanol < water < phenol.
This is because the phenoxide ion obtained after the removal of H+ is resonance stabilised, while the ethoxide ion obtained after the removal of H+ is destabilised by +1 effect of ethyl group. Thus phenol is a stronger acid than ethanol.
Now, ethanol is a weaker acid than water because the electron releasing ethyl group increases the ethanol density on oxygen and consequently the proton will not be released easily. There is no such effect is water.
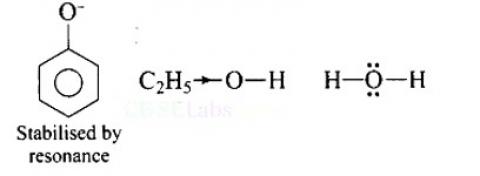
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
This can be explained as under:
(i) In phenol, the conjugation of unshared electron pairs over oxygen with aromatic ring results in partial double bond character in C – O bond.
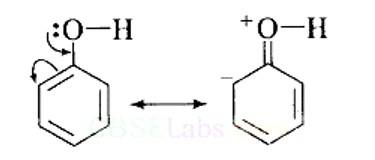
In methanol, no such conjugation (resonance) is possible.
(ii) In phenol, oxygen is attached to sp2 hybridised carbon while in methanol, oxygen attached to sp2 hybridised carbon. An sp2 hybridised carbon is more electronegative (because of greater 5-character) than sp3 hybridised carbon atom. Therefore, the bond between oxygen and sp2 hybridised carbon is more stable than the bond between o
New Question
10 months agoNew Question
10 months agoContributor-Level 8
Admissions to Backstage Pass Institute of Gaming and Technology are now open. Interested candidates must visit the official website of the Institute and fill out the application form. After a successful application submission, candidates will be shortlisted based on the provided eligibility criteria of Class 12 or graduation aggregate, depending on the UG and PG level programme, candidates opt for. For final admission, candidates must pay the fees and confirm the seat.
New Question
10 months agoContributor-Level 10
The application steps that a candidate must take are as follows:
Step 1: Visit the official website of the college.
Step 2: Go to the applications section and fill out the form
Step 3: Upload the required documents.
Step 4: Fill out the form for the required entrance exam as well.
Candidates who follow the above mentioned steps can complete the application process.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Due to the presence of intermolecular H- bonds in alcohol the energy required to break H – bonds is more than the energy required to break simple dipole bonds present in ethers . Thus, the boiling point of alcohol is higher than the boiling point of ethers.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
