Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
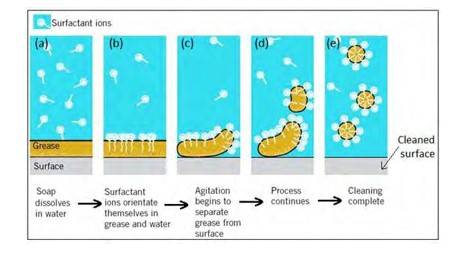
16.30 Soap molecules form micelles around an oil droplet [dirt] in such way that the hydrophobic parts of the stearate ions attach themselves to the oil droplet and the hydrophilic parts projects outside the oil droplet. Due to the polar nature of the hydrophilic parts, the stearate ions [along with the dirt] are pulled into the water, thereby removing the dirt from the cloth.
New Question
11 months agoContributor-Level 10
According to Henry's law,"At a constant temperature, the amount of a given gas that dissolves in a given type and volume of liquid is directly proportional to the partial pressure of that gas in equilibrium with that liquid."
Stated as,
p = KHx
Where, P = partial pressure of the solute above the solution
KH = Henry's constant
x = concentration of the solute in the solution
Given,
Solubility of H2S in water at STP is 0.195 m
We know,
At STP pressure p = 0.987 bar
0.195 mol of H2S is dissolved in 1000g of water
Moles of water = 1000/18
= 55.56 g/mol
∴ the mole fraction of H2S = Moles of H2S / Moles of H2S + Moles of w
New Question
11 months agoContributor-Level 10
16.29
Soaps can be very easily used for checking the hardness of water as soap lathers with soft water and with hard water, soap is precipitated.
On the other hand, synthetic detergents are soluble in hard water and soft water. Hence it cannot be used for checking the hardness of water.
New Question
11 months agoContributor-Level 10
(a) Molality, also called molal concentration, is a measure of the concentration of a solute in a solution in terms of amount of substance in a specified amount of mass of the solvent. Molar mass of KI = 39 + 127 = 166 g/mol.
20% aqueous solution of KI means 200 g of KI is present in 1000 g of solution. Therefore,
Molality = Moles of KI / Mass of Water in kg
= (200/166) / (0.8) = 1.506 m
(b) Molarity is the concentration of a solution expressed as the number of moles of
solute per litre of solution.
Given,
Density of the solution = 1.202 g/mL
Volume of 100 g solution = mass/ density
= 100/1.202
= 83.1
New Question
11 months agoContributor-Level 10
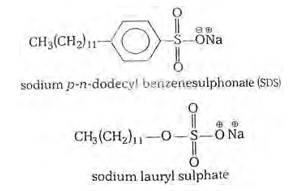
16.28
The commonly used soaps are the sodium and potassium salts of higher fatty acids. They are soluble in water and give good lather to it. On the other hand, calcium and magnesium salts of higher fatty acids (calcium and magnesium soaps) are insoluble in water and do not produce lather. Hard water contains Ca2+and Mg2+ ions. When a sodium or potassium soap is added to hard water, it gets converted into an insoluble calcium or magnesium soap as shown ahead.
Due to the conversion of sodium or potassium soaps into calcium or magnesium soaps in the presence of hard water, the common soaps are unable to emulsify the greasy dirt and c
New Question
11 months agoContributor-Level 10
2.5 kg of 0.25 molal aqueous solution.
Molar mass of urea (NH2CONH2) = (2 (1 * 14 + 2 * 1) + 1 * 12 + 1 * 16)
= 60 g/mol
1000 g of water contains 0.25 mol = (0.25 * 60) g of urea.
= 15 g of urea.
Means, 1015 g of solution contains 15 g of urea
Therefore,
2500 g of solution contains = 15 X 2500 / 1015
= 36.95 g
Hence, mass of urea required is 37 g (approx).
New Question
11 months agoBeginner-Level 5
In case a candidate does not report to the institute allotted to him or her during the NEET SS counselling process, then the security deposit paid by him or her will be forfeited. After that, they will be eligible to participate in the further rounds of counselling after re-registration and choice filling and locking. However, their registration fee will not be refunded.
New Question
11 months agoContributor-Level 10
16.27
It has been found that the detergents having a great deal of branching in the hydrocarbon tail are not biodegradable and cause pollution in rivers and waterways. This is because the presence of side chains in the hydrocarbon tail stops bacteria from attacking and breaking the chains.
This results in slow degradation of detergent molecules leading to their accumulation in water ways. Alkylbenzene sulphonates having branched chain alkyl groups possess poor biodegradability and cause pollution in rivers and water Such detergents are termed as non-biodegradable or hard detergents.
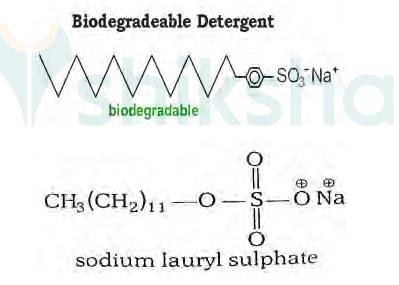
The alkyl sulphates do not possess branching and a
New Question
11 months agoContributor-Level 10
Molarity = Moles of Solute / Volume of Solution in liter
(a) Given, In 4.3 L of solution there is 30 g of Co (NO3)2. 6H2O
Molar mass of Co (NO3)2.6H2O = (1 × 59 + 2 × (1 × 14 + 3 × 16) + 6 × 18)
= 291 g/mol.
∴ Moles = Given Mass / Molar Mass = 30/291 = 0.103 mol.
Now, Molarity = 0.103 mol / 4.3 L
= 0.023 M
(b) Given, 30 mL of 0.5 M H2SO4 diluted to500 mL.
In 1000 mL of 0.5 M H2SO4, number of moles present is 0.5 mol.
∴ In 30 mL of 0.5 M H2SO4, number of moles present = 30X 0.5 / 1000 mol.
= 0.015 mol.
∴ Molarity = 0.015 mol / 0.5L
= 0.03 M.
New Question
11 months agoContributor-Level 9
With an acceptance rate of only 12.8%, Boston University can be considered quite selective as out of every 100 applications, only 12 to 13 students are rolled out an offer letter. International students are required to meet ceratin admission requirements.
Below are the UG admission requirements for Boston University:
- Secondary School Records: Higher Secondary Certificate / ISC (State Board exams may also be examined for admission). Also, submit copies of all qualifying exams that have been taken throughout the secondary school
- English language proficiency test results
- Financial support documentation
- Passport data page
- SAT&n
New Question
11 months agoContributor-Level 10
Let the total mass of the solution be 100 g and the mass of benzene be 30 g.
∴ Mass of carbon tetrachloride = (100 - 30) g = 70 g
Molar mass of benzene (C6H6) = (6 × 12 + 6 × 1) g mol -1
= 78 g mol -1
∴ Number of moles of C6H6 =30/78 mol
= 0.3846 mol
Molar mass of carbon tetrachloride (CCl4) = 1 × 12 + 4 × 35.5
= 154 g mol -1
∴ Number of moles of CCl4 = 70/154 mol
= 0.4545 mol
Thus, the mole fraction of C6H6 is given as:
Number of moles of C6H6 / Number of moles of C6H6 + Number of moles of CCl4
= 0.3846 / (0.3846 + 0.4545)
New Question
11 months agoContributor-Level 10
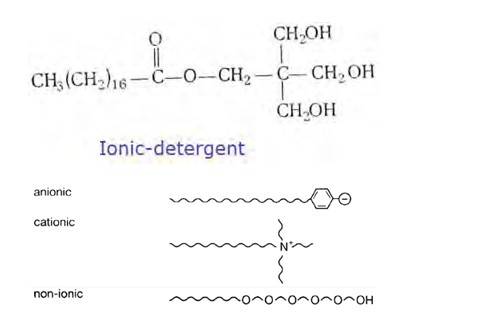
16.26
(i) Cationic detergents- Cationic detergents are quaternary ammonium salts [chlorides, bromides, acetates, etc.] having long chain alkyl groups. The cationic detergents are more expensive than anionic detergents and hence they find only limited use. However, they possess germicidal properties and are used quite extensively as germicides. The examples of cationic detergents are as follows:
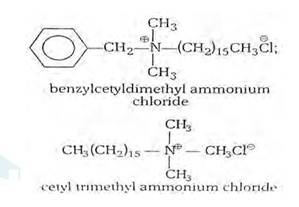
(ii) Anionic detergents- A detergent is said to be anionic when the large part of its molecule is an anion and is involved in the cleansing action. The anionic detergents are also effective in slightly acidic In slightly acidic solutions,
New Question
11 months agoContributor-Level 10
Mass of Solution = Mass of Benzene + Mass of Carbon Tetrachloride
= 22 g + 122 g = 144 g
Mass percentage of Benzene = Mass of Benzene / Mass of Solution X 100 = 22/144 X 100 = 15.28%
Mass percentage of CCl4 = Mass of CCl4 / Mass of Solution X 100 = 122/144 X 100 = 84.72%
New Question
11 months agoContributor-Level 10
Jaypee Business School offers scholarships for its students. The scholarships are mainly offered by the institute via the government. JBS Noidan mainly offers one scholarships: William Webster Merit cum Means Scholarship. Candidates can only avail the scholarship once they are officially admitted to the BSc course under JBS Noida. As per the scholarship, selected students will get a tuition fee waiver for the year up to a maximum of one semester Tuition Fee. All scholarship amounts will be adjusted against the tuition fees of the year in which approved.
New Question
11 months agoContributor-Level 10
16.25
Synthetic detergents are superior cleansing agents as compared to soaps. This is due to the following reasons.
1. Detergents are soluble even in hard water. This is because of calcium and magnesium ions present in hard water form soluble salts with detergents. Hence, detergents can be used both in soft as well as in hard water. On the other hand, soaps form insoluble salts with calcium and magnesium ions and cannot be used in hard water.
2. The aqueous solutions of detergents are usually neutral. Therefore, they do not damage delicate fabrics and can be used for washing almost all types of On the other hand, aqueous solu
New Question
11 months agoContributor-Level 10
Given-
Mass of K2SO4, w = 25 mg = 25 X 10-3 g,
Molar mass of K2SO4 = (39×2) + (32×1) + (16×4) = 174 g mol-1
Volume V = 2 liter
T = 250C + 273 = 298 K (add 273 to convert in Kelvin)
The reaction of dissociation of K2SO4 is written as,
K2SO4 → 2K + + SO42-
Number if ions produced = 2 + 1 = 3, hence vant Hoff’s factor, I = 3
Here, we use vant Hoff’s equation for dilute solutions, given as,
πV = inRT
where, n is the number of moles of solute, R is solution constant which is equal to the gas constant (0.082) and T is the absolute temperature (298 K).


Hence, the osmotic pressure of a solution is 5.27x10-3atm
New Question
11 months agoContributor-Level 10
16.24
Alitame is a high potency artificial sweetener and it is not possible to control the sweetness imparted to food by alitame. Alitame is an artificial sweetener that is 2,000 times as sweet as sugar. This sweetness is very high as compared to the natural sugar and use of such sweetener is very critical while preparing sweet dishes. The structure of alitame is as follows:
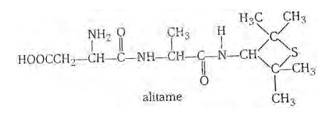
New Question
11 months agoContributor-Level 10
Given-
Vant Hoff’s factor, I = 2.47
osmotic pressure, π = 0.75 atm
Volume of solution = 2.5L.
To determine the amount of CaCl2, we use vant Hoff’s equation for dilute solutions, given as,
πV = inRT
where, n is the number of moles of solute, R is solution constant which is equal to the gas constant and T is the absolute temperature.

Hence, the amount of CaCl2 dissolved is 3.425g
New Question
11 months agoContributor-Level 10
Given-
KH for O2 = 3.30 * 107 mm Hg,
KH for N2 = 6.51 * 107 mm Hg
Percentage of oxygen (O2) = 20 %
Percentage of nitrogen (N2) = 79%
Total pressure = 10 atm
Using Henry's law,

where, p is the partial pressure of gas in the solution and KH is Henry's constant.

Thus, the mole fraction of oxygen in solution, xoxy = 4.61x10-5
and the mole fraction of nitrogen in solution, xnit is 9.22x10-5
New Question
11 months agoContributor-Level 10
16.23
Artificial sweetening agents like Saccharin, Alitame, Sucrolose can be used in the preparation of sweets for a diabetic patient as they do not add any calories to the body.
Diabetic people are advised to consume low-calorie diet [fewer carbohydrates & more proteinaceous and fiber rich]. The refined sugar like sucrose adds calorie to the diet but complex sugar and starches do not add to the calorie intake of a person and at the same time impart a sweet taste to diet. Artificial sweeteners are generally either complex sugar or protein in nature
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts