Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The decreasing order of reactivity of alcohol with sodium metal:
Tertiary Alcohols > Secondary Alcohols > Primary Alcohols
When alcohol reacts with active metals e.g. Na. K etc., the O-H bond of alcohols breaks to form the corresponding alkoxide. The alkyl group is electron donating and has +I effect due to which the O-H bond becomes strong and hence, the reactivity of alcohol decreases.
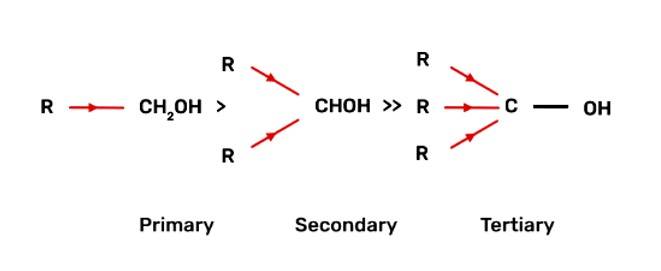
Fig: +I effect of alkyl group of alcohol
New Question
10 months agoContributor-Level 10
Cath Lab Technician course provides you with practice and clinical skills like:
- Operating cath lab imaging equipment and monitoring vital signs
- Assisting with procedures (angioplasty and pacemaker insertions)
- Knowing cardiac anatomy and reading diagnostics
- Following appropriate safety protocols and ensuring sterilization
- Responding to emergencies and acting fast in critical cases
New Question
10 months agoContributor-Level 10
If you want to learn about heart therapies, enjoy science, and comfortable in clinical environments, Cath Lab Technician course may be for you. Here are a few signs:
- You enjoy learning about how the heart operates and how it can be medically treated without surgery.
- You are not afraid to observe procedures or work in hospitals.
- You enjoy completing technical tasks but also enjoy helping the patient/customer through the treatment.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The increasing order of acidity:
o-cresol < Phenol < o-nitrophenol
The electron-withdrawing group on the substituted phenol increases the acidity due to increasing the polarity of the O-H bond and thus, the acidity increases and vice versa. -NO2 is an electron-withdrawing group whereas the -CH3 group is an electron-donating group.
New Question
10 months agoContributor-Level 10
Students who have excelled in Cath Lab Technician typically show a few commonalities:
- Science Background: Students with a background in Biology and/or Physics are generally better able to adapt to the heavier medical and technical content of the course.
- Focus and Precision: Students who are precise and can still focus and remain calm under pressure perform quite well in the clinical environment.
- Interest in Heart Care: Students with a genuine passion for providing care for people with cardiovascular disease and want to support patients through their journey are successful.
- Comfort with machines: Students who are comfortable and have fun us
New Question
10 months agoContributor-Level 10
Selecting the best Cath Lab Technician college ensures quality education and future employment prospects. Below are some things to think about:
- Hospital Tie-Ups: Opt for a college that provides real-time cath lab exposure in hospitals.
- Accreditation & Recognition: Make sure the course is accredited or recognised by appropriate health / educational organizations.
- Faculty Experience: Good faculty members can harness their skill & experience to break-down complex concepts and practical skills.
- Placement Record: A good college should be able to demonstrate strong student placements in hospitals.
- Location & Fees: Choose a colle
New Question
10 months agoContributor-Level 10
Even though college ranking isn't everything, it can have an impact on education quality, exposure to professional equipment, and access to clinical learning in some of the best hospitals.
Oftentimes, the higher-rated colleges, especially in health-care programs, have excellent faculty, better lab facilities, and placement options. But every mid-ranked institute can still provide access to clinical experiences through hospitals and offer practical learning, which can lead to very positive career outcomes after Cath Lab Technician course.
New Question
10 months agoContributor-Level 10
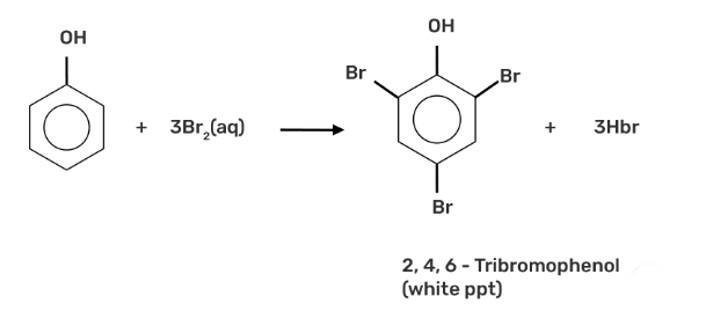
This is a short answer type question as classified in NCERT Exemplar
When phenol is treated with bromine water, it forms the white precipitate of 2, 4, 6-tribromophenol. In the bromination reaction of phenol, water is ionized. And also bromine gets ionized to produce bromonium ions to a larger extent. Phenol gets ionized to produce an ortho-para directing phenoxide ion. Bromine water is mostly used as a test for C=C double bond. During this reaction white precipitates form at the end and bromine water is decolourised.
Hence, the formation of bromonium ions and strong ortho-para directing species indicates that the product formation is
New Question
10 months agoContributor-Level 10
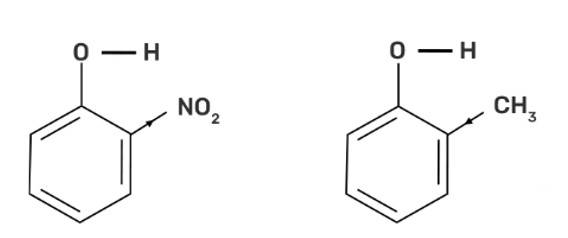
This is a short answer type question as classified in NCERT Exemplar
Out of o-nitrophenol and o-cresol, o-nitrophenol is more acidic due to the -I effect and -R effect of -NO2 group as the -NO2 group is an electron withdrawing group whereas the -CH3 group is electron releasing group and has +I effect on the conjugate base which increases the electron density and decreases the polarity of O-H bond and hence less acidic.
New Question
10 months agoContributor-Level 6
Yes, there are choices in MBOSE SSLC question paper. Students will get choice in almost all sections. They are advised to read the question paper carefully and select the questions wisely.
New Question
10 months agoContributor-Level 10
To get admission for BCom (Hons) at Amity University, candidates need not have studied Mathematics to be qualified as per the university has not mentioned any such subject requirement. BCom (Hons) is a full-time course, offered to students who have completed their class 12 with at least 55%.
New Question
10 months agoContributor-Level 10
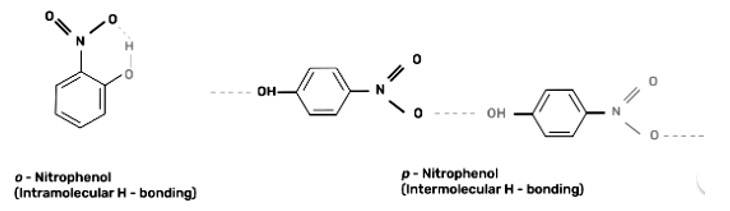
This is a short answer type question as classified in NCERT Exemplar
Out of o-nitrophenol and p-nitrophenol, o-nitrophenol is more volatile due to presence of intramolecular hydrogen bonding in o-nitrophenol whereas p-nitrophenol has intermolecular hydrogen bonding.
New Question
10 months agoContributor-Level 6
The MBOSE SSLC sample papers are released on the official website- mbose.in only. Students can click on MBOSE 10th sample question papers to download them. To get them offline, they can take a printout of the question papers and start solving them for better preparation of the exams.
Solving the MBOSE SSLC sample question papers will help students know their weak areas and work on them.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The strong oxidising agents such as acidified KMnO4 or K2Cr2O7 reagents are used for the conversion of ethanol to ethanoic acid. These reagents are so strong that they convert the alcohols to carboxylic acids.
New Question
10 months agoNew Question
10 months agoContributor-Level 6
Yes, questions are of the same pattern as given in the sample papers. Students must also solve and practice the practice questions and previous years question papers as released by the board.
New Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The reagent used for the conversion of ethanol to ethanal is pyridinium chlorochromate (PCC) which is a complex formed by CrO3, pyridine and HCl which converts the primary alcohols to aldehydes.
CH3—CH2—OH ![]() CH3—CHO
CH3—CHO
Ethanol Ethanal
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
2-chloroethanol is more acidic than ethanol because the conjugate base of 2-chloroethanol is more stable than that of ethanol due to the -I effect of chlorine atom.
Stability: Cl—CH2—CH2—OΘ > CH3—CH2—OΘ
Acidity: Cl—CH2—CH2—OH > CH3—CH2—OH
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts