Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
5.29
Catalysis by zeolites is dependent on shape. Because zeolites are shape-selective catalysts. They are alumino silicates which are microporous in nature. It has Honeycomb structure. That makes them shape selective. In zeolites, some si atoms are replaced by Al to form Al-O-Si network.
Reactants are very sensitive to the pore size of zeolites. Zeolites are used in petrochemical industry. Ex- ZSM-5 used to convert alcohol into gasoline.
New Question
11 months agoContributor-Level 10
(i) CH3CH2CH2NH2 + HCl → CH3CH2CH2N+H3Cl-
The final product is (N-propyl ammonium chloride.)
(ii) (C2H5)3N + HCl → (C2H5)3N+HCl-
The final product is (Tri ethyl ammonium chloride)
New Question
11 months agoContributor-Level 10
8.10 The 5f orbitals (in actinoids) have a poorer shielding effect than 4f orbitals (in lanthanoids). Thus, the effective nuclear charge experienced by electrons in outer shells in case of actinoids is much more that experienced by lanthanoids.
As the effective nuclear charge experienced is high, the electrons are attracted with much force, hence the size of the atom decreases. Hence, actinoid contraction is greater from element to element than lanthanoid contraction.
New Question
11 months agoContributor-Level 10
5.28
Activity - It is the ability of the catalyst to accelerate the Reaction. It mostly depends upon the Chemisorption strength.
Selectivity - It is an ability to direct reaction to yield of a particular product i.e., One catalyst cannot be a catalyst for other reactions.
New Question
11 months agoContributor-Level 10
1- Alkyl group contribute inductive effect which increases the basic strength of
NH3
Then C6H5NH2 is having –I effect that reduces strength. And C6H5CH2NH2 increases the basic strength but not as much as C2H5 group.
Hence final order will be C6H5NH2<
2- By taking into consideration –R effect and steric hindrance of groups we can arrange them in the order
C6H5NH2< C2H5NH2< (C2H5)3N< (C2H5)2NH.
Because (C2H5)3N has a lot of steric hindrances that reduces the basic strength.
3- In C6H5NH2, N is directly attached to the ring that causes delocalization of electrons of the benzene ring. Whereas in case of C6H5CH2NH2 it is not directly connected to benze
New Question
11 months agoContributor-Level 10
Securing admission into Unitec Institute of Technology in New Zealand is easier based on academic record and English proficiency. The institution's acceptance rate has been about 91% over the past years. Documents that are usually required are certified academic transcripts, English proficiency proof, and identification documents. The institute has different admission requirements for different countries. Following are the requirements for students applying admissions:
- For undergraduate programs, student must have the All India Senior School Certificate (10+2) or an equivalent.
- For post graduate programs students must have bachelor's deg
New Question
11 months agoContributor-Level 10
8.9 The stability in aqueous condition depends on the hydration energy of the ions when they bond to the water molecules. And, the hydration energy is the amount of heat released as an ionic substance is dissolved and its constituent ions are hydrated or surrounded by water molecules.
Now, in Cu2+ and Cu+ ion, Cu2+ has a greater charge density than the Cu+ ion and so forms much stronger bonds releasing more energy. Therefore, in an aqueous medium, Cu2+ ion is more stable than Cu+ ion. This is because the energy required to remove one electron from Cu+ to Cu2+, is compensated by the high hydration energy of Cu2+.
New Question
11 months agoContributor-Level 10
The MBA eligibility requires candidates to have completed their graduation in any field (Science, Commerce, Arts) with an aggregate of 50% or more. Students can also check the list of reasons below to pursue MBA course from Swaminarayan University:
- Promising Career: The simplest approach to advance your career is to earn a Master of Business Administration. Students concentrate on the employment possibilities that follow earning a Master of Business Administration. MBA courses are becoming increasingly important to have managerial abilities and expertise as the globe becomes more and more competitive.
- Develop Manage
New Question
11 months agoContributor-Level 10
Benzene into aniline
When Benzene is treated with HNO3/H2SO4 it forms nitrobenzene. When Nitrobenzene reduced with Sn/HCL it forms Aniline because Sn/HCl is a reducing Mixture.
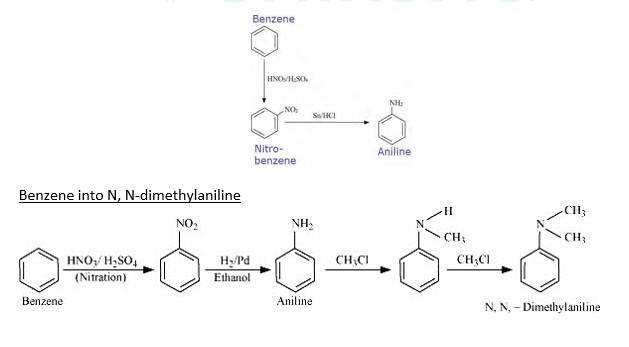
When Benzene is reacted with nitrating mixture it forms nitrobenzene. When it Reduced H2/Pd in ethanol or Sn/HCl, it forms Aniline. When Aniline reacts 2 times with CH3Cl It forms N, N- dimethylaniline.
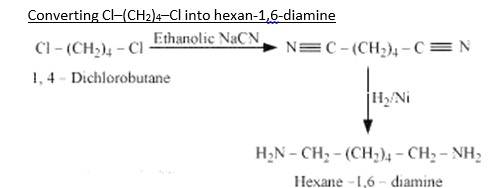
When 1,4-dichlorobutane reacts with NaCN it forms Di cyanide compound, After Hydrogenation it forms the Hexane 1,6-Diamine.
New Question
11 months ago
Contributor-Level 10
MBA course is a two-year postgraduate level degree program focusing on Management, Business, and Entrepreneurial concepts. Students can check the list of career options available after completing MBA from Swaminarayan University:
| Job Profiles | Description | Average Salary (in INR) |
|---|---|---|
| Investment Analyst | A Business Analyst is a professional who works within an organisation to analyze, understand, and document the business processes and systems. | 9.5 LPA |
| Investment Banker | An Investment Banker is a finance professional who works within an investment bank or a financial institution, specializing in providing financial advisory services and raising capital for corporations, governments, and other entities. | 5 LPA |
| Chief Financial Officer | The CFO is a high-level executive responsible for managing an organization's financial actions, strategies, and operations. | 47.1 LPA |
New Question
11 months agoContributor-Level 10
8.8 Given, Z=atomic number = 27 = [Ar]3d7 4s2
⇒ M2+ = [Ar]3d7
Hence, 3 unpaired electrons are present.
The spin only magnetic moment μ= √n(n+2)
Where n is the number of unpaired electrons.
Hence, μ =√(3×(3+2))
μ = √15 BM or μ = 3.87 BM
New Question
11 months agoContributor-Level 10
(i) & (ii) There are total 8 geometrical isomers of the given compound.
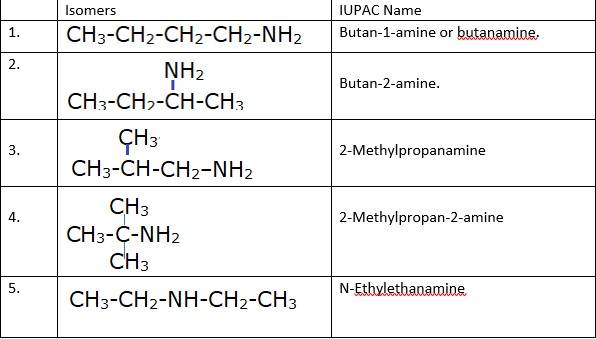
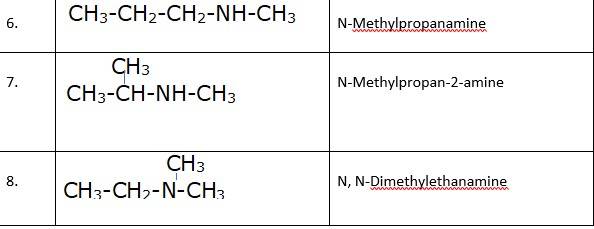
(iii) a) Pairs 1,2,6,7 Exhibit Position isomerism; means the change in position of the substituent.
b) Pairs 1,3 and 1,4 and 2,3 and 2,4 exhibit chain isomerism i.e. in this type of isomerism the different structures can be produced by changing the chain of the
c) Pairs 5,6 and 5,7 exhibit metamerism; e. different group on either side of the central atom.
d) All Primary amines exhibit functional isomers. All secondary amines share functional isomerism and same for The functional isomerism means same functional group.
New Question
11 months agoContributor-Level 10
5.27
Oxidation of sulphur dioxide into sulphur trioxide in the presence of
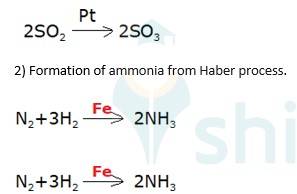
(3) Oxidation of ammonia into nitric acid in presence of platinum gauze in Ostwald's process
→ 4NH3 (g)+ 5O2 (g) Pt (s) 4NO +6H2O
(4) Hydrogenation of vegetable oils in presence of Nickel as catalyst
Vegetable oil (l)+H2 (g) Ni → Vegetable ghee (s)
New Question
11 months agoContributor-Level 10
8.7 The following reactions are involved when Cr2+ and Fe2+ act as reducing
Cr2+ = Cr3+ ( E? Cr3+/ Cr2+ = - 0.41 V)
Fe2+⇒ Fe3+ (E? Fe3+/ Fe2+ = +0.77 V)
Since, Cr has less potential value, Cr2+ gets oxidised easily than Fe2+. Therefore, Cr2+ is a better reducing agent that Fe3+.
New Question
11 months agoContributor-Level 10
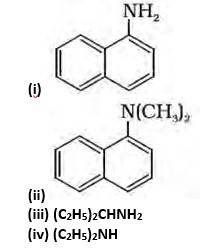
(ii) Tertiary, because the Nitrogen atom is attached to the 3 carbon atoms.
(iii) Primary, because the nitrogen atom is attached to the only 1 carbon atom.
(iv) Secondary, because the nitrogen atom is attached to the only 1 carbon.
New Question
11 months agoContributor-Level 10
Swaminarayan University application forms are also available online. Candidates meeting the eligibility criteria can apply for the preferred course. Below are the steps to apply for admission:
Step 1: Visit the official SU website (admission.swaminarayanuniversity.ac.in/) and click on the 'Apply Now' tab to register as a new user. In the registration form, provide details such as name, email ID, and password.
Step 2: Verify the verification email sent to the registered email address.
Step 3: Once verified, candidates will receive an email on the registered email with the user ID and password.
Step 4: Log in to the admission
New Question
11 months agoContributor-Level 10
5.26
Soap is sodium or potassium salts of fatty acids (long chain). Represented as RCOO–Na+. When dissolved in Water it dissociates in the RCOO– and Na+. In first ion there are two parts R and COO–. R is hydrophobic tail and COO– Hydrophilic head. RCOO– ions present at the surface with COO– group inside and R group outside.
Increasing concentration to critical micelle concentration anions are pulled inside the bulk and Form a spherical shape of hydrocarbon chain pointing towards the centre and COO– pointing outside this are called as micelle.
Cleansing action of soap: Soap molecules form micelle around oil droplets. So
New Question
11 months agoContributor-Level 10
In order to get admission at Swaminarayan University candidates must keep certain documents ready. Student can check the list of documents below:
- Scanned Class 10, Class 12 and graduation marksheet
- Leaving Certificate
- Aadhaar Card
- Passport-size photograph
- Signature
Note: The list of documents mentioned above are the general documents. For more details visit the official website.
New Question
11 months agoContributor-Level 10
8.6 The oxidation state increases when an atom loses its Example: When Fe loses 2 electrons, its oxidation state becomes +2 from 0.
Oxygen (O) and fluorine (F) are very strong oxidizing agents. Both oxide and fluoride ions are highly electronegative and have a very small size, so they attract the electrons from metal atoms. Hence, they oxidize the metal to its the highest oxidation state.
New Question
11 months agoContributor-Level 9
According to the official statistics for Fall 2024, over 82,000 applications were received by the University of Southern California, of which more than 8,000 students were admitted, and only 3,489 got enrolled. Out of the total enrollment, 26.6% of students were international. Students can check the enrollment statistics and ethnicity breakdown for the Fall admission below:
Particulars | Headcount 2024 |
|---|---|
Fall Applicants | 82,027 |
Fall Admits | 8,050 (9.8% admitted) |
Fall Enrolls | 3,489 (43.3% yield) |
Ethnicity Breakdown:
Ethnicity | Percentage of students |
|---|---|
Asian | 19.8% |
Black/African American | 6.1% |
Hispanic | 17.1% |
White/Caucasian | 21.9% |
International | 26.6% |
Other | 8.5% |
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts