Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
8.13 The oxidation states displayed by the first half of the first row of transition metals are given in the table below.
METALS | Sc ( [Ar] 3d14s2) | Ti ( [Ar] 3d24s2) | V ( [Ar] 3d34s2 ) | Cr ( [Ar] 3d54s1) | Mn ( [Ar] 3d54s2) |
OXIDATION STATES |
| +2 | +2 | +2 | +2 |
+3 | +3 | +3 | +3 | +3 | |
| +4 | +4 | +4 | +4 | |
|
| +5 | +5 | +6 | |
|
|
| +6 | +7 |
Except for Sc, all others metals display +2 oxidation state. This is because as the atomic number increases, the number of electrons in the valence shell increases. +2 oxidation state is attained by the loss of the two 4s electrons by these metals. As the number of electron increases, the possibility of an ion with +2 oxidation state being stable (by attaining half-filled structure) also increases. Finally, Mn2+ ions have half-filled structure and are very stable.
New Question
11 months agoContributor-Level 10
5.34
Alcohol- a colloidal solution having alcohol as the dispersion medium and a solid substance as the dispersed Ex- colloidal sol of cellulose nitrate in ethyl alcohol.
Aerosol- a colloidal solution having gas as the dispersion medium and a solid substance as the dispersed Ex-Smoke.
Hydrosol- a colloidal solution having Water as the dispersion medium and a solid substance as the dispersed Ex-Gold sol.
New Question
11 months agoContributor-Level 10
1- When 3-methylaniline treated with NaNO2 + HCl it gets converted into chlorine complex.
When that complex reacted with HBF4 It gets converted into Barium Fluoride complex. This complex reacts with NaNO2 in presence of copper to give 3-Nitrotoluene.
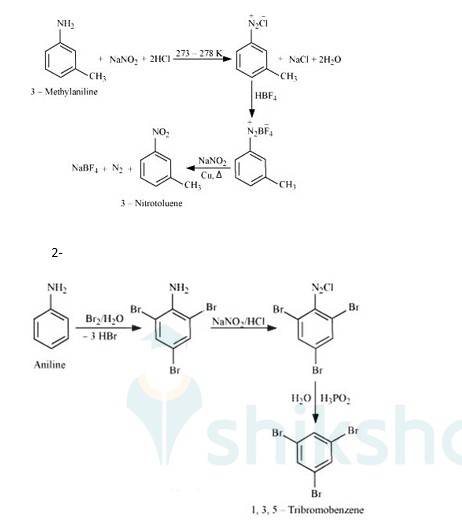
When aniline reacts with Br2 water it gets converted into 2,4,6 tribromobenzamine. When this further reacted with NaNO2/HCl it forms Chloride complex. This complex forms 1,3,5 tribromobenzene after treating with H3PO2 in presence of water.
New Question
11 months agoContributor-Level 10
For admission to the MBA course at Swaminarayan University, students need to fulfil the minimum eligibility criteria in the first place. The selection criteria for this course is merit-based. The eligibility criteria for this course are to pass graduation with a 50% aggregate. The duration of the course is two years, spread over four semesters.
New Question
11 months agoContributor-Level 10
8.12 Electronic configuration of Mn2+ is [Ar]183d5 and Electronic configuration of Fe2+ is [Ar]18 3d6 . It is known that half-filled and fully-filled orbitals are more stable. Therefore, Mn in (+2) state has a half-filled stable configuration, whereas the Fe in +3 oxidation state has partially filled subshells, which are relatively unstable. This is the reason Mn2+ shows resistance to oxidation to Mn3+. Also, Fe2+ has 3d6 configuration and by losing one electron, it attains half- filled stable Hence, Mn2+ compounds more stable than Fe2+ towards oxidation to their +3 state.
Mn+2→
Manganese has the atomic no. 25 and its electronic con
New Question
11 months agoContributor-Level 10
The different isomers of the molecular formula: C3H9N are given in the table. However only 1° amines will liberate nitrogen gas on the treatment with h=the nitrous acid are given as follows:
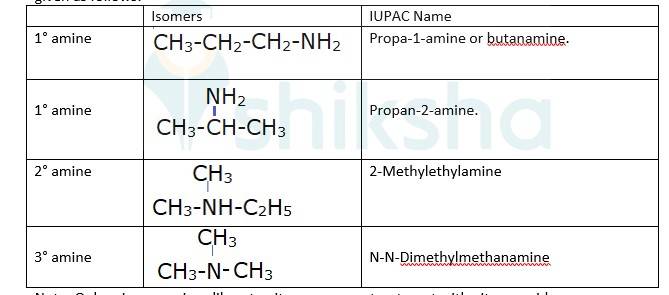
Note: Only primary amines liberate nitrogen gas on treatment with nitrous acid.
New Question
11 months agoContributor-Level 10
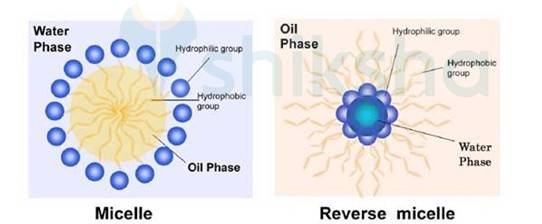
5.33
Soaps contain hydrophobic and hydrophilic part when dissolved in the water they arrange themselves in such a way that they form a spherical structure having hydrophobic part towards the centre and hydrophilic part away from centre. This cluster is known as Micelle. Ex-Sodium stearate + Water
(CH3 (CH2)16COO-Na + H2O)
New Question
11 months agoContributor-Level 10
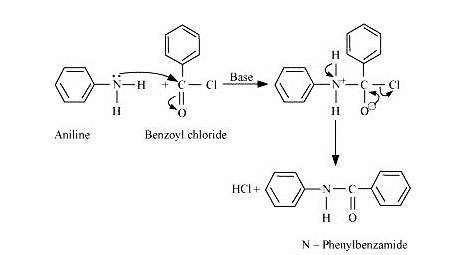
When aniline is treated with benzoyl chloride in the presence of base it gets converted into N-Phenylbenzamide.
New Question
11 months agoContributor-Level 10
For admission in a BCom course of PES University, it is crucial that the aspirants fulfil the course-specific eligibility requirements. Aspirants who are Class 12 or equivalent exam pass with at least 50% aggregate can apply for admission. The qualifying exam should have been passed from a recognised board. Moreover, the aspirants must hold the relevant documents during the application procedure and course admission.
New Question
11 months agoContributor-Level 10
Unitec Institute of Technology does not specify a minimum GPA requirement for international students applying to an undergraduate or postgraduate programme but does offer overall academic requirements. The requirements are different for different countries. For UG programs, the student must have high school graduation certificate with a min of 70% in at least 4 academic subjects. For PG programs applicants must have bachelor's degree in the relevant field from a recognised university, with an average grade of around B- or above. There can be some programme specific requirements hence it is recommended to refer the official programme pa
New Question
11 months agoContributor-Level 10
For MBA course admission, candidates must meet the eligibility criteria set by Swaminarayan University. Aspirants should pass graduation with a minimum 50% aggregate. The admission criteria for the course is merit-based. The Full Form of MBA is Master of Business Administration which is a two-year PG degree designed to equip students with advanced skills and knowledge in business management and leadership.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
5.32
Uses of Emulsions-
It is used in making of medicines,
Cleansing action of soaps is based on this emulsion
Digestion of fats in intestine takes place by the process of
Antiseptics and disinfectant added to water form emulsion for
New Question
11 months agoContributor-Level 10
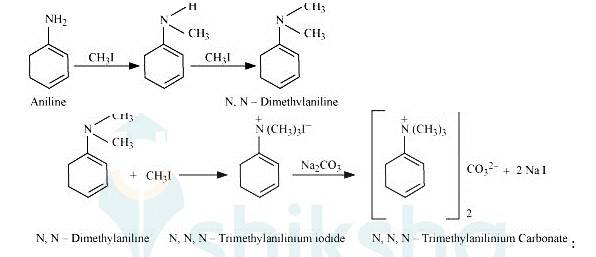
On excessive alkylation with methyl iodide aniline gets converted into N, N-Trimethylanilinium iodide. After reacting it with sodium carbonate it get converted into N, N-Trimethylanilinium carbonate.
New Question
11 months agoContributor-Level 10
5.31
Electrophoresis: The movement of colloidal particles under an applied electric potential is called When an electric potential is applied to two platinum electrodes dipping in colloidal solutions the colloidal particle move towards the Oppositely charged electrodes.
Coagulation: The process of settling of colloidal particles is called coagulation. When the charge is removed from colloidal solution somehow, then particle start coagulation and settling due to the force of
Dialysis: It is a process of removing a dissolved substance from AC colloidal solution by means of diffusion through a suitable the animal membrane or parchment paper
New Question
11 months agoContributor-Level 10
Unitec Institute of Technology consist of a diverse student community of over 20,000 individuals. It offers a multicultural environment to international students, welcoming applicants from more than 80 different countries across the world. The university provides over 150 programs in both undergraduate and postgraduate levels, in a variety of fields like Applied Science, Technology, Engineering, Business, Health, and Social Sciences. The acceptance rate of Unitec Institute of Technology has been around 91% over the years, which shows easier admissions. The university has strong student support groups to help international students thro
New Question
11 months agoContributor-Level 10
5.30
The catalytic reaction that depends upon the pore structures of the catalysts and the size of reactant and product molecules is called shape selective catalysis. Zeolites are good shape- selective catalysts.
New Question
11 months ago
Contributor-Level 10
In the 2025 Andhra Pradesh EAMCET, GMR Institute of Technology (GMRIT) Rajam is considered to be a good option for students with a rank between 6,000 and 8,000, based on the Andhra Pradesh EAMCET marks vs rank analysis. For B.Tech in Computer Science and Engineering (CSE), the closing rank in Round 1 was 6765 according to Shiksha. For ECE, the closing rank in Round 1 was 16085.
GMRIT NC (Non-Constituent) Ranking:
B.Tech in CSE:
GMRIT Rajam's cutoff rank for B.Tech in Computer Science and Engineering (CSE) is around 7,000 as per Shiksha.
B.Tech in ECE:
The closing rank for B.Tech in Electronics and Communication Engineering (ECE)
New Question
11 months agoContributor-Level 10
8.11 (i) The atomic number of Cr is 24 and the electronic configuration is [Ar] 3d54s1. When 3 electrons are removed, it becomes Cr3+. The electronic configuration of Cr3+: 1s2 2s2 2p6 3s2 3p6 3d3 Or [Ar]3d3
(ii) The atomic number of Pm is 61 and the electronic configuration is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f5 5s2 5p6 6s2 or [Xe] 4f56s2. When 3 electrons are removed, it becomes Pm+3, having the electronic configuration,
Pm3+: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f4
Or, [Xe]544f4
(iii) The atomic number of Cu is 29 and has the electronic configuration of [Ar] 3d104s1. On removing one electron, the Cu+
New Question
11 months agoRegister to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts
