Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The electron withdrawing group (-NO2), withdraws electrons and disperses the negative charge. Therefore, -NO2 group stabilizes the phenoxide ion. Hence p-nitrophenol is more acidic than phenol.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The occurrence of intermolecular hydrogen bonds due to the H- bonds in alcohol compounds play a major role . With the increasing number of alkyl groups the molecular mass increases due to which the polar nature of these compounds get suppressed . Hence, the solubility factor of alcohols is indirectly proportional to the molecular mass.
New Question
10 months agoContributor-Level 8
Admission to these courses is both merit & entrance-based at Backstage Pass Institute of Gaming and Technology. The Institute conducts an entrance exam for admission to UG courses. Backstage Pass Institute of Gaming and Technology application process is conducted in online mode only. Therefore, candidates must fill out the online application form and submit it. After a successful submission of the application form, candidates will be shortlisted based on the merit results. For final admission, candidates must pay the fees and confirm the seat.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The bond angle C—O—H in alcohols is less than tetrahedral angle because of the repulsion between the lone pairs present on the oxygen atom, which pushes the bond C—O—H closer. Therefore, C—O—H bond angle in alcohols is slightly less than the tetrahedral angle.
In ethers, the lone pairs on oxygen atoms are also present but the two bulky alkyl groups also have repulsive force which increases the C—O—C bond angle. Therefore, the C—O—C bond angle in ether is slightly greater than tetrahedral ang
New Question
10 months agoContributor-Level 8
Backstage Pass Institute of Gaming and Technology offers BSc as a UG level programme for the duration of 4 years in full-time mode. The Institute offers admission to the respective course on the basis of merit and valid scores of college level entrance exam. Candidates must for the apply to the Backstage Pass Institute of Gaming and Technology by visiting the official website of the Institute. The Institute offers BSc with a total tuition fees worth INR 6 Lakh.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Ethers are prepared by Williamson synthesis by the reaction of alkyl halide with the stadium alkoxide. But this is not possible in case of di-tert-butyl ether. In case of preparation of di-tert-butyl ether, tert-butyl halide must react with sodium tert-butoxide. Alkoxides are nucleophiles but they are strong bases as well, due to which elimination is favoured instead of substitution and leads to the formation of 2-methylprop-1-ene.
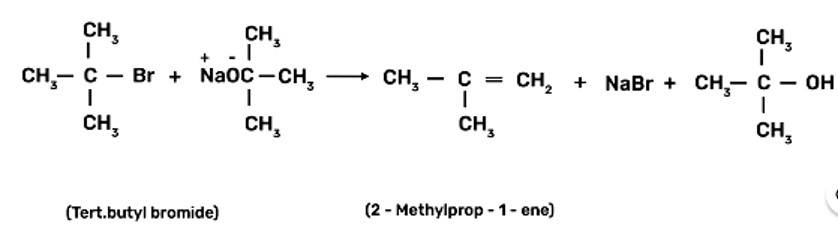
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
In phenol, the electron pairs on oxygen atom of -OH group are in conjugation (or resonance) with phenyl ring which decreases the polarity of the C-OH bond whereas in methanol the polarity of C-OH bond is more due to electron releasing group of -CH3.
New Question
10 months agoContributor-Level 10
For BTech admission at North East Frontier Technical University (NEFTU), students must submit: mark sheets and certificates, address proof, a government-issued ID, affidavit form (available on the NEFTU website), migration certificate from previous board/university, and five passport-size photographs. All documents must be accurate and in the format specified by NEFTU.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Phenoxide ion is more reactive than phenol to electrophilic aromatic substitution reaction due to the more in electron density of phenoxide ion and hence, phenoxide ion easily undergoes the electrophilic substitution reaction than phenol by the weak CO2 electrophile.
New Question
10 months agoContributor-Level 10
The final step for admission into the BTech programme at North East Frontier Technical University (NEFTU) is payment of the tuition fee. The total fee is approximately INR 3.2 Lakh. Candidates must complete this step to confirm their seat. Scholarships are also available for eligible students enrolled in the BTech course.
New Question
10 months agoContributor-Level 8
Admission to these courses is both merit & entrance-based at Backstage Pass Institute of Gaming and Technology. The Institute considers college level entrance exam for the admission, along with merit. Candidates must meet the eligibility criteria to secure admission at the Institute. Furthermore, candidates must secure a minimum aggregate in Class 12 and graduation, depending on the course opted for.
New Question
10 months agoContributor-Level 10
The selection process for the BTech programme at North East Frontier Technical University (NEFTU) is based on performance in national-level exams such as JEE or through the university's own entrance test and/or personal interview. Candidates must also meet the basic eligibility of passing Class 12 with a Science stream.
New Question
10 months agoContributor-Level 10
Ranchi University BCom eligibility criteria state the conditions to be fulfilled by candidates seeking admission. Only those candidates who fulfil the requirements are eligible to apply for the preferred course. Tabulated below are the eligibility conditions to be met:
Course | Eligibility Criteria |
|---|---|
BCom | Students willing to apply for BCom courses offered by Ranchi University must have passed Class 12 from a recognised board of India |
New Question
10 months agoContributor-Level 10
To apply for the BTech programme at North East Frontier Technical University (NEFTU), visit the official website and click on 'Apply Now.' Select the BTech course, fill in the required academic and personal details, upload necessary documents, pay the application fee, and submit the completed form online to begin the admission process.
New Question
10 months agoContributor-Level 6
BEST Innovation University powered by Sunstone has a Placement Opportunity Program (POP) for its students. The programme assures placement opportunities or a refund of first-year tuition fee if placements don't meet the commitment. In 2025, the highest and average package offered was INR 26 LPA and INR 4.6 LPA. Additionally, the top recruiters who regularly visits the campus Nestle, Amul, Paytm, SBI General, and Axis Bank, among others.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The nitration of aromatic compounds occurs by using HSO4 and HNO3 which leads to the formation of NO2+ (nitronium ion) electrophile.
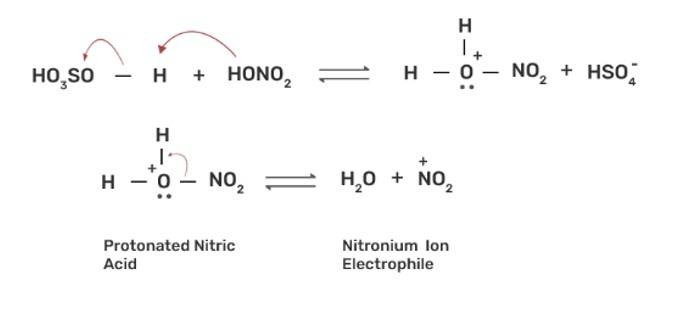
Due to the formation of NO2+ electrophile, we can say that nitration is an example of aromatic electrophilic substitution. The electron releasing group on the benzene ring increases the rate of nitration and vice-versa.
Phenol is more easily nitrated than benzene as the hydroxyl (-OH) group on the benzene ring is an electron releasing group which increases the electron density at the ortho and para position due to +R effect of -
New Question
10 months agoContributor-Level 6
You can share solved paper with teacher, who will help know if the answers are correct and can fetch one full marks.
It is always suggested to make notes of all important points while studying. Try getting the concepts clear and writ them down in your own words in exam.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
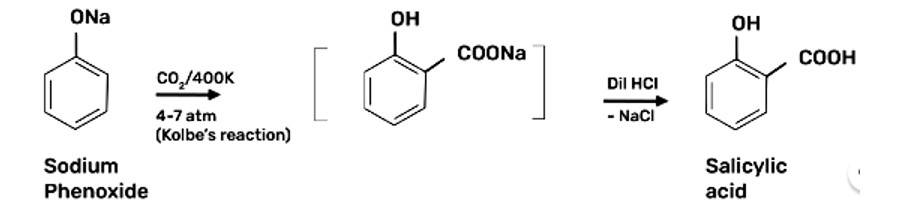
Step 1: Addition of NaOH to obtain sodium phenoxide
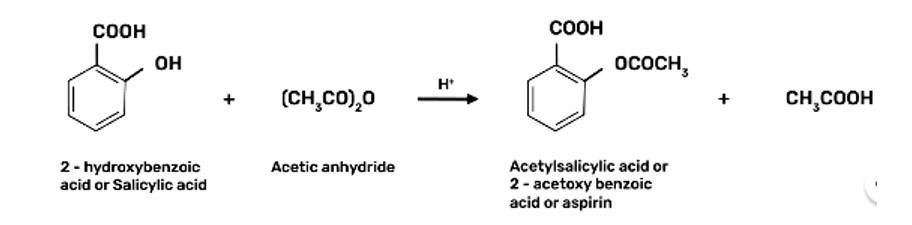
Step 2: Sodium Phenoxide undergoes Kolbe’s reaction at high temperature and pressure in presence of carbon dioxide (CO2) gas. The product is further hydrolysed to obtain salicylic acid.
Step 3: Acetylation of salicylic acid occur when it is treated with acetic anhydride

Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
