Ask & Answer: India's Largest Education Community
All Questions
New Question
11 months agoContributor-Level 10
The candidates who are Class 12 or equivalent exam pass can apply for a BCom/ BCom (H) course of Cluster University Srinagar. Additionally, the qualifying exam should have been passed from a recognised board and with minimum aggregate as prescribed by the university. During form filling and admission, the aspirants must hold the relevant documents for verification purposes.
New Question
11 months agoContributor-Level 10
4.22 Half life of first order reaction is, t1/2 = ln2/K where t1/2 is half life of first order reaction, K is rate constant of First order reaction.
(i) t1/2 = ln2/200 s-1
⇒t1/2 = 0.693/200 s-1 (? ln2 = 0.693)
⇒t1/2 = 0.003465 sec.
(ii) t1/2 = ln2/2 min-1
⇒t1/2 = 0.693/2 min-1 (? ln2 = 0.693)
⇒t1/2 = 0.3465 min
(iii) t1/2 = ln2/4 year-1
⇒t1/2 = 0.693/4 years -1 (? ln2 = 0.693)
⇒t1/2 = 0.17325 year.
Half life of 3 reactions are 0.003465 sec, 0.3465 min, 0.17325 year, respectively.
New Question
11 months agoContributor-Level 10
4.21 As reaction is first order with respect to A and zero Order with respect to B. Then changing the concentration of B won't affect the rate of reaction and increasing concentration of A 'n' times will increase the rate by 'n' times. By this logic lets fill the table- In first blank space concentration of A will be 0.2 mol L-1 because the rate is doubled. In second blank space, Rate will be 8 * 10-2mol L-1min-1 because the concentration of A is increased 4 Times. In third blank space concentration of A will be 0.1 mol L-1 because the rate is same as in experiment I.
Experiment | [A]/mol L-1 | [B]/mol L-1 | Initial rate/mol L-1 min-1 |
I | 0.1 | 0.1 | 2.0 * 10-2 |
II | 0.2 | 0.2 | 4.0 * 10-2 |
III | 0.4 | 0.4 | 8.0 * 10-2 |
IV | 0.1 | 0.2 | 2.0 * 10-2 |
New Question
11 months agoContributor-Level 10
Repeating connectors can lower your lexical resource score in IELTS exam. So, candidates should use synonyms or related linking phrases to maintain variation.
New Question
11 months agoContributor-Level 10
One should aim for using 5–8 connectors per report. Candidates should focus on quality and variety rather than quantity.
New Question
11 months ago
Contributor-Level 10
As per the latest report, 17 BPharma students were placed during the 2024 placement drive at Guru Nanak College Dehradun. Earlier, 29 students of the BPharma batch 2024 were placed in reputed companies. The top recruiters of Guru Nanak College Dehradun in 2024 included NECLIFE, Windlas Biotech Limited, and Swarnimaksh Life Sciences.
New Question
11 months agoContributor-Level 10
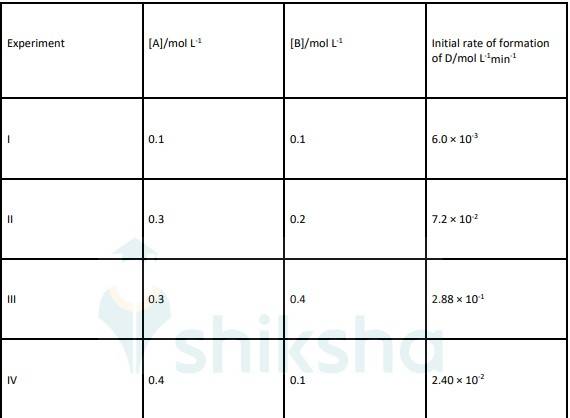
4.20 By comparing Experiment I and IV if we increase the concentration of A by 4 times then Rate also increased by 4 times. That means order with respect to A is 1.
By comparing Experiment II and III if we double the concentration of B Rate increases by 4 times that means order with respect to B is 2.
Rate law of reaction will be, Rate = k [A] [B]2
To find K, K = rate/ [A] [B]2 i.e. K = 6.0 * 10-3/ [0.1] [0.1]2
K = 6 mol-2L2sec-1
Order with respect to A and B is 1 and 2 respectively. And value of K (rate constant) is = 6 mol-2L2sec-1
New Question
11 months agoContributor-Level 10
CUET UG scores are not compulsory to get admissions into the BBA course at Dr. CV Raman University. However, the university is one of the universities that considers CUET UG scores for admission to its several undergraduate courses. Therefore, candidates with CUET UG scores can get admission on the basis of their secured scores. Moreover, the university also grants merit-based admissions after evaluating candidates' Class 12 scores.
New Question
11 months agoContributor-Level 10
The below-mentioned steps to complete MKU DDE admission procedure:
1: Apply for the course of choice.
2: Fill out the form
3: Submit the required documents.
4: Shortlisted candidates will receive a letter via mail.
5: Pay the course admission fees.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
4.19 When concentration of B is changed then rate of reaction doesn't change that means order with respect to B is 0. But when the concentration of A is doubled rate increased by 2.82 times i.e.21.5 = 2.82.Hence order with respect to A is 1.5.
Order with respect to A and B is 1.5 and 0, respectively.
New Question
11 months agoBeginner-Level 4
Crescent Institute of Science and Technology offers Work-Integrated Learning (WIL) programs aimed at bridging the gap between academic knowledge and industry needs. These programs are developed in collaboration with leading industry partners, notably IBM, to provide students with hands-on experience, practical skills, and exposure to real-world challenges.
A key highlight is the IBM Software Lab for Emerging Technologies on campus, where students gain access to IBM's advanced software, specialised curriculum, and mentorship opportunities. Specializations in Artificial Intelligence & IoT and Big Data Analytics under the B.Tech CSE progra
New Question
11 months agoNew Question
11 months agoContributor-Level 10
4.18 (i) Order is power raised to reactant in rate law, hence,
Rate = k [A] [B]2
(ii) When the concentration of B is increased three times then the rate is affected by the square of The rate is increased by 9 Times.
(iii) When the concentration of reactant both A and B is doubled then the rate will have affected as square of reactant B and Two times of Reactant Overall increase in rate is 8 times
(a) When the concentration of B is increased by three times, the rate is increased by nine times
(b) When of both reactants is doubled, the Rate increases 8 times.
New Question
11 months agoContributor-Level 10
4.17 (i) Average rate of reaction over interval is [change in concentration]/ [time taken] e.
[0.31 - 0.17] / [60-30] = 0.00467 mol L-1 sec-1
(ii) the pseudo first-order rate constant can be calculated by K = (2.303/t) log (Ci/Ct)
where K is Rate constant,
t is time taken,
Ci is initial concentration
Ct is Concentration at time t.
K = (2.303/30) log (0.55/0.31)
? K = 1.9 * 10-2 sec-1
(i) Average rate between 30 to 60 sec is 0.00467 mol L-1sec-1
(ii) Pseudo first order rate constant is 1.* 10-2sec-1
New Question
11 months agoContributor-Level 10
Yes, admissions are open for the BBA course at Dr. C.V. Raman University Bihar for the academic batch of 2025. Interested candidates can apply through the official website of the university. Moreover, before applying for the course, candidates must ensure that they meet the basic eligibility criteria required by the university to get enrolled in the BBA course. The selection process involves acceptance of CUET and release of merit on the basis of candidates' achieved scores in their higher senior secondary.
New Question
11 months agoNew Question
11 months agoContributor-Level 10
The BA specialisations offered at St. Xavier's College Goa are as follows:
B.A. in Mass Communication
B.A. in Journalism
B.A. in Sociology
Candidates are required to follow the merit based admission process to get a chance at securing admission to their desired programme. Candidates can choose from the specialisations while filling out the application form of the college.
New Question
11 months agoContributor-Level 10
5.7
According to Hardy-Schulze law
'The greater the valence of the flocculating ion added, the greater is its power to cause precipitation.
As this law takes into consideration of only the charge present on the ion and not the size of the ion. So when the size of the atom is considered, smaller the size of an atom more will be its polarising power.
So Hardy-Schulze can be modified in terms of the polarising power of the flocculating ion as 'The greater the polarising power of the flocculating ion added, the greater is its power to cause precipitation.
New Question
11 months agoContributor-Level 10
Nagaland University – Kohima Campus offers one of the most affordable MBA programmes in the region, with a total tuition fee of just INR 30,800 for the entire two-year course. In comparison, ICFAI University charges around INR 3.3 lakh, and St. Joseph University's fee is about INR 1.2 lakh, making Nagaland University a cost-effective option for students seeking quality education at a lower cost.
Register to get relevant
Questions & Discussions on your feed
Ask & Answer
Panel of Experts