Ask & Answer: India's Largest Education Community
All Questions
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: (i) Due to the electrostatic forces between the two opposite charges, ionic bonds are non-directional. The bonding direction does not matter as the electrostatic field of an ion is non-directional. Whereas, the formation of covalent bonds happens with the overlap of the atomic orbitals. The direction of the bonds is given by the direction of overlapping.
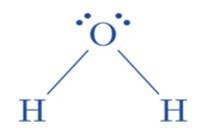
(ii) The hybridization of the oxygen atom in water molecules is 3 sp due to the presence of two lone pairs of electrons on the oxygen atom. Tetrahedral geometry is acquired by these four 3 sp hybridized or
New Question
10 months agoNew Question
10 months agoContributor-Level 7
Interesting Question! So, While BFA is a rewarding field, there are several challanges in this fiel also such as:
- Financial Uncertainty - Art careers can take time to become profitable.
- Freelance Struggles - Many jobs are project-based with irregular income.
- Competition - High competition in industries like animation, design, and photography.
- Lack of Awareness - Many people undervalue artistic careers, leading to family/social pressure.
- Continuous Learning Required - Artists need to constantly upgrade skills in software, digital trends, and new media.
I can understand these challenges may sound a little bit terrifying but in todays world the
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: (i) N2 → N2+ + e-
The electronic configuration of N2 is:
σ1s2 σ*1s2 σ2s2 σ*2s2 π2p2x = π2p2y σ2p2z
So, its bond order will be 3
The electronic configuration of N2+ is:
σ1s2 σ*1s2 σ2s2 σ*2s2 π2p2x = π2p2y σ2p1z
Its bond order will be 2.5
Hence the bond order decreases in this reaction N2 → N2+ + e-
(ii) O2→ O2+ + e-
The electronic configuration of O2 is:
σ1s2 σ∗1s2 σ2s2 σ∗2s2 s2pz2 π2p2y π2p2x π*2p1y π*2p1x
Its bond order will be 2
The elec
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The general sequence of the energy level of the molecular orbital is σ1s < σ*1s < σ2s< σ*2s < π2px = π2py < σ2pz
N2 = σ1s2 σ*1s2 σ2s2 σ*2s2 π2p2x = π2p2y σ2p2z
N2+ = σ1s2 σ*1s2 σ2s2 σ*2s2 π2p2x = π2p2y σ2p1z
N2- = σ1s2 σ*1s2 σ2s2 σ*2s2 π2p2x= π2p2y σ2p2z σ2p2x
N22+ = σ1s2 σ*1s2 σ2s2 σ*2s2 π2p2x = π2p2y
The formula for finding the bond order (B. O) for any molecular species is:
New Question
10 months agoNew Question
10 months agoContributor-Level 10
BSc Agri. Practical subjects include
- Agronomy
- Horticulture
- Soil Science
- Plant Pathology
- Entomology
- Animal Husbandry
- Agricultural Engineering
BSc Agri. Practical Exercises:
- Measuring irrigation water.
- Determining soil moisture content.
- Calculating consumptive use of water.
- Exercises on drainage and irrigation requirements.
- Calculating irrigation water use efficiency.
- Visiting irrigation and drainage projects.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: The Lewis structures of the compounds are:
Lewis structure for nitric acid:
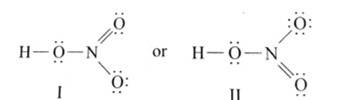
Lewis structure of nitrogen dioxide:
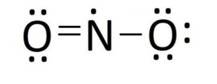
Lewis structure of sulphuric acid:
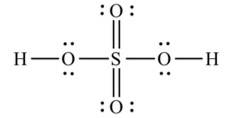
New Question
10 months agoContributor-Level 10
The key highlights of IIT Guwahati placements 2024 and 2025 for M.Tech are presented below:
Particulars | Placement Statistics (2024) | Placement Statistics (2025) |
|---|---|---|
| the highest package | INR 65.70 LPA | INR 39.35 LPA |
| Average Package | INR 14.55 LPA | INR 14.17 LPA |
| Median Package | INR 11 LPA | INR 11 LPA |
| the lowest Package | INR 4.20 LPA | INR 6 LPA |
| Students Placed | 313 | 209 |
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Although the hybridisation of the central atom oxygen in both the molecules is 3 sp but dimethyl ether will have a higher bond angle than water molecule.
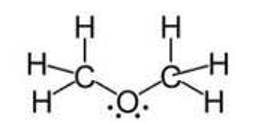
Due to the presence of two bulky methyl groups in dimethyl ether, the repulsive forces will be greater in them than the two hydrogens in water molecules. In dimethyl ether the -CH3 is a group attached to three hydrogen atom through s bonds. Thus, the C- H bond pairs increase the electron density on the carbon atom which results in lone pair-bond pair repulsions. Due to this lone pair-bond pair repulsions, the bon
New Question
10 months agoContributor-Level 10
If a students drops one year, he or she will have to learn syllabus given by board. They can take help from their teachers for 10th board syllabus. They can download syllabus from official website of board.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: In PCl5, P has 5 valence electrons in orbitals and make 5 bonds with 5 Cl atoms, it will share one of its electrons from 3s to 3d orbital, therefore the hybridization will be sp3d and the geometry will be trigonal bipyramidal. IF5, the Iodine atom has 7 valence electrons in molecular orbitals it will form 5 bonds with 5 Cl atoms using 5 electrons from its molecular orbital, two electrons will form one lone pair on Iodine atom, which gives the square pyramidal geometry.
New Question
10 months agoNew Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
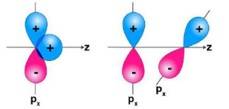
Ans: In the first figure given above, the area which is under + overlapping is equal to the area under +- overlap. Both the overlaps cancel out with each other as they are oppositely charged. Due to cancelling out of the overlaps the net overlap will be zero. In the second figure given above, both the p-orbitals are perpendicular to each other. Due to the, px py orbitals being perpendicular with each other, no overlap will be possible.
New Question
10 months agoContributor-Level 10
For the first year of the MA programme at the School of Government, MIT WPU, students are required to pay INR 1,55,000. This includes tuition and may cover additional components as specified by the institute. It's important to verify the latest fee details from the official website before making payments.
New Question
10 months agoContributor-Level 10
At the last stage of Ganpat University BSc admissions, students who are offered admission are required to pay the first installment of the first-year fees. In general, students are required to pay one-time fees and some part of the tuition fees. To know more, students can visit the official website of the university.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
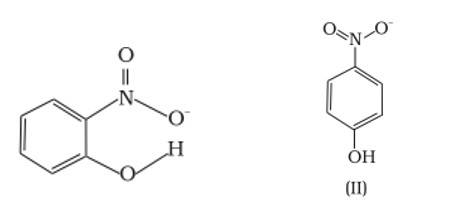
Ans: (a) Which of the two compounds will have intermolecular hydrogen bonding and which compound is expected to show intramolecular hydrogen bonding.
Ans: The intramolecular hydrogen bonding is shown by compound (I) and the intermolecular hydrogen bonding is shown by compound (II) . In compound (I) the NO2 and OH group are close together in comparison to that in compound (II) . So, that is why compound (I) shows intramolecular hydrogen bonding. The intermolecular hydrogen bonding in compound (II) is shown as:
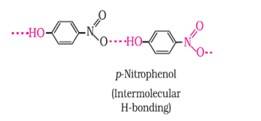
(b) The melting point of a compound depends on, among other
New Question
10 months agoContributor-Level 10
Even if a student fails in pre board exam, he or she can appear for the Class 10 board exam. Model exams help students to know how well they are prepared for the Board exam. A student cannot be detained from appearing in the Class 10 Board exam, if failed in pre board exam. Hence students need not worry, if they fail in pre board. They are advised to prepare well within the remaining time and appear for the board exam.
New Question
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: In BrF5 the central bromine atom has 7 valence electrons. It makes five bonds with the fluorine atom and one lone pair of electron is left. Due to the lone pair-bond pair repulsions, BrF5 makes a structure of square pyramidal geometry. Due to the distortion of fluorine ions, each fluorine ion makes an angle of 90° . The square pyramidal shape of BrF5 is
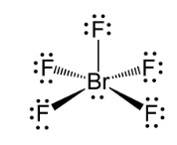
New Question
10 months agoContributor-Level 10
The total tuition fee for the MA programme in School of Government, for two years is INR 3.1 Lakh. This fee is subject to revision by the institute. Students are advised to check the updated fee structure carefully before applying.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts