Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
The depression in freezing point of water observed for the same amount of acetic acid, trichloroacetic acid and trifluoroacetic acid increases in the pattern,
Acetic acid< trichloroacetic acid< trifluoroacetic acid.
This is because fluorine is more electronegative than chlorine. So, trifluoracetic acid is a stronger acid in comparison to trichloroacetic acid and acetic acid. And also, acetic acid is the weakest of all.
Explanation: Stronger acid produces more number of ions, therefore it has more? Tf (depression in freezing point), hence lower freezing point. As the acidic strength increases, the acid gets more and more ionised.
Trifluoracetic acid ionizes to the largest extent. Hen
New Question
11 months agoContributor-Level 10
Volume of the solution = 250mL = 0.25L
Let the no. of moles of solute be n
Molarity = No. of moles of solute/volume of solution
⇒ 0.15 = n/0.25
⇒ n = 0.0375moles
Molar mass of C6H5OH = 6×12 + 5×1 + 16 + 1 = 94g
Moles = mass/molar mass
⇒ 0.0375 = m/94
Mass of benzoic acid required = 3.525g.
New Question
11 months agoContributor-Level 10
16.17
Phenol is one such particular substance that can act both as an antiseptic as well as a disinfectant depending upon the concentration of the solution used. An antiseptic is used to prevent the growth of germs in cuts and wounds.
For instance, a phenol of 0.2% concentration acts as antiseptic and phenol of concentration 1% acts as a disinfectant.
New Question
11 months agoContributor-Level 7
Yes, KKHSOU offers scholarships to students based on both need and merit basis. KKHSOU students belonging to reserved categories, such as Scheduled Caste (SC) and Scheduled Tribes (ST), are eligible for fee exemption or reimbursement from KKHSOU. However, all of it depends on the KKHSOU university scholarship rules and regulations. Additionally, all these students are eligible for government scholarships as well. Some of the KKHSOU scholarships are:
- Scholarship for Female Civil Service Aspirants
- Merit-Based Scholarships
- Financial Aid for Economically Weaker Sections
- SC/ ST Post-Matric Scholarship
- Minority Scholarship
- Scholarship for Persons
New Question
11 months agoContributor-Level 10
Molar mass of Nalorphene = 311g/mol
Now 1000g of solution contains 1.5 × 10-3 moles of Nalorphene (Molality of solution = moles of solute/mass of solvent (in kg)
⇒ 1.5 × 10-3 moles of Nalorphene = 1.5 × 10-3 × 311 = 0.4665g of Nalorphene
Therefore, total mass of the solution = (1000 + 0.4665) g
⇒ total mass = 1000.4665 g
This implies that the mass of the solution containing 0.4665 g of nalorphene is 1000.4665 g.
Therefore, mass of the solution containing 1.5 mg of nalorphene is:
Mass = 1000.4665 X 1.5 X 10-3 / 4.665 g
⇒ mass of solution containing required ions = 3.22 g
Hence, the mass of aqueous solution
New Question
11 months agoNew Question
11 months agoContributor-Level 10
16.16
Magnesium or Aluminium hydroxides do not tackle with the root cause of acidity that is they reduce the acidity by reacting with excess acid present in the stomach [neutralization reaction].
On the other hand cimetidine and ranitidine relieve acidity by preventing interaction of histamine with receptors of stomach walls and hence release of excess acid by stomach walls is stopped. So the root cause of acidity is totally eliminated.
Therefore cimetidine and ranitidine relieve the acidity by tackling the root cause whereas magnesium or aluminum hydroxide just relieves by healing only the mere cause of acidity.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
Total mass of solution = 6.5g + 450g = 456.5g
Therefore mass percentage of aspirin in solution = (mass of aspirine/total mass of solution) = 6.5/456.5
⇒ mass % of aspirine = 1.424%
New Question
11 months agoContributor-Level 10
16.15
Antiseptics and disinfectants are effective against micro-organisms. Antiseptics are applied to the living tissues such as wounds, cuts, ulcers, and diseased skin surfaces, while disinfectants are applied only to inanimate objects such as floors, drainage system, instruments, etc. Disinfectants are harmful to the living tissues. Iodine is an example of a strong antiseptic. Tincture of iodine (2 − 3 percent of solution of iodine in alcohol − water mixture) is applied to wounds. 1 percent solution of phenol is used as a disinfectant.
New Question
11 months agoContributor-Level 10
The Solubility product of CuS (ksp) = 6 × 10-16
CuS → Cu ++ + S2-
Let the s be solubility of CuS in mol/L
Ksp = [ Cu ++ ] [S2]
Ksp = solubility product
6 × 10-16 = s × s = s2
⇒ S = 2.45 × 10-8 mol/L
Hence, the maximum molarity of CuS in an aqueous solution is 2.45 × 10-8 mol/L
New Question
11 months agoContributor-Level 10
16.14
Antibiotics that are effective against a wide range of gram-positive and gram-negative bacteria are known as broad spectrum antibiotics. Chloramphenicol is an example of a broad spectrum antibiotic. It has the following structure: -
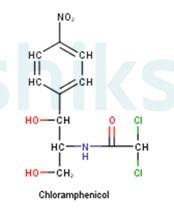
It can be used for the treatment of typhoid, dysentery, acute fever, pneumonia, meningitis, and certain forms of urinary infections. Two other examples of broad spectrum antibiotics are vancomycin and ofloxacin. Ampicillin and amoxicillin? synthetically modified from penicillin are also broad spectrum antibiotics.
New Question
11 months agoContributor-Level 10
Mass of ions = 92g
Molar mass of ions = Na+ = 23g (neglect the mass lost due to absence of a electron)
Moles of ions = mass of ions/molar mass
⇒ n = 92/23 moles
⇒ n = 4moles
Molality of solution = moles of solute/mass of solvent (in kg) Molality = 4/1 = 4M
New Question
11 months agoBeginner-Level 5
It is mandatory to save choices during the NEET SS counselling. If candidate does not lock the choice submitted, the choices saved by him/her will be automatically locked on the last date of choice locking. Also, once the choice locking deadline is over, candidates will not be allowed to modify the NEET SS counselling choices.
New Question
11 months agoContributor-Level 10
16.13
Anti-depressant drugs are needed to counteract the effect of depression. These drugs function by inhibiting the enzymes catalysing the degradation of the neurotransmitter, noradrenaline. As a result, the important neurotransmitter is slowly metabolised and then it can activate its receptor for longer periods of time.
Two anti-depressant drugs are:
(i) Iproniazid
(ii) Phenelzine
New Question
11 months agoContributor-Level 10
- Water is a polar compound (due to electronegativity difference between O and H) . We know that “like dissolves like”. So, a non-polar compound will be more soluble in non-polar solvent as compared to polar compound.
- Phenol has the polar group -OH and non-polar group –C6H5 and it can not form H bonding with water (presence of bulky non-polar group) . Thus, phenol is partially soluble in water
- Toluene has no polar Thus, toluene is insoluble in water.
- Formic acid (HCOOH) has the polar group -OH and can form H-bond with water. Thus, formic acid is highly soluble in water
- Ethylene glycol (OH-CH2-CH2-OH) has polar -OH group and can form H-
New Question
11 months agoContributor-Level 10
Now n-octane is non-polar solvent due to long chain saturated structure. We know that “like dissolves like” so a non-polar compound will be more soluble in non-polar solvent as compared to polar compound.
So cyclohexane is non-polar due to symmetric structure. KCl is ionic in nature as it will dissociate into K + and Cl- ions. CH3CN is polar as mentioned above and CH3OH is also polar in nature.
The order of increasing polarity is:
Cyclohexane < CH3CN < CH3OH < KCl (O is more electronegative than N)
Therefore, the order of increasing solubility is:
KCl < CH3OH < CH3CN < Cyclohexane
New Question
11 months agoContributor-Level 10
No, CIMAGE College admissions are primarily entrance-based. The college admits candidates based on a college-level Entrance-cum-Scholarship test. Those who clear the test are then further called in for personal interview. Those who clear both selection rounds in addition to fulfilling the eligibility are offered seats. However, students may visit the college's help desk to get more information about the admission chances.
New Question
11 months agoContributor-Level 10
16.12
Specific drugs affect only some specific or particular receptors. Antacids and anti-allergic drugs work on different receptors. This is the reason why antacids and anti-allergic drugs do not interfere with each other's functions, but interfere with the functions of histamines.
New Question
11 months agoContributor-Level 10
(i) Both the compounds are non-polar and they do not attract each other because they do not form any polar ions. Vanderwaals forces of attraction will be dominant in between them as vanderwaals forces of attraction are not a result of any chemical or electronic bond.
(ii) now here both the compounds are non-polar because in I2 both the atoms are same so they have same electronegativity and hence there will be no displacement of electron cloud, it will be in the centre. In case of CCl4 molecule, it has tetrahedral shape so two Cl atoms will cancel the attraction effect from two opposite Cl atoms, hence molecule as a whole is non polar.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
