Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
11.23
Step 1:- Protonation of ethene to form carbocation by electrophilic attack of H3O+.
Step 2:- Nucleophilic attack of water on carbocation.
Step 3:- Deprotonation to form ethanol.
New Question
11 months agoContributor-Level 10
This is the simplest form of chromatography. Here a strip of paper acts as an adsorbent. It is based on the principle which is partly adsorption. The paper is made of cellulose fibres with molecules of water adsorbed on them. This acts as stationary phase. The mobile phase is the mixture of the components to be identified prepared in a suitable solvent.
New Question
11 months agoContributor-Level 10
The application fee at BVIMR is INR 1,200. Candidates must fill out the application form to register for courses such as MBA, BBA, and BCA. Candidates are required to accurately fill in their personal and academic details and upload the scanned documents in the correct mentioned dimensions to successfully submit their application for the desired course.
New Question
11 months agoContributor-Level 10
11.22
There are many ways to this conversion. Two of them are given below:-
(a) In the above conversion, the chlorobenzene is treated with a base such as NaOH, KOH etc. (strong base). The base abstracts the hydrogen from the C-2 position (it can also abstract the hydrogen from the C-6 position, as both are equally acidic) leaving the negative charge at that position.
In the next step Cl- leaves, leaving behind the positive charge at that carbon. Both the negative charge and positive charge forms a bond resulting Benzyne as the intermediate.
After the formation of the Benzyne intermediate OH- of the base attacks at the C-1 position and fu
New Question
11 months agoContributor-Level 10
Estimation of halogens: It involves oxidising the organic substance with fuming nitric acid in the presence of silver nitrate. The halogen of the substance is thus converted to silver halide which is separated and weighed:
Weight of organic compound = W gm
weight of silver halide = x g.
% of halogen =
Estimation of sulphur: The organic substance is heated with fuming nitric acid but no silver nitrate is added. The sulphur of the substance is oxidised to sulphuric acid which is then precipitated as barium sulphate by adding excess of barium chloride solution. From the weight of BaSO4 so obtained the percentage of sulphur can
New Question
11 months agoContributor-Level 10
(i) Dumas method: The nitrogen containing organic compound, when heated with copper oxide in an atmosphere of carbon dioxide, yields free nitrogen in addition to carbon dioxide and water
(ii)Kjeldahl's method: A known mass of the organic compound is heated strongly with conc. H2SO4, a little potassium sulphate and a little mercury (a catalyst). As a result, the nitrogen present in the organic compound is converted to ammonium sulphate.
New Question
11 months agoContributor-Level 10
11.21
The conversion of Phenol from Cumene requires the air oxidation of
The air oxidation of cumene (isopropyl benzene) leads to the production of both phenol and acetone (costlier than phenol).
The air oxidation of cumene gives cumene hydro peroxide as an intermediate which on further hydrolysis (H3O+) gives phenol and acetone.
New Question
11 months agoContributor-Level 10
Lassaigne's test: In organic compounds, nitrogen, sulphur and halogens are covalently bonded. Their detection in 'Lassaigne's test' is possible if they are in the ionic form. This can be achieved by fusing the organic compound with sodium metal.
Chemistry for test for nitrogen:
Sodium fusion extract is boiled with ferrous sulphate and acidified with sulphuric acid. Sodium cyanide reacts with ferrous sulphate and forms sodium hexacyanoferrate (II). On heating with sulphuric acid, some ferrous is oxidized to ferric hexacyanoferate (II) Fe4 [Fe (CN)6]3 which is prussian blue in colour.
Chemistry of the test for sulphur:
Aceti
New Question
11 months agoContributor-Level 10
Sumandeep Vidyapeeth BSc offers degree programmes in multiple streams, such as Radiology Imaging Technology, Cardiac Care Technology, Nursing, Neuro Science Technology, Perfusion Care Technology, Anaesthesia Care Technology, etc. The institute also offers good infrastructure that gives it an edge when compared with other colleges that offer the same courses.
New Question
11 months agoContributor-Level 10
Distillation is used in case of volatile liquid mixed with non-volatile impurities.
Distillation under reduced pressure: This method is used to purify such liquids which have very high boiling points and which decompose at or below their boiling points.
Steam distillation is used to purify steam volatile liquids associated with water immiscible impurities.
New Question
11 months agoContributor-Level 10
Once DSMNRU graduates have a BCom degree, they have range of career choices to make. These include private sector as well as government sector job opportunities. Some of the popular government job opportunities for DSMNRU BCom holders are given below:
Job Profiles | Average Salary |
|---|---|
Bank Probationary Officer | INR 8.6 LPA |
Clerk | INR 5 LPA |
Specialist Officer | INR 8.5 LPA |
Insurance Agent | INR 2.4 LPA |
Assistant Station Master | INR 3.4 LPA |
Assistant Audit Officer | INR 11.3 LPA |
Assistant Accounts Officer | INR 11.3 LPA |
Inspector of Income Tax | INR 7 LPA |
NOTE: The above details are referred to from multiple sources. They are still subject to change and, accordingly, are indicative.
New Question
11 months agoContributor-Level 10
11.20
In ortho nitrophenol there is intra-molecular H bonding, whereas in para-nitrophenol there is inter-molecular H bonding, as shown below:
And because of that para-nitrophenol get tightly bounded with water and ortho nitrophenol is steam volatile and it will leave the solution.
New Question
11 months agoContributor-Level 8
RRB NTPC result is announced on the region-wise official websites of RRBs. The direct link to download the result will be provided on this page. RRB NTPC result is made available in PDF form and comprises the roll numbers of qualified candidates.
New Question
11 months agoContributor-Level 10
Fractional crystallisation is the method that can be used to separate two compounds with different solubilities in a solvent S. A hot saturated solution of these two compounds is allowed to cool, the less soluble compound crystallises out while the more soluble remains in the solution. The crystals are separated from the mother liquor and the mother liquor is again concentrated and the hot solution again allowed cooling when the crystals of the second compound are obtained. These are again filtered and dried.
New Question
11 months agoContributor-Level 10
(a) Crystallisation: In this process the impure solid is dissolved in the minimum volume of a suitable solvent. The soluble impurities pass into the solution while the insoluble ones left behind. The hot solution is then filtered and allowed to cool undisturbed till crystallisation is complete. The crystals are then separated from the mother liquor by filtration and dried.
Example: crystallisation of sugar.
(b) Distillation: The operation of distillation is employed for the purification of liquids from non-volatile impurities. The impure liquid is boiled in a flask and the vapours so formed are collected and condensed to give
New Question
11 months agoContributor-Level 9
New Question
11 months agoContributor-Level 10
11.19
The different forms of cresol is formed with given molecular formula:
(I) 2-methylphenol
(II) 3-methylphenol
(III) 4-methylphenol
New Question
11 months agoContributor-Level 10
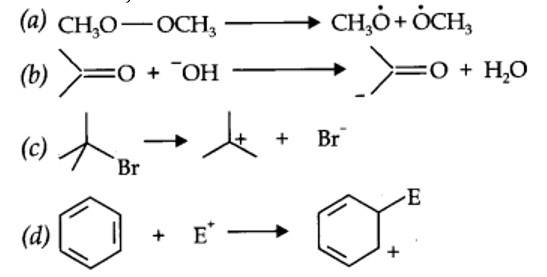
Inductive effect: The inductive effect refers to the polarity produced in a molecule as a result of higher electronegativity of one atom compared to another. Atoms or groups which lose electron towards a carbon atom are said to have +I Effect. Examples of +I effect are (Electron releasing)
(CH3)2C—, (CH3)2CH—, CH3CH2— CH3— etc.
Those atoms or groups which draw electron away from a carbon atom are said to have -I Effect. Examples of -I effect are:
NO2, F, Cl, Br, I, OH etc.
Electromeric effect: The electromeric effect refers to the polarity produced in a multiple bonded compound as it is approached by a reagent.
The atom A has lost
New Question
11 months agoContributor-Level 10
11.18
The hydroboration-oxidation reaction is a two-step reaction that converts an alkene into a neutral alcohol by the net addition of water across the double bond. The hydrogen and hydroxyl group are added in a syn addition leading to the cis configuration. Hydroboration- oxidation is an anti-Markovnikov reaction, with the hydroxyl group attaching to the less substituted carbon. In first step Addition of Hydroborate group is done and in next step, it is oxidized by hydrogen peroxide.
For example: - When propene undergoes hydroboration-oxidation reaction, then it produces propan-1-ol as product. In this reaction diborane i.e., (BH
New Question
11 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts