Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoNew Question
11 months agoContributor-Level 10
In case of MBA, the admission criteria of JBS Noida is completely entrance-based. The institute accepts various national-level entrance exams such as CAT, MAT, CMAT, XAT, CUET, etc. Apart from the aforementioned entrance exams, Jaypee Business School also accepts the in-house entrance test conducted by Jaypee Institute of Information Technology, called JIIT UGET, for admission to the BBA course. Therefore, candidates cannot get direct admissio to MBA course at Jaypee Business School.
New Question
11 months agoContributor-Level 10
16.22
Artificial sweetening agents refer to those compounds which impart sweet taste to any food product but at the same, they do not add any calories to the body. Example: Aspartame, Alitame, Saccharin etc.
New Question
11 months agoContributor-Level 10
The Ptotal for the values given in the graph is found out and plotted in the graph.
ptotal (mm Hg) | 632.8 | 603.0 | 579.5 | 562.1 | 580.4 | 599.5 | 615.3 | 641.8 |

It can be observed from the graph that the plot for the p total of the solution curves downwards. Therefore, the solution shows negative deviation from the ideal behaviour.
New Question
11 months agoContributor-Level 10
Some topics in BSc Physiology may be tougher due to their complexity and technical depth.
Neurophysiology
Endocrinology
Molecular biology
Biochemistry
Advanced human anatomy
New Question
11 months agoContributor-Level 10
16.21
At elevated temperatures, aspartame is unstable and break down to give a tasteless compound because of which aspartame is limited to cold foods and drinks.
Aspartame is methyl ester of the dipeptide obtained from phenylalanine and aspartic acid. It is about 180 times as sweet as cane sugar. The structure of aspartame is as follows:
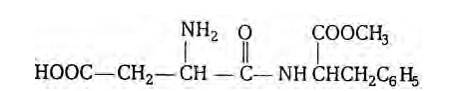
New Question
11 months agoContributor-Level 10
Most colleges ask for a standard set of academic and identity documents for BSc Physiology admission.
Class 10 & 12 mark sheets
Transfer and Migration certificates
Birth certificate or Age proof
Passport-size photographs
Category/Caste certificate (if applicable)
Aadhaar card or other ID proof
New Question
11 months agoContributor-Level 10
Given-
Mass of liquid A, WA = 100g, Molar mass, MA = 140 g mol-1
Mass of liquid B, WB = 1000 g, Molar mass, MB = 180 g mol-1
Using the formula below calculate the no. of moles in liquid A and B.
Number of moles = Mass / Molar Mass
Number of moles of liquid A, MA = 100/140 = 0.714 mol-1
Number of moles of liquid B, MB = 1000/ 180 = 5.556 mol-1
Using the formula,
mole fraction of a liquid = No. of moles of the liquid / total no of moles
we calculate the mole fraction of liquids A and B.
→ Mole fraction of A,
xA = 0.714 / (0.714 + 5.556)
∴ xA = 0.114
→ Mole fraction of B,
xB = 1- xA = 1 - 0.114
∴ xB = 0.886
Vapour pressure of p
New Question
11 months agoContributor-Level 10
16.20
Food preservatives are chemicals that prevent food from spoilage due to microbial growth. Table salt, sugar, vegetable oil, sodium benzoate (C6H3COONa), and salts of propanoic acids are some common examples of food preservatives.
New Question
11 months agoContributor-Level 10
Given-
Henry's law constant KH = 4.27X 105 mm Hg,
p = 760mm Hg,
Using Henry's law,

Using the formula of lowering vapour pressure,

Thus, the solubility of methane in benzene is 0.023 moles
New Question
11 months agoContributor-Level 10
Given- Vapour pressure of water,
PA0 = 17.535 mm Hg
WB= 25 g of glucose
WA = 450g of water
Molar mass of water, H2O = 1 + 1 + 16 = 18 g mol-1
Molar mass of glucose, C6H12O6 = (12*6) + (1*12) + (16*6) = 180 g mol-1
Using Raoult's law for solution of non-volatile solute,
PA0 - PA / PA0 = xB? Equation 1
where xB is the mole fraction of the solute
xB = WB/MB X MB/WB
=25/180 X 18 / 450
xB = 1/180
Substituting the value of xB in equation 1, we get,

Thus, the vapour pressure of water at 293 K at the given conditions is 17.437 mm Hg
New Question
11 months agoContributor-Level 10
If you are unsre about pursuing BSc Physiology then ask yourself these questions to reflect on your genuine interest:
You Enjoy Learning About the Human Body
You find systems like the nervous, respiratory, or cardiovascular fascinating.You're Curious About How Diseases Affect the Body
You want to know how body functions change in illness or trauma.You've Done Well in Biology and Chemistry
Academic performance in these subjects shows your compatibility with this field.You Want to Help Advance Medical Knowledge
You're more drawn to behind-the-scenes healthcare and research than direct patient care.You've Explored Career Paths Like Lab Researc
New Question
11 months agoContributor-Level 10
Given- w1 = 500g
W2 = 19.5g
Kf = 1.86 K kg mol-1
Molar mass of CH2FCOOH = 12 + 2 + 19 + 12 + 16 + 16 + 1
= 78 g mol-1
The depression in freezing point is calculated by,

→ (where, m is the molality)
= 1.86 X 19.5 / 78 X 1000/500
= 1.86 X 19.5 / 78 X 2
=0.93
∴ Δtf (calculated) = 0.93
To find out the vant Hoff’s factor, we use the formula,
i = observed Δtf / calculated Δtf
i = 1.0 (given) / 0.93
∴ i= 1.07
CH2FCOOH → CH2FCOO- + H +
To find out the degree of dissociation α, we use

Thus, the vant Hoff’s factor is 1.07 an the dissociation constant is 2.634x10-3
New Question
11 months agoContributor-Level 10
Jaypee Business School Noida, MBA admission is conducted via the entrance exam scores. To get admission in the MBA cousre, candidates can choose any one admission procedure as per their qualification. Candidates must follow the below steps for admission:
- Eligibility Criteria for MBA: Bachelor degree in any discipline with 50% (45% for SC/ST) aggregate marks or CGPA score and valid score of CAT/MAT/CMAT/XAT/GMAT/ATMA/CUET-PG. (Candidates graduating by 31 July can also apply).
- Application will be shortlisted based on valid exam's score.
- Shortlisted candidates will be called for Group Discussion and Personal interview.The fina
New Question
11 months agoContributor-Level 10
BSc Physiology builds scientific, analytical, and practical abilities essential for the healthcare and research fields.
Human anatomy and physiology understanding
Laboratory techniques and experimentation
Critical thinking and data analysis
Scientific writing and documentation
Research methodology and ethical practices
Interpersonal and communication skills for healthcare settings
New Question
11 months agoContributor-Level 8
The Bachelor of Computer Applications (BCA) program at Navajyothi College of Arts and Science, Cherupuzha, Kannur, has a total fee of ?1,80,000 for the 3-year duration, amounting to ?60,000 per year. Additional costs include application fees, exam fees, and library/technology fees.
New Question
11 months agoContributor-Level 10
16.19
Iodine is a powerful antiseptic. It is employed as tincture of iodine which is an alcohol-water solution containing 2-3 percent of iodine. Iodine is widely used in healing and treatment of wounds as it kills all the microbes present in the wounded region.
New Question
11 months agoContributor-Level 10
If you're considering BSc Physiology, it's important to know whether your interests and strengths align with the nature of this course.
Biology Enthusiasts
Students who are genuinely interested in how the human body functions at a cellular and systemic level.Detail-Oriented Learners
You enjoy diving deep into processes like respiration, circulation, and nervous system activities.Aspiring Researchers or Healthcare Professionals
Those aiming for careers in healthcare, lab research, or higher education in biosciences.Comfortable with Science Subjects
A strong foundation in Biology and Chemistry helps in understanding complex concepts easily.Cu
New Question
11 months agoContributor-Level 10
Mass of CH3CH2CHClCOOH = 10 g
Mass of water = 250g
Ka = 1.4 × 10–3,
Kf = 1.86 K kg mol–1
Molar mass of CH3CH2CHClCOOH = 12 + 3 + 12 + 2 + 12 + 1 + 35.5 + 2 + 16 + 16 + 1
= 122.5 g mol–1
Number of moles of solute = Mass of Solute / Molar Mass
→ No. of moles = 10g / 122.5 g/mol
∴ No. of moles = 8.6 X 10–2 mol
Now, Molality is given as,
M = Number of moles of solute / kg of solvent
M= 8.6 X 10–2 X 1000 g/mol / 250 g
M = 0.3264 kg/mol
CH3CH2CHClCOOH = CH3CH2CHClCOO- + H +
Initial moles | 1 | 0 | 0 |
Equilibrium moles |
(1-α) |
α |
α
|
Total moles at equilibrium = (1-α) + 2 α
= 1 + α
In order to find out the depression in freezing point,

values o
New Question
11 months agoContributor-Level 10
16.18
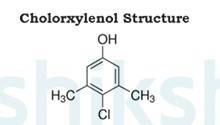
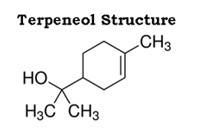
Dettol is a well known antiseptic. It is a mixture or combination of chloroxylenol and terpineol in a suitable solvent.
The structure of chloroxylenol is as follows:
The structure of terpineol is as follows:
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts






