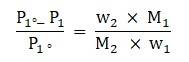Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months ago
Contributor-Level 10
Quacquarelli Symonds Rankings are known to be one of the top ranking bodies around the world and is known for publishing university rankings for various global universities. Some of the Quacquarelli Symonds world rankings of Carnegie Mellon University are given as follows:
| Year | Quacquarelli Symonds World Rank |
|---|---|
| 2024 | #52 |
| 2025 | #58 |
| 2026 | #52 |
New Question
11 months agoContributor-Level 10
With so many options available, it's important to consider a few key factors that can impact your academic experience and career path after BSc Physiology.
Following are the points to keep in mind while choosing the right colleges for BSc Physiology:
Check College Accreditation and Affiliation
Ensure the college is recognised by UGC or your state university and has a good academic reputation.Look at Faculty and Lab Facilities
Well-qualified faculty and fully equipped physiology labs are essential for practical learning and research exposure.Review the Curriculum and Electives
Go through the syllabus to see if it covers core physiology topic
New Question
11 months agoNew Question
11 months agoContributor-Level 10
Given:
2% non-volatile solute in an aqueous solution means that if the solution is of mass 100 g then the mass of solute in the solution is,
Mass of solute = [2 × 100]/100
= 2 g
∴ Mass of Solvent = 100-2
= 98 g
Vapour pressure of solution at normal boiling point, P1 = 1.004 bar
Vapour pressure of pure water at normal boiling point, P1° = 1 atm = 1.013 bar
To find: Molar mass of solute
Formula:
From Raoult’s Law, we have:
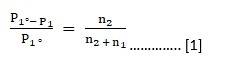
Now we know that number of moles of solute is given by the following relationship,
⇒ Number of moles = Mass of solute / Molecular mass of solute
Using the above relationship the equation [1] can be mod
New Question
11 months agoContributor-Level 6
A high ATS score or the Aptitude Test Score means higher chances of getting a Personal Interview call from IPMAT Indore. The ATS score is based on the performance of the students in the IPMAT exam. The ATS is calculated using the below-mentioned formula:
ATS Score = (Candidate's score / Maximum score) X 100
An ATS score is a defining factor in deciding the candidate's chances of being called for a Personal Interview.
New Question
11 months agoContributor-Level 10
Choosing the right course after 12th can be confusing. Here are a few key checkpoints to help you decide if BSc Physiology aligns with your interests and goals:
Strong Interest in Biology: You enjoy learning about how the human body works, especially systems like the brain, heart, muscles, and hormones.
Curious About Medical Research: You're fascinated by lab work, clinical studies, and want to explore how the body reacts to different conditions.
Aim for a Career in Healthcare or Research: You're planning for roles like physiologist, researcher, or preparing for advanced studies like MSc or medical fields.
Comfortable with Science Subjects
New Question
11 months agoContributor-Level 10
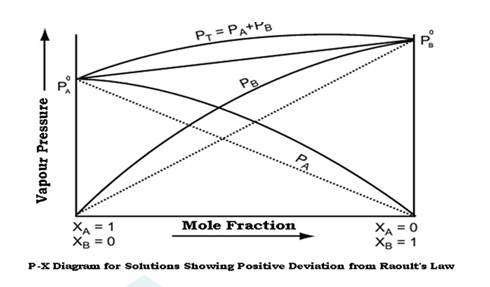
Raoult’s law states that at a given temperature, the vapour pressure of a solution containing non volatile solute is directly proportional the mole fraction of the solvent.
Non ideal solutions show positive and negative deviations from ideal behaviour.
Non ideal solutions showing positive deviations from Raoult’s law-
A solution is said to show positive deviation from Raoult’s Law when at any composition, its vapour pressure is higher than that given by Raoult’s Law.
The positive deviation is shown by those liquid pairs in which the A-B molecular interaction forces are weaker than the corresponding A-A or B-B molecu
New Question
11 months agoContributor-Level 10
The BA graduates of Cluster University looking for government jobs must have the required skill set. To secure a government job, degree holders have to go through the preset selection procedure. These candidates can also take government entrance examinations. Some of the popular government job profiles among BA passouts are:
- IAS
- IPS
- SSC officer
- Bank PO
- Social Worker, etc.
New Question
11 months agoContributor-Level 10
Given:
Mass of ethane in solution A = 6.56 × 10–3 g
Partial pressure of solution A = 1 bar
Mass of ethane in solution B = 5.00 × 10–2 g
To find: Partial Pressure of gas
Formula:
By Henry’s law:
Mass of dissolved gas M = k × P Where
k = proportionality constant
P = Partial Pressure
Solution:
⇒ M1 = k × P1. [1]
⇒ M2 = k × P2. [2]
Dividing the [2] by [1], we get
M2/M1 = P2/P1
P2 = M2 X P1 / M1
P2 = 5 X 10–2 X 1 / 6.56 X 10–3
P2 = 7.62 bar
Therefore the partial pressure of the gas is 7.62 bar.
New Question
11 months agoContributor-Level 10
The solubility of a gas in water depends on following three parameters:
- Nature of gas
- Temperature
- Pressure
The solubility decreases with increase in temperature. Temperature and pressure follow inverse proportionality. So solubility increases with increase with pressure. A quantitative relation between pressure and solubility of a gas in a solvent was given by W. Henry [1803]. This relationship is known as Henry's law.
Statement:
Henry's law can be expressed as follows.
At constant temperature, the solubility of a gas in a liquid is directly proportional to the pressure of the gas.
Mathematically,
Solubility? Pressure of the gas
Some of the imp
New Question
11 months agoContributor-Level 10
Whenever a gas is dissolved in a liquid, a small amount heat is liberated in the process. So dissolving a gas in liquid is overall an exothermic process.
So according to the LeChatelier principle, whenever the temperature is increased for a reaction which is exothermic in nature, the equilibrium shifts backwards and the reaction proceeds in backward direction that means the solution gets dissociated and will give off gas and hence solubility of gas decreases.
So with the increase in temperature, the solubility of the gases in liquids decreases.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
The lower members of alcohols are completely miscible [highly soluble] with water but the solubility decreases with increase in the molecular weight. The lower members of the alcohol group have the capability to form intermolecular hydrogen bonding with water molecules as alcohols are polar molecules in nature.
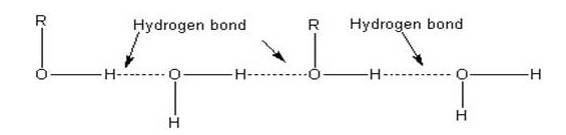
Alkyl groups are hydrophobic [prevents formation of hydrogen bonds with water] in nature. In lower alcohols, the alkyl group is small and the –OH group of alcohol is effective in making hydrogen bonds with water.
But with the increase in the size of alkyl group, the hydrophobic [water hating] nature of alkyl group dominates over
New Question
11 months agoContributor-Level 10
16.2
Basically drugs can be classified in a number of ways. For example classification on basis of their chemical structure, classification on the basis of their action etc.
In the above statement, the classification is based on the pharmacological effect of drugs on the human body.
Antacids refer to those class of drugs which relieve the acidity by either reacting with the excess acid in the stomach [eg – magnesium hydroxide] or by preventing the secretion of an excess of acids by the stomach cells [eg – ranitidine].
New Question
11 months agoContributor-Level 10
Given:
Level of contamination = 15 ppm [by mass]
To find: Mass Percentage and Molality
Formula:
Molality = Number of moles of solute / Mass of solvent in kg
Mass Percentage of Solute = Mass of solute / Mass of solution X 100
Solution:
Calculation of Mass Percentage:
15 ppm means 15 parts of Chloroform in 106 parts of drinking water
⇒ Mass Percentage = Mass of choloroform / Total mass X 100
= 15 / 106 X 100
= 1.5 × 10-3
Calculation of Molality:
⇒ Molecular Mass of Chloroform, CHCl3 = [12] + [1] + [35.5 × 3]
= 119.5 g
⇒ Number of Moles of Chloroform = [15 / 119.5]
= 0.1255 moles
Molality = Number of moles of solute
New Question
11 months agoContributor-Level 10
Cluster University Srinagar offers BA and BA (Hons) courses across different specialisations. The mode of study is full-time for all these programmes. Admission is granted based on merit in CUET. Mentioned below are some of the BA and BA (H) courses offered by the university:
- BA in Psychology
- BA (H) in Islamic Studies
- BA in Political Science
- BA0 (H) in Economics, etc.
New Question
11 months ago
Beginner-Level 4
To be eligible for the PG Diploma at Vinayak Ganesh Vaze College, candidates must hold a bachelor's degree.
The college prefers graduates with a background in B.Sc./M.Sc., B.Pharm/M.Pharm, or B.Tech/M.Tech with Chemistry as a major or subsidiary subject.
Professionals with three years of relevant experience are also considered.
New Question
11 months agoContributor-Level 10
Given:
Mass of ethylene glycol (C2H6O2) = 222.6 g
Mass of water = 200 g
Density, d = 1.072 g/ml
To find: Molality and Molarity of solution
Formula:
Molality = Number of moles of solute/ Mass of solvent in kg
Molarity, Mo = number of moles of solute/ volume of solution in litres
Density, d = Mass (M) / volume (V)
Calculation of Molality:
⇒ Molecular Mass of ethylene glycol (C2H6O2) = [12 × 2] + [6 × 1] + [16 × 2]
= 24 + 6 + 32
= 62 g
⇒ Number of moles of ethylene glycol (C2H6O2) = [222.6/62]
= 3.59 moles
⇒ Mass of water = 200 g
⇒ Molality = numebr of moles of solute/ Mass of solvent in kg
= 3.59 / 200 X 1000
= 17.95
New Question
11 months agoNew Question
11 months agoContributor-Level 10
16.1
Sleeping pills belong to tranquilizers class of drugs. Sleeping pills induce artificial sleep in people suffering from insomnia by depressing the activities of the central nervous system [brain and spinal cord].
When such drugs are taken for a prolonged time, such drugs can become addictive and the person can become addicted to such drugs and the aftereffects of such drugs are drowsiness, hallucinations, slow heart rate etc. and in extreme cases, the person can enter a state of coma and death can also take place.
Therefore it is highly recommended that sleeping pills should be administered to people suffering from insomnia but
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts