Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
The B Tech in ECE syllabus is planned out in a manner as to ensure a balance between both classroom learning through books and hands-on training via lab sessions, project work, and workshops. There is a strong focus on both aspects of the coursework, ensuring that you have good understanding of the important concepts of the field and can look forward to good earning opportunities because of the practical knowledge and technical proficiency in the ECE field.
Useful Link:
New Question
11 months agoContributor-Level 10
The BTech ECE syllabus is a combination of core subject modules and elective subjects coupled with lab sessions, industrial projects, internships, and workshops. When it comes to the core subjects, there is no such scope for customization, as the subjects are meant to give the foundational knowledge needed to have a strong hold on the field basics to do well at the job front in the future.
However, in the case of the elective subject choices, you can choose the subjects based on your interests, the ones that you find scoring, fun to study, easy to grasp, and aligned with your future plans in mind. This allows you to customize the BTech
New Question
11 months agoContributor-Level 10
To join IIT Madras after Class 12 candidates must meet the eligibility criteria set by the college. IIT Madras course admissions are primarily entrance-based. The institute offers various programmes at the undergraduate courses. BTech at IIT Madras is a four-year programme offered across 10 specialisations in full-time mode. Admission to IIT Madras BTech courses is completely entrance-based. IIT Madras BTech admission is based on the JEE Main, followed by the JEE Advanced score. The centralised counselling process is managed and regulated by the JoSAA, based on which candidates are allotted
New Question
11 months agoContributor-Level 9
Yes, candidates can get admission in Jaypee Business School without CAT exam. As per the school admission guidelines, admission to the MBA course is based on various national-level entrance exams. Candidates must also check the eligibility criteria before applying for the courses offered at Jaypee Business School. The basic eligibility criteria is that the candidate must have a bachelor's degree or its equivalent in any discipline from a statutory university with at least 50% aggregate marks or equivalent CGPA. Few of the accepted entrance exams are as follows:
- MAT or Management Aptitude Test or MAT
- XAT or Xavier Aptitude Test
- CMAT o
New Question
11 months agoContributor-Level 10
The general time span for a PhD in Electronics & Communication Engineering is 3 to 6 years, depending on your chosen research topic. A PhD degree in ECE requires extensive research on a particular topic, which must be backed by authentic research findings that need to be presented at the end of the course duration in the form of a comprehensive thesis under your supervisor's guidance.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
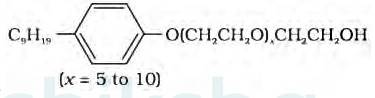
The functional group present in the molecule is:
1. Ether
2. Primary Alcoholic group
Alcohol group can recognize by the presence of –OH group present in the given organic compound and on the other hand, the ether group can be identified by the presence of R-O-R bonds where R represents the alkyl group present in the compound.
New Question
11 months agoContributor-Level 10
You should start your prep at least 8 months before.
- Go through the exam syllabus in detail and note down all the important topics.
- Prepare a weekly timetable.
- Practice previous year's question papers, get familiar with the test pattern.
Know more about -
New Question
11 months agoContributor-Level 10
They are generally considered moderate to difficult. Requiring a strong hold over Physics, Chemistry, and Mathematics subjects. A good general aptitude and logical reasoning. The difficulty level of the entrance test also depends on individual preparation, the number of applicants, last year's results, and student feedback. You should start your preparation as early as possible and cover the maximum syllabus to score well in the exam.
New Question
11 months agoContributor-Level 10
16.4
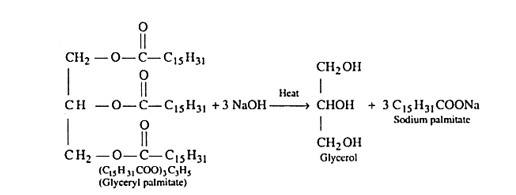
Soaps are metallic salts of higher fatty acids such as palmitic acid, stearic acid, oleic acid etc. The process of saponification involves the hydrolysis of an oil or fat with an alkali (sodium hydroxide or potassium hydroxide). Oils and fats are glyceryl esters of fatty acids and may be regarded as mixed glycerides. Sodium soaps are prepared by heating the fats by sodium hydroxide.
(i) (C15H31COO)3C3H5 – Glyceryl palmitate
(ii) (C17H32COO)3C3H5 – Glyceryl oleate
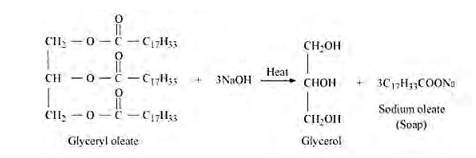
New Question
11 months agoNew Question
11 months agoContributor-Level 10
16.3
Diabetes mellitus is the third leading cause of death in the world. It is a medical condition in which the glucose is not consumed in the body due to inefficiency in the secretion of insulin [hormone which converts glucose to glycogen and store in liver]. Diabetes is the precursor for many other diseases like obesity, heart attacks, stroke etc.
Therefore Diabetic people are advised to consume low-calorie diet [fewer carbohydrates & more proteinaceous and fiber rich]. The refined sugar like sucrose adds calorie to the diet but complex sugar and starches do not add to the calorie intake of a person and at the same time impart a
New Question
11 months agoContributor-Level 10
Given:
Molar mass of non-volatile solute = 40g
Let no. of moles of solute be n.
Mass of octane = 114g
Molar mass of octane (C8H18) = 12 × 8 + 1 × 18 = 114g/mol
Moles of octane = given mass/molar mass
⇒ n = 114/114 moles
⇒ n = 1 mole
Molar fraction of solute,
x2 = moles of solute / moles of solute + moles of octane
⇒ x2 = n/n + 1
Let the vapour pressure of original solvent (without solute) be p1?
Accordingly after addition of solute vapour pressure of solution reduces to 80% i.e.
0.8 p1? = p1
Applying the formula:

⇒ n/n + 1 = 0.2
⇒ 0.2n + 0.2 = n
⇒ n = 0.25 moles
Hence, mass of solute is:
moles = given mas
New Question
11 months agoContributor-Level 10
The fees for 2-year MBA in Jaypee Business School is around INR 11 Lakh. The MBA fee might include multiple components that are to be paid during the admission period. The application form for the Jaypee Institute of Information Technology is available online on the official website of the institute. In addition to the course fees, applicants are required to pay a JSB Noida application fee of INR 1,000. For those interested in hostel accommodations, a hostel fee ranging from INR 4 Lacs is required to be paid while applying for the hostel.
NOTE: This information is sourced from the official website/ sanctioning body and i
New Question
11 months agoContributor-Level 10
Given: 1 molal solution means 1 mole of solute present in 1000g of water solvent)
Molecular weight of water = H2O = 1 × 2 + 16 = 18g/mol
No. of moles of water, n = given mass /molecular weight
⇒ n = 1000/18 = 55.56 gmol-1
Mole fraction of solute in solution, x2 = moles of solute/ (moles of solute + moles of water)
⇒ x2 = 1/ (1 + 55.56)
⇒ x2 = 0.0177
Given vapour pressure of pure water at 300k is 12.3 kpa
Apply the formula:
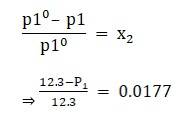
⇒ P1 = 12.0823kpa
which is the vapour pressure of the solution.
New Question
11 months agoContributor-Level 10
IIT Madras BTech admission is based on the JEE Main, followed by the JEE Advanced score. The centralised counselling process is managed and regulated by the JoSAA, based on which candidates are allotted seats. The seat allotment is based on the institute cut-off and a candidate's preference for courses and colleges. Given below is the BTech eligibility and selection criteria along with the admission details for the integrated BTech course:
| Course Name | Eligibility | Selection Criteria |
|---|---|---|
| BTech | Class 12 with a minimum of 75% aggregate in PCM | JEE Main + JEE Advanced + JoSAA Counselling |
| BTech + M.Tech (Dual degree) | Class 12 with a minimum of 75% aggregate in PCM | JEE Main + JEE Advanced + JoSAA Counselling |
New Question
11 months agoContributor-Level 10
The eligibility requirements to get admissions into BTech courses at Manipal University are that candidates must complete their Class 12 with a 50% aggregate with Physics, Mathematics and English as compulsory subjects along with chemistry/biotechnology/biology/any vocational subject as optional. Moreover, the university accepts valid MET scores for admissions to BTech courses. Moreover, the provision of direct admissions has not been mentioned on the official website of the university.
New Question
11 months agoContributor-Level 10
As of now, the BSc average package has not been released by the SRM University yet. However, student reviews present on Shiksha provide students' opinions on the placement scenario at the university. As per that, the highest package bagged by the BCom students during the recent placement drive stood at INR 57 LPA.
New Question
11 months agoContributor-Level 10
SRM University has not released the average package for the BSc department yet. However, the student reviews on Shiksha talk about the university's placement stats. As per the reviews, the average package offered to BSc students participating in the recent SRMU Andhra Pradesh placement drive was INR 7.9 LPA.
Note: The above-mentioned data is sources from student reviews.
New Question
11 months agoContributor-Level 10
Given: Temperature = 373k
Vapour pressure of pure heptane (p10 ) = 105.2 kpa and that of octane (p20 ) = 46.8 kpa
Mass of heptane = 26 g
Mass of octane = 35 g
Molecular weight of heptane = C7H16 = 12 × 7 + 1 × 16 = 100 gmol-1
Molecular weight of octane = C8H18 = 114 gmol-1
Moles of heptane, n1 = given mass /molecular weight = 26/100
⇒ n1 = 0.26mol
Moles of octane, n2 = given mass /molecular weight = 35/114
⇒ n2 = 0.307mol

∴ Partial pressure of heptane, p1 = χ1 × p10
⇒ p1 = 0.456 × 105.2 = 47.97kpa
∴ Partial pressure of octane, p1 = χ2 × p20
⇒ p2 = 0.544 × 46.8 = 25.46 kpa
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts