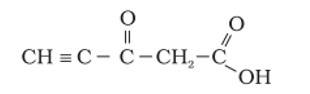Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
Candidates have to pay an application fees of INR 1,000 in order to submit their SBBSU BTech applications. The fee payment can be done online or via demand draft. Students shall keep the fee payment receipt safe for future references.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: (i)

(ii) 
New Question
10 months agoContributor-Level 10
Yes, there are many about 4 best private BMLT colleges in Maharashtra. Some of them are mentioned below along with their tuition fees:
Private Colleges | Tuition Fee |
|---|---|
Dr. D.Y. Patil Vidyapeeth, Pune Admission | INR 2.25 lakh |
DY Patil University, Navi Mumbai Admission | INR 6 lakh |
Alard University Admission | INR 5 lakh |
Ajeenkya DY Patil University Powered by Emversity Admission | INR 8 lakh |
ITM - Institute of Health Sciences Admission | INR 2.1 lakh |
Disclaimer: This information is sourced from official website and may vary.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: (i) According to the electronic configuration, a total of 8 electrons must be present in the valence shell of an element. Element X has 4 valence electrons, so it will share the remaining 4 electrons for the formation of the bond, the molecular formula will be XH4 . The element Y has 5 valence shell electrons, so it will form 3 bonds and the formula will be YH3 . The element Z has 7 valence shell electrons, so it will form one bond with hydrogen and has the molecular formula H-Z .
(ii) Elements X, Y and Z having, 5 and 4 7 valence electrons respectively belongs
New Question
10 months agoBeginner-Level 5
The Interjections can be divided into five broad types:
· Primary Interjections
· Secondary Interjections
· Volitive Interjections
· Emotive Interjections
New Question
10 months agoContributor-Level 10
Yes, Creative Techno College admission 2025 is open. Interested students can visit the official website to apply for the respective course. The college offers three courses at the UG level. To be admitted to the Creative Techno College, candidates must meet the eligibility criteria set by the college. The admissions are based on merit.
New Question
10 months agoContributor-Level 10
The students should read the guidelines given on Class 10 board exam question paper. Doing both options will take time. So they should read the questions and the answer should be written for the one they know well.
New Question
10 months agoContributor-Level 10
Listed below are the top BMLT colleges in Maharashtra along with their NIRF rankings over the past three years:
Top Colleges | NIRF 2022 | NIRF 2023 | NIRF 2024 |
|---|---|---|---|
17 | 15 | 11 | |
Datta Meghe Institute of Higher Education and Research Ranking | NA | 25 | 23 |
NA | NA | 30 |
Disclaimer: This information is sourced from official website of the ranking body listed and may vary.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: BeCl2 has a linear structure
HOCl is also non-linear in structure.
H2O has a V-shaped structure.
Cl2O has a V-shaped structure.
New Question
10 months agoContributor-Level 10
As per NIRF ranking 2024, Dr. D. Y. Patil Vidyapeeth emerged to be as the top BMLT college in Maharashtra, secured 11th position followed by Datta Meghe Institute of Higher Education and Research and Armed Force Medical College.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The hybridization of Carbon 1 is sp, carbon 2 is sp, carbon 3 sp2, carbon 4 is sp3 and carbon 5 is sp2. The triple bond has 2 pie bonds and one sigma bond. Each double bond has one sigma and one pie bond. Every single bond is a sigma bond. Thus, the total number of sigma bonds is 11 and pie bonds are 4.
New Question
10 months agoContributor-Level 10
About 25+ best BMLT colleges are there in Maharashtra. Students can secure admission after passing Class 12 boards. Some of the top Medical Laboratory Technology colleges in Maharashtra include Dr. D.Y. Patil Vidyapeeth Pune, Datta Meghe Institute of Higher Education and Research, Armed Forces Medical College, DY Patil University Navi Mumbai, University of Mumbai [MU], Oasis College of Science and Management, AIIMS Nagpur, Council of Education and Development Programmes, and many others. Of these, 11 colleges are privately owned and 6 colleges are owned by public/government organisations.
New Question
10 months agoContributor-Level 10
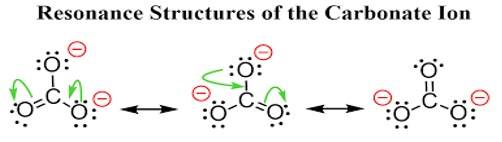
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The carbonate ion CO32- can be best represented by its resonating structures. The carbonate ion cannot be represented by a single Lewis structure because the three carbon oxygen bond lengths are the same. This cannot be shown by a single Lewis structure. For showing the similar lengths of all the carbon to oxygen bonds three hybrid structures are constructed which are in resonance with each other.
New Question
10 months agoBeginner-Level 5
An interjection is one of the eight parts of speech, which are defined by one or a couple of words used to express the mood or emotion. Although the Interjection does not impact the meaning of the sentence as a whole but acts as an ornamentation in the sentence.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
N—H, F—H, C—H and O—H
Ans: The ionic character in a molecular species is decided by the electronegativity difference between the two bonded atoms. Greater the electronegativity difference between two bonded pairs, the greater will be the ionic character. The electronegativity difference of the given species is
C - F = (2.5 - 2.1) = 0.4
N -H = (3.0 - 2.1) = 0.9
O - H = (3.5 - 2.1) = 1.4
F - H = (4.0 - 2.1) = 1.9
According to the above given difference in the electronegativities, the order of increasing ionic character is:
C – H< N - H
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Ionic bonds are such bonds in which complete transfer of electrons from one atom to another atom. Due to complete transfer positive and negative ions are formed in this bond. The ions in this bond are held together by electrostatic force of attraction. The formation of calcium fluoride CaF2 leads to the formation of an ionic bond.
Ca→ Ca2+ + e - The electronic configuration of Ca= [Ar] 4s2 and Ca2+= [Ar]
F +e -→F- The electronic configuration of F= [He] 2p5 and F = [He ]2p6
Thus Ca2+ +2F- → CaF2
A covalent bond
New Question
10 months agoContributor-Level 10
Sant Baba Bhag Singh University BTech admission cycle starts with the application process. The eligible students can apply in both online and offline modes. SBBSU selection committee scans the received applications and shortlist them. The selection is done based on performance in the last qualifying examination. However, to be eligible for admission, candidates should be Class 12 pass with required aggregate and subject combination. These selected students have to then get their documents verified and pay the required fee amount.
New Question
10 months agoContributor-Level 10
Candidates can get admission to the IISER Pune MSc course without IIT JAM scores. However, it completely depends on the specialisation candidates have selected. Candidates can get admission into MSc in Mathematics and MSc in Physics (Quantum Technology) without IIT JAM scores. For MSc Physics, candidates with relevant scores in the GATE exam with subject codes PH, EE, EC, IN, MA, ST, CS, and DA can also apply, and for MSc Mathematics, the institute accepts its in-house-entrance test.
New Question
10 months agoContributor-Level 10
The Gujarat Technological University offers admission to BSc course for students shortlisted by the GCAS. The GCAS releases a merit list which is based on the score of Class 12. Therefore, there are no provisions for direct admission for the students. It is a must to clear the eligibility for getting admission.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts