Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
For admission in a MBBS programme at Swaminarayan University, it is crucial that the aspirant fulfills the course-specific eligibility criteria. Thus, only the aspirants who have passed Class 12 with at least 50% aggregate are eligible for admission. In addition, student should have passed the qualifying examination from a recognised board. Candidate must also hold the Class 12 mark sheet during application form filling and at the time of admission for verification purposes.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
Amity University, Kolkata has had decent placement opportunities over the years. The students are given excellent placement support every year by placing them in leading MNCs. Check out the table below to know the key highlights of the Amity University, Kolkata placements for BTech CSE:
Particulars | Placement Statistics (2023) |
|---|---|
the highest Package | INR 32.21 LPA |
Top Recruiters | Amazon, TCS, Cognizant, PWC, etc. |
Note- The institution has not released the placement report for 2024, once released it will be updated here.
New Question
11 months agoContributor-Level 10
7.1
Pentahalides (like MX5) means the compounds in which metal is bonded with five halogen atoms. Thus, the oxidation state of metal here is +5. Similarly, trihalides (like MX3) means the compounds in which metal is bonded with three halogen atoms.
Thus, the oxidation state of metal here is +3. Now, as the polarizing power is directly proportional to the charge, the metals with the higher charge will have higher polarizing power. Hence, Pentahalides are more covalent than trihalides.
Note: Polarizing power is the ability of a cation to distort an anion.
New Question
11 months agoContributor-Level 10
Amity University Kolkata provides good placements to its graduating students. The students are given excellent placement support every year placing them in top companies. Check out the table below to know the key highlights of the Amity University Kolkata placements for BTech CSE:
Particulars | Placement Statistics (2023) |
|---|---|
the highest Package | INR 32.21 LPA |
Top Recruiters | Amazon, TCS, Cognizant, PWC, etc. |
Note- The institution has not released the placement report for 2024, once released it will be updated here.
New Question
11 months agoContributor-Level 10
Yes, MBBS course is available at Swaminarayan University. The university offers MBBS courses at the UG level. Candidate must meet the eligibility criteria to enrol for MBBS course admission set by the college. Aspirants must pass Class 12 with a 50% aggregate. Swaminarayan University's admission process is entrance-based.
New Question
11 months agoContributor-Level 10
Amity University, Kolkata is known for its excellent placement record. Check out the tabulated data given below to know more about Amity University, Kolkata recent placements for BTech CSE:
Particulars | Placement Statistics (2023) |
|---|---|
the highest Package | INR 32.21 LPA |
Top Recruiters | Amazon, TCS, Cognizant, PWC, etc. |
Note- The institution has not released the placement report for 2024, once released it will be updated here.
New Question
11 months agoContributor-Level 10
The fee range for the MBBS programme at Swaminarayan University is INR 55 lakh. This information is sourced from official website/ sanctioning body and is subject to change. The fee covers various academic expenses like tuition fees, laboratory charges, examination fees, library fees, and other relevant charges. It is important to note that the fee structure is subject to change, and candidates are advised to refer to the official website of the university for the latest and most accurate fee structure.
New Question
11 months agoNew Question
11 months agoGuide-Level 12
Pursuing an MA in Sociology offers numerous benefits, both academically and professionally. It provides you with a deep knowledge of social structures, relationships, and patterns and gives you a unique lens to view and understand the complexities of human societies.
Here are key reasons why sociology is a valuable field to pursue:
Sociology gives you an in-depth understanding of society where you learn how societies function, their impact, and their relationships with each other.
Sociology gives an idea of international problems and helps governments promote the welfare of tribal and marginalized communities.
It also enhances your skills
New Question
11 months agoContributor-Level 10
Yes, PES University offers BCom and BCom (H) courses of three-year duration. The university also provides a BCom evening programme. Admission in the UG course is entrance-based. Selection is done on the basis of performance in PESSAT. The entrance exam is conducted by the university. Before applying for admission, aspirants must check that they meet the course-specific eligibility criteria.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
8.15 For answering this question, we can compare the electronic configuration of standard elements and then write their corresponding oxidation
S. No | Electronic configurations in ground state | Stable oxidation states |
1 | 3d3 Vanadium | +2, +3, +4, +5 |
2 | 3d5 Chromium | +3, +4, +6 |
3 | 3d5 Manganese | +2, +4, +6, +7 |
4 | 3d8 Nickel | +1, +2, +3, +4 |
5 | 3d4 | 3d4 configuration is not stable at ground state |
New Question
11 months agoContributor-Level 10
There are several factors that candidates can consider while comparing the BBA course of Dr. C.V. Raman University with other universities and colleges. Firstly, candidates are suggested to identify their concern; for example, if fees are the concern, then candidates must compare the colleges on the basis of their fee structure and affordability. In the same way, candidates can compare the colleges and institutions on the basis of various factors:
- Shiksha Rating
- Location
- Seats
- Cutoff
- Admission Criteria, etc.
New Question
11 months agoContributor-Level 10
The best test for distinguishing methyl amine and dimethylamine is the Carbylamines test.
Carbylamine Test: Aliphatic and aromatic primary amines on heating with chloroform and ethanolic potassium hydroxide form foul-smelling isocyanides or carbylamines.
In this case, Methylamine (which is an aliphatic primary amine) gives a positive carbylamine test while dimethylamine wont.
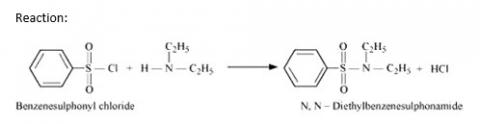
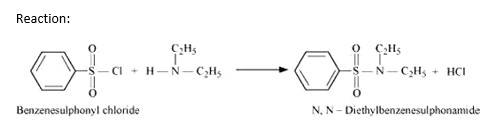
Hinsberg's reagent (benzenesulphonyl chloride, C6H5SO2Cl). can be used to distinguish secondary and tertiary amines.
Hinsberg Test: Secondary amines react with Hinsberg's reagent to form a product that is insoluble in an alkali. For example, N, N? diethylamine reacts
New Question
11 months agoContributor-Level 10
8.14 The elements in the first-half of the transition series exhibit many oxidation states with Mn exhibiting a maximum number of oxidation states (+2 to +7). The stability of +2 oxidation state increases with the increase in atomic number. This happens as more electrons are getting filled in the d-orbital.
However, Sc ( [Ar] 3d14s2) does not show +2 oxidation state, instead, it loses all the three valence electrons to form Sc3+. The +3 oxidation state of Sc is very stable as it attains stable configuration.
For Mn ( [Ar] 3d54s2), +2 oxidation state is very stable because after losing two electrons, it attains stable half-filled str
New Question
11 months agoContributor-Level 10
1 - Methylethanamine
The root name is based on the longest chain with the -NH2 attached. The chain is numbered so as to give the amine unit the lowest possible number. The longest chain is ethane chain which is further suffixed with ‘amine’.
2 - Propan-1-amine
The longest chain here is propane. The naming is such that amine unit should get a the lowest possible number. Propane-1-amine can also be written as 1-propylamine.
3 - N−Methyl-2-methylethanamine
The chain is numbered so as to give the amine unit the lowest possible number. The other alkyl group is treated as a substituent, with N as the locant. The N locant is list
New Question
11 months agoContributor-Level 10
5.35
We can say that colloid is not a substance but a state of a substance which is dependent on the size of particle colloidal state is intermediate between a true solution and a suspension.
When a size of substance is between 1nm to1000nm it behaves as colloid otherwise not.
New Question
11 months agoContributor-Level 10
To get into Swaminarayan University for an MBA course, candidates must meet the eligibility criteria set by the university. Students must pass graduation with a minimum of 50% aggregate. Swaminarayan University offers full-time MBA courses of two years' duration. Students can get into this course based on merit. Apart from the selection criteria, students must pass graduation (common eligibility).
New Question
11 months agoContributor-Level 10
Hi, as far as my research goes, there are around 1466 MA Sociology colleges in India. Out of these, 509 colleges are government entities & 632 are private institutions.
Read more: M.A. Sociology Colleges in India
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts