Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The reaction of alcohol with the conc. HCl and ZnCl2 (Lucas reagent) leads to the formation of a carbocation through SN1 mechanism. The more stable the carbocation, the faster the reaction will be.
Tertiary carbocation is the most stable due to hyperconjugation and inductive effect then is the secondary alcohol and the primary alcohol is the least stable.
Stability of carbocation:
Tertiary carbocation > Secondary carbocation > Primary carbocation
According to stability of carbocation, the reactivity order of the alcohol is:
Tertiary alcohol > Secondary alc
New Question
10 months agoContributor-Level 6
You will get medical college according to you marks and college cutoff. You should make a list of colleges according to your marks. Then participate in counselling for admission process.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
In O? C? O, the dipole moment is zero, hence non-polar while in R—O—R the dipole moment is non-zero due to which it is polar.
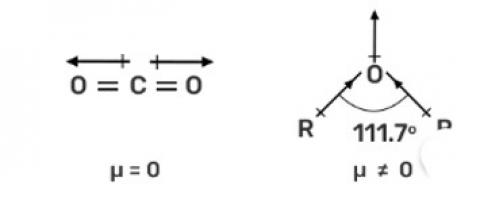
The CO2 is a linear molecule and the dipole moment is equal and in the opposite direction due to which the dipole moment is zero. While in R—O—R, has bent structure and lone pair of electrons due to which the net dipole moment is nonzero and hence, it is a polar molecule.
The CO2 is a linear molecule and the dipole moment is equal and in the opposite direction due to which the dipole moment is zero. While in R&md
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Step 1: Protonation of alkene due to the presence of double bond which attacks the H3O+ ion and forms carbocation.
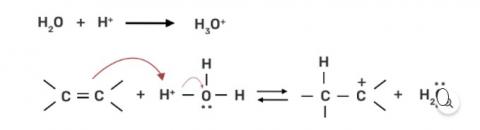
Step 2: Water molecule act as nucleophile and attack the carbocation.

Step 3: Deprotonation occur to get the alcohol and hydronium ion forms
New Question
10 months agoContributor-Level 10
Yes, students can do BCom from Ranchi University. This course is only offered via colleges affiliated with the university and not the main campus. The key highlights of this course are as follows:
Particulars | Statistics |
|---|---|
Duration | Three/four years |
Mode | Full-time |
Eligibility | Passed Class 12 |
Selection Criteria | CUET UG/Class 12 |
Note: The above information is taken from official sources. However, it is subject to change.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
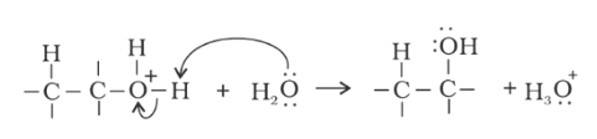
The C-OH bond in phenols has double bond character due to resonance of electron pairs of oxygen atom with the pi electrons of phenyl ring, which makes the C-OH bond strong and hence, the nucleophilic substitution of a nucleophile with the -OH group of alcohol is not possible as the C-OH does not break easily.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The electron pairs of oxygen atom of hydroxyl (-OH) group in phenols are in conjugation with the pi electrons of phenyl ring and hence, the bond C-OH bond has double bond character due to which OH group in phenols more strongly held as compared to OH group in alcohols.
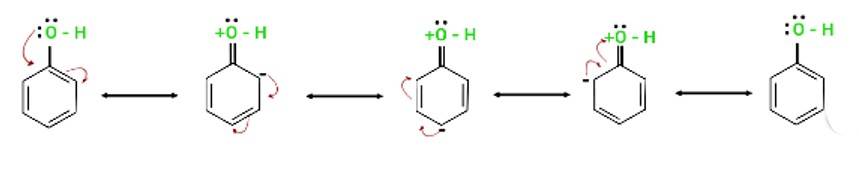
Resonance in phenol
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The alcohol with molecular formula C4H10O is butanol has 4 isomers, these are:
(a) CH3-CH2CH2-CH2OH (Butane-1-ol)
(b) CH3-C*H-CH2-CH3 (Butane-2-ol)
|
OH
(c) CH3-CH-CH2-OH (2-methylpropane-1-ol)
|
CH3
CH3
&nbs
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The propan-2-one can be converted into tert-butyl alcohol by using CH3MgBr grignard reagent to form the additional product tert-butyl alcohol.
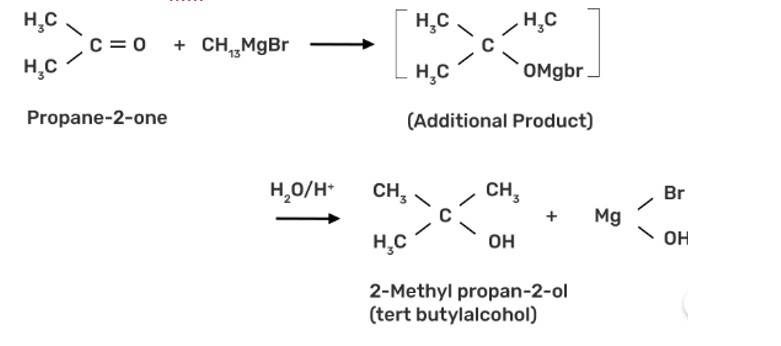
New Question
10 months agoNew Question
10 months agoContributor-Level 10
The documents required to fill out the application form for PK DAS Institute of Medical Sciences - Hospital and Medical College are as follows:
Copies of the scanned educational credentials (e.g., Class 10 and Class 12 marksheets)
signature and a scanned image in the appropriate format
These materials must be uploaded in the designated format. Additionally, candidates cannot submit their application for admission before the required papers have been submitted.
New Question
10 months agoContributor-Level 10
An important date related to BTech admission at NEFTU is the release of the JEE Main Session 2 result, which plays a key role in the selection process. Students who appeared for the exam should keep track of result updates and follow the JoSAA counselling process closely.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The enzyme invertase, which is present in yeast, is used to convert sucrose to glucose and fructose. The glucose and fructose are further converted to ethanol by zymase enzyme (also present in yeast).
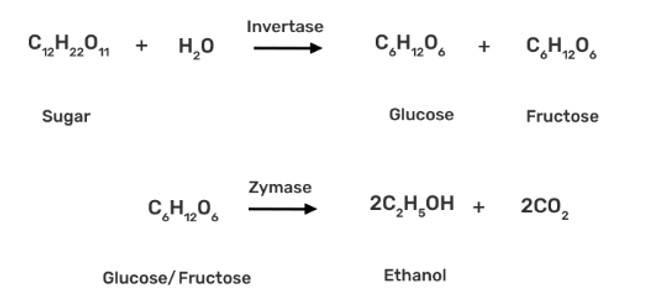
New Question
10 months agoContributor-Level 10
As of now, the BTech admission process at North East Frontier Technical University (NEFTU) is underway. Candidates should stay updated through the official website for counselling schedules, seat allotments, and other related announcements to avoid missing any important deadlines.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The decreasing order of acidity:
H2O > ROH > HC ≡ CH
The more the stability of the conjugate base of the given compounds, the more acidity will be.
? OH > ? O-R > ? C ≡ CH
The negative charge on the electronegative oxygen atom is more stable than carbon atom. The + I effect of the alkyl group does not stabilize the negative charge on the electronegative oxygen atom, hence less stable than ? OH.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
When benzene diazonium chloride is heated with water, it forms phenol along with nitrogen and hydrochloric acid as by-products as shown below:
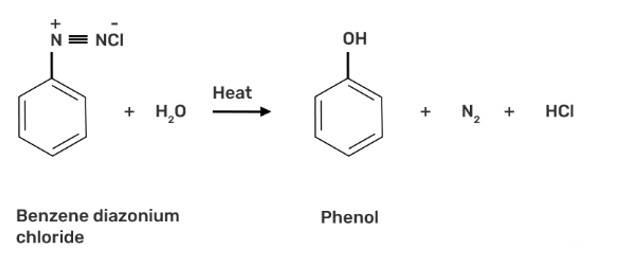
New Question
10 months agoNew Question
10 months agoContributor-Level 10
If you're still uncertain if Cath Lab Technician course is for you then use these methods:
- Go to a hospital: Be a spectator to see Cath Lab Technicians in action during the actual procedures.
- Watch medical videos: There are numerous cath lab operations online, view already completed cath lab cases to understand the daily work involved.
- Interview a technician: Have chat with either current students or a working technician to better understand the profession.
- Attend a short workshop: Sign up for a short course in health care or cardiac care before making a full commitment.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
