Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The electronic configurations of O2+ and O2 - according to molecular orbital theory is:
O2+: σ1s2 σ∗1s2 σ2s2 σ∗2s2 s2pz2, π2p2y , π2p2x, π*2p1x
O2- : σ1s2 σ∗1s2 σ2s2 σ∗2s2 s2pz2, π2p2y , π2p2x, π*2p2y π*2p1x
The bond order of O2+
Bond order = ( Nb- Na)
Bond order = (10-5)=2.5
The bond order of O2-
(10-7)= = 1.5
As the bond order of O2+ is higher, it is more stable than O2 -, because higher the bond order more stable is the bond. Both the molecular species
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: In the Lewis structure of PCl3, the phosphorus atom is surrounded by three bond pairs (chlorine atoms) and one lone pair. These four electron pairs are arranged in a tetrahedral geometry around the central phosphorus atom. Due to the presence of lone pair of electron on the phosphorus atom, PCl3 will have a distorted tetrahedral geometry. Thus, it will form a pyramidal shape and is non-linear in structure.
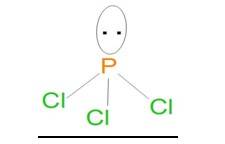
In H2S, the central sulphur atom is surrounded by two bond pairs and two lone pairs of electrons. It can be said that these four electron pairs are arranged in
New Question
10 months agoContributor-Level 10
The Gujarat Technological University offers students with the BSc course in different specialisation. Computers Science and Biotechnology being among them. Therefore, students looking forward to BSc course in the mentioned specialisations can chcek the eligibility criteria and get themselves registered with the GCAS portal.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: Option (iii)
Molecules from hydrogen to nitrogen shows this type of electronic configuration:
σ1s σ*1s< σ2s < σ*2s< [ π2px = π2py] < σ2pz< [ π*2px = π*2py] σ*2pz in which σ2pz is filled after π2px and π2py
So, amongst the given elements only nitrogen will show this type of electronic configuration in which σ2pz molecular orbital is filled after π2py and π2px
The electronic configuration of N2 is:
σ1s2 σ∗1s2 σ2s2 σ∗2s2, π2p2x , π2p2y, s2pz2 π*2p1x
New Question
10 months agoContributor-Level 10
Yes, Sant Baba Bhag Singh University is currently accepting applications for various courses. BTech course aspirants who meet the eligibility requirements can apply. The students can visit the official website to apply. For offline forms, the admission office shall be contacted. The aspirants are advised to apply as soon as possible.
New Question
10 months agoContributor-Level 9
No, candidates are not allowed to get direct admissions to MSc courses at IISER Pune. Indian Institute of Science Education and Research, Pune, is one of the seven Indian Institutes of Science Education and Research, and candidates are not allowed to get direct admission into any of its offered courses. Moreover, to get admissions, candidates are required to appear in the relevant entrance exam associated with the offered specialisation.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: Option (ii)
(i) The electronic configuration of O2 is:
σ1s2 σ∗1s2 σ2s2 σ∗2s2 s2pz2 π2p2y π2p2x π*2p1y π*2p1x
Bond order = ( Nb- Na)
Bond order = (10-6)=2.0
The electronic configuration of N2 :
σ1s2 σ*1s2 σ2s2 σ*2s2 π2p2x, π2p2y σ2p2z
Bond order = (10-4)=3.0
So, the bond order of O2 , N2 will not be equal.
(ii) The electronic configuration of O2+ is:
σ1s2 σ∗1s2 σ2s2 σ∗2s2 s2pz2,π2p2y , π2p2x, π*2p1x
Bond order = (10-5)=2.5
The electronic conf
New Question
10 months agoContributor-Level 10
Completed a 3-year graduation in any stream can apply for the School of Government, MIT WPU MA. A minimum of 50% aggregate marks is required (45% for Reserved Class category candidates from Maharashtra State only). Meeting the eligibility criteria is mandatory to proceed with the admission process.
New Question
10 months ago
Contributor-Level 10
Creative Techno College course admissions are based on merit. To enroll for course admission, candidates must complete Class 12. The college offers BSc in Computer Science, BCA, and BBA programmes at the undergraduate level. The courses are offered in the field of Science, Computer Application and Business Administration.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: Option (ii)
Nodal plane is a plane which passes through the nucleus and the probability of finding an electron on a nodal plane is zero. Amongst the above given molecular orbital, only π*2px contains two nodal planes. Rest of the molecular orbitals, σ∗1, π2px, π*2py contains one nodal plane.
New Question
10 months agoContributor-Level 8
Candidates for KIITEE Law 2026 (5-year integrated B.A./BBA/B.Sc LL.B) must fulfil the following requirements:
Academic Requirement: Successfully completed or appearing in 10+2 for any stream; however, for B.Sc. LL.B., the qualifying 10+2 must be in Science.
Minimum Scores: 40% for SC/ST/OBC categories and at least 45% overall for General and PwD categories
- According to the guidelines set forth by the Bar Council of India, there is no maximum or lower age limit.
- Candidates in the final year of high school are eligible to apply, as long as they pass the test before being admitted.
New Question
10 months agoContributor-Level 10
No, the pre board exam marks will not be considered in the Class 10 Board examination marks. Pre-Board exams are conducted to make students familiar with the Class 10 board exam. This will help students to understand the pattern of exam, difficulty level, time management etc. It also help them to know how well they are prepared for the Class 10 Board examination.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: (i) The electronic configuration of dioxygen is:
σ1s2 σ∗1s2 σ2s2 σ∗2s2 s2pz2 π2p2y π2p2x π*2p1y π*2p1x
(ii) As it can be seen from the electronic configuration of oxygen atom π*2p1X π*2p1Y the are partially. So, the statement given is incorrect.
(iii) The statement given is correct because there are five bonding molecular orbitals and four antibonding molecular orbital in oxygen molecules. Hence, the bonding and antibonding molecular orbitals are no equal.
(iv) The filled bonding orbitals are not the sam
New Question
10 months agoContributor-Level 10
The MA programme offered by the School of Government, MIT WPU is a two-year full-time course. It is divided into four equal semesters, helping students build a strong foundation in Political Leadership and Government. The structure ensures a balanced academic load across the duration of the programme.
New Question
10 months agoContributor-Level 10
For admissions to IISER Pune MSc courses, the candidates are obliged to perform well and secure good scores in the accepted entrance exams, such as IAT, IIT JAM, GATE, etc. To apply, fill out the IISER Pune application form visit the official website of the institution, fill out the form with the required details, pay the application fee and submit it. Moreover, the candidates who got shortlisted in the merit list can pay the course fee to confirm their seat.
New Question
10 months agoContributor-Level 10
Aspirants looking forward to take admission in the BSc programme at the GTU should check for the eligibility criteria as the admissions to the university is granted on the basis of Class 12 merit. Students who are not appearing for the CUET examinations are also eligibile if the have completed their class 12 with valid score and from a recognised board.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: (i) Different types of hybridization in a carbon atom are:
(a) sp hybridization: The carbon atoms forming triple bonds with each other determines hybridized carbon. ( Cº C ).
(b) sp2 hybridization: The carbon atoms forming double bonds with each other determine sp2 hybridized carbon. (C=C). (c) sp3 hybridization: The carbon atoms forming single bonds with each other determine sp3 hybridized carbon. ( C-C).
(ii)The starred C atom is sp2 hybridised.
B] The starred C atom is sp3 hybridised.
C] The starred C atom is sp2 hybridised.
D] The starred C atom is sp3 hybridis
New Question
10 months ago
Contributor-Level 10
The KLAT 2026 registration is closed. Candidates could apply for the K LAT Exam 2026 until January 9. Aspirants need to complete slot booking, take mock tests, and appear for the exam on January 18.
New Question
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: Hybridisation in the case of PCl5 :
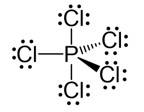
The hybridisation of phosphorus PCl5 is sp3d and it has a trigonal bipyramidal geometry. The axial bonds in PCl5 are slightly longer as compared to the equatorial bonds because axial bonds experience greater repulsive forces from other bonds as compared to the equatorial bonds.
Hybridisation in the case of SF6:
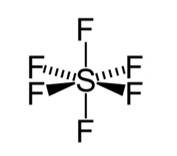
The hybridisation of sulphur in SF6 is sp3d2 and the molecule has an octahedral geometry. The bond length of axial as well as the equatorial bonds are similar because all the bonds in octahedral geometry experience similar r
New Question
10 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts